Scalable Low-Temperature Enzymatic Synthesis for High-Purity Cephalosporin Intermediates
Scalable Low-Temperature Enzymatic Synthesis for High-Purity Cephalosporin Intermediates
The pharmaceutical industry continuously seeks robust methodologies for producing beta-lactam antibiotics with superior purity profiles and reduced environmental footprints. Patent CN1063491C introduces a transformative approach to the preparation of cephalosporins, specifically leveraging immobilized penicillin acylase under rigorously controlled low-temperature conditions. Unlike traditional chemical acylation methods that often require harsh solvents and multiple protection-deprotection steps, this biocatalytic route operates efficiently in aqueous media at temperatures ranging from 0°C to +20°C. The innovation lies not merely in the enzyme selection but in the synergistic combination of低温 (low temperature), ambient pH maintenance, and a high molar ratio of substrates. This triad of conditions effectively mitigates the formation of hydrolytic byproducts that typically plague enzymatic syntheses conducted at physiological temperatures. For R&D directors and process chemists, this patent represents a significant leap forward in achieving high-yield condensation of 7-amino cephalosporanic acid nuclei with alpha-amino acid derivatives, ensuring a cleaner impurity profile essential for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, enzymatic synthesis of cephalosporins, as described in earlier literature such as United States Patent 3,816,253, relied heavily on operating temperatures between 20°C and 40°C, with 37°C being the standard. While enzymes function optimally at these physiological temperatures regarding their intrinsic catalytic rate, the thermodynamic landscape favors unwanted side reactions. Specifically, the hydrolysis of the activated amino acid derivative and the subsequent hydrolysis of the formed cephalosporin product become kinetically competitive at these elevated temperatures. This results in a significant loss of yield, often capping conversion rates around 63% even under optimized conditions. Furthermore, the necessity to maintain a constant pH using buffered solutions adds substantial complexity to the downstream processing. The removal of buffer salts and the separation of hydrolytic byproducts increase the operational expenditure and generate considerable aqueous waste. Consequently, despite the theoretical elegance of biocatalysis, the economic viability of these conventional enzymatic routes has often been inferior to classical organic chemistry methods, limiting their adoption in large-scale commercial manufacturing of pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN1063491C fundamentally alters the reaction paradigm by shifting the operating window to a cryogenic range of 0°C to +20°C, preferably 0°C to 5°C. This counter-intuitive strategy capitalizes on the differential activation energies of the desired condensation reaction versus the undesired hydrolysis pathways. By drastically lowering the thermal energy of the system, the rate of hydrolytic degradation is suppressed far more significantly than the rate of the enzyme-catalyzed acylation. To compensate for the natural reduction in enzyme activity at these lower temperatures, the process employs a high molar ratio of the alpha-amino acid derivative to the cephalosporin nucleus, typically between 3:1 and 5:1. This mass action effect drives the equilibrium toward the product. Additionally, the process eliminates the need for rigid pH control, allowing the reaction to proceed at ambient pH values which naturally drift from approximately 7.0 to 6.0. This simplification removes the requirement for expensive buffering agents. The reaction involves the condensation of a 7-amino cephalosporanic acid derivative (Formula II) with an alpha-amino acid derivative (Formula III), as illustrated below:
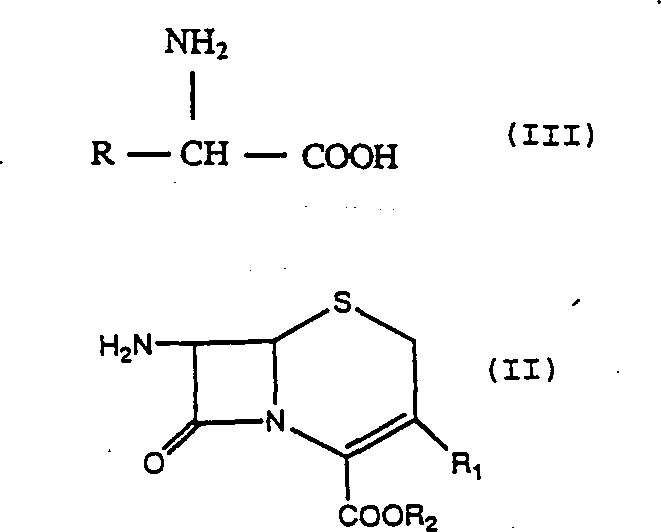
This streamlined approach not only enhances the chemical yield but also drastically simplifies the isolation of the final cephalosporin product, making it highly attractive for industrial scale-up.
Mechanistic Insights into Immobilized Penicillin Acylase Catalysis
The core of this improved process relies on the precise interaction between the immobilized penicillin acylase and the substrates within the active site of the enzyme. The mechanism proceeds through the formation of a covalent acyl-enzyme intermediate. Initially, the alpha-amino acid derivative, which acts as the acyl donor, binds to the serine residue within the enzyme's active center, releasing the leaving group (typically an alcohol from the ester). This forms a stable acyl-enzyme complex. Subsequently, the nucleophilic attack by the 7-amino group of the cephalosporin nucleus (Formula II) on this acyl-enzyme intermediate results in the formation of the new amide bond and the release of the free enzyme. The structural integrity of the cephalosporin product (Formula I) is maintained throughout this mild enzymatic process, preserving the sensitive beta-lactam ring which might otherwise be susceptible to chemical degradation under harsher synthetic conditions. The general structure of the resulting cephalosporin derivatives is depicted below, where R represents various substituted hydrocarbon or heterocyclic rings, allowing for the synthesis of a diverse library of antibiotic precursors.
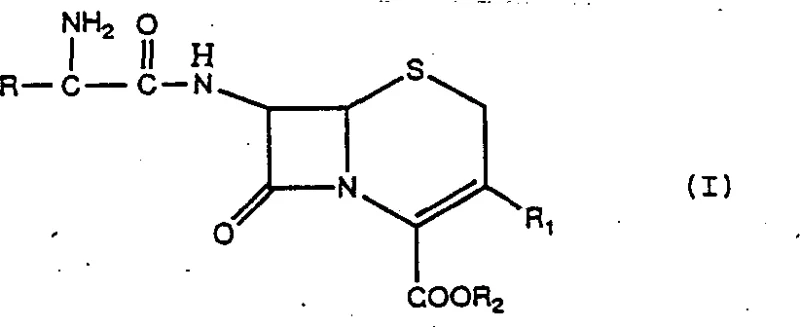
Crucially, the immobilization of the enzyme on supports such as cