Advanced Imidocarb Manufacturing: High-Purity Veterinary Intermediate Production via Green Catalysis
Advanced Imidocarb Manufacturing: High-Purity Veterinary Intermediate Production via Green Catalysis
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical active ingredients and intermediates. Patent CN102924380A introduces a transformative preparation method for Imidocarb, a vital antiprotozoal agent used extensively in veterinary medicine to treat babesiosis and trypanosomiasis. This technology represents a significant leap forward in process chemistry, shifting away from hazardous, waste-intensive traditional methods toward a streamlined, catalytic approach. By leveraging phosphorus-molybdenum-vanadium (P-Mo-V) heteropolyacids and palladium-catalyzed hydrogenation, the process achieves exceptional purity levels exceeding 99% and molar yields stabilized above 71%. For global procurement leaders and R&D directors, this patent outlines a pathway to secure a reliable veterinary drug intermediate supplier capable of meeting stringent quality standards while drastically reducing the environmental footprint associated with large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Imidocarb and its derivatives has been plagued by significant operational and environmental challenges that hinder cost-effective production. Traditional routes, such as those described in US Patent No. 3338917 and Chinese patent CN101348465A, often rely on the use of phosgene or triphosgene for urea formation, posing severe safety risks due to the extreme toxicity of these reagents. Furthermore, the reduction of nitro-intermediates in older processes frequently utilizes iron powder in acidic media, a method that generates massive quantities of iron mud sludge. This solid waste is not only difficult to separate from the product liquor but also requires expensive and complex disposal procedures to meet environmental regulations. Additionally, these legacy methods often suffer from poor reaction control, leading to inconsistent yields typically hovering around 65%, which necessitates larger reactor volumes and higher raw material consumption to meet production targets, thereby inflating the overall cost of goods sold.
The Novel Approach
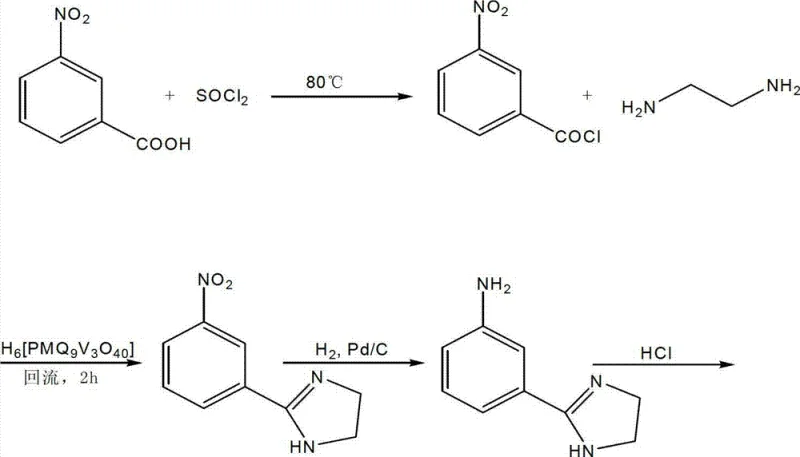
In stark contrast, the methodology disclosed in CN102924380A offers a sophisticated, multi-step synthesis that prioritizes atom economy and operational safety. The process initiates with the conversion of m-nitrobenzoic acid to m-nitrobenzoyl chloride using thionyl chloride, followed by a novel cyclization step utilizing ethylenediamine and a reusable P-Mo-V heteropolyacid catalyst. This catalytic system facilitates the formation of the imidazoline ring under mild reflux conditions in acetonitrile, avoiding the harsh conditions of previous methods. Crucially, the reduction step employs catalytic hydrogenation with palladium on carbon (Pd/C) instead of iron powder, completely eliminating the generation of heavy metal sludge. The final condensation with urea in dimethylformamide (DMF) proceeds with high efficiency, yielding a white solid product with a melting point of 350-352°C and content greater than 99%. This holistic redesign of the synthetic route ensures a cleaner, faster, and more economically viable production cycle suitable for modern industrial demands.
Mechanistic Insights into P-Mo-V Heteropolyacid Catalyzed Cyclization
The core innovation of this patented process lies in the strategic application of phosphorus-molybdenum-vanadium heteropolyacids during the cyclization of m-nitrobenzoyl chloride with ethylenediamine. Heteropolyacids are known for their strong Brønsted acidity and redox properties, which act synergistically to activate the carbonyl group of the acid chloride towards nucleophilic attack by the diamine. Unlike traditional Lewis acids that may form stable complexes with the product and require aqueous workups that generate wastewater, the P-Mo-V catalyst in this system demonstrates excellent stability in organic solvents like acetonitrile. The mechanism likely involves the protonation of the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon, thereby facilitating the rapid formation of the amide bond and subsequent intramolecular cyclization to form the 2-(3-nitrophenyl)imidazoline ring. This catalytic efficiency allows the reaction to proceed at temperatures between 75°C and 85°C with short reaction times of 1.5 to 2.5 hours, significantly enhancing throughput compared to non-catalytic thermal cyclizations.
Furthermore, the integration of palladium-catalyzed hydrogenation in the subsequent step provides a mechanistic advantage regarding impurity control and product isolation. In the reduction of the nitro-imidazoline intermediate, the use of Pd/C under hydrogen pressure (0.01-0.95 MPa) ensures a highly selective reduction of the nitro group to the amine without affecting the sensitive imidazoline ring structure. This selectivity is critical because over-reduction or ring-opening side reactions are common pitfalls in the synthesis of such heterocycles. The process includes a precise pH adjustment step (pH 4.0-8.0) prior to crystallization, which effectively removes residual catalyst and byproducts, ensuring the resulting 2-(3-aminophenyl)imidazoline hydrochloride salt is of high purity. This rigorous control over the reduction environment minimizes the formation of azo or hydrazo impurities, which are difficult to remove in later stages, thereby guaranteeing the high purity (>99%) of the final Imidocarb API intermediate.
How to Synthesize Imidocarb Efficiently
The synthesis of Imidocarb via this patented route requires precise control over reaction parameters to maximize yield and safety. The process is divided into four distinct operational units: acylation, catalytic cyclization, hydrogenation, and condensation. Each step has been optimized in the patent embodiments to demonstrate scalability from laboratory to pilot plant levels. Operators must pay close attention to temperature gradients during the exothermic acylation with thionyl chloride and maintain strict hydrogen pressure controls during the reduction phase to ensure safety. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to implement this superior manufacturing protocol.
- React m-nitrobenzoic acid with thionyl chloride at 75-85°C to form m-nitrobenzoyl chloride.
- Cyclize m-nitrobenzoyl chloride with ethylenediamine using P-Mo-V heteropolyacid catalyst in acetonitrile.
- Reduce the nitro-imidazoline intermediate using Pd/C catalyst under hydrogen pressure (0.01-0.95MPa).
- Condense the resulting amine salt with urea in DMF at 155-165°C to obtain high-purity Imidocarb.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN102924380A process translates directly into tangible operational efficiencies and risk mitigation. The elimination of iron powder reduction removes a major bottleneck in waste management, significantly reducing the logistical burden and cost associated with hazardous sludge disposal. Moreover, the ability to recycle key reagents such as acetonitrile, thionyl chloride, and the heteropolyacid catalyst creates a closed-loop system that minimizes raw material consumption. This circular approach to manufacturing not only lowers the variable cost per kilogram but also insulates the supply chain from volatility in raw material pricing. The simplified post-processing operations, characterized by straightforward filtration and crystallization steps, reduce the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The transition from stoichiometric iron reduction to catalytic hydrogenation fundamentally alters the cost structure of Imidocarb production. By removing the need for large quantities of iron powder and the associated acid waste neutralization chemicals, the process drastically cuts down on consumable costs. Additionally, the high molar yield stabilized above 71% means that less starting material is required to produce the same amount of final product compared to the 65% yield of older methods. This improvement in mass efficiency directly correlates to a lower cost of goods sold, providing a competitive pricing advantage in the global veterinary pharmaceutical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the complexity of the manufacturing process. Traditional methods involving iron mud often face production delays due to filtration clogging and equipment fouling, which can disrupt delivery schedules. The new method utilizes homogeneous or easily filterable heterogeneous catalysts that maintain reactor efficiency over long campaigns. Furthermore, the use of commercially available and stable reagents like m-nitrobenzoic acid and urea ensures that raw material sourcing remains secure and unaffected by niche supply constraints. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery requirements of downstream formulation manufacturers.
- Scalability and Environmental Compliance: As regulatory pressures on chemical manufacturing intensify globally, the eco-friendly nature of this process offers a significant strategic advantage. The absence of phosgene and iron sludge aligns the production facility with stringent environmental, health, and safety (EHS) regulations, reducing the risk of regulatory shutdowns or fines. The process is inherently designed for scale-up, with reaction conditions that are easily manageable in large stainless steel reactors. The ability to recycle solvents and catalysts further reduces the facility's total waste output, supporting corporate sustainability goals. This compliance readiness ensures long-term operational viability and protects the brand reputation of partners relying on this supply chain for critical veterinary medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Imidocarb synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in patent CN102924380A, providing clarity on yield expectations, purity profiles, and environmental benefits. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term value proposition of this manufacturing route.
Q: What is the primary advantage of the P-Mo-V heteropolyacid catalyst in Imidocarb synthesis?
A: The P-Mo-V heteropolyacid catalyst enables a highly efficient cyclization step with easy separation and recyclability, significantly reducing waste compared to traditional Lewis acids.
Q: How does the new hydrogenation method improve environmental compliance?
A: By replacing iron powder reduction with Pd/C catalytic hydrogenation, the process eliminates the generation of toxic iron mud sludge, simplifying wastewater treatment and lowering disposal costs.
Q: What purity levels can be achieved with this patented Imidocarb process?
A: The optimized process consistently achieves liquid phase purity greater than 99%, with molar yields stabilized above 71%, surpassing previous industry benchmarks of 65%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidocarb Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of effective veterinary therapeutics. Our team of expert chemists has thoroughly analyzed the CN102924380A patent and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this advanced Imidocarb process to life. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the >99% purity benchmark established by this innovative technology. Our state-of-the-art facilities are equipped to handle the specific catalytic hydrogenation and heteropolyacid recycling steps safely and efficiently, ensuring a consistent supply of this vital intermediate.
We invite global partners to collaborate with us to leverage this superior synthetic route for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of Imidocarb that drives your veterinary product portfolio forward.
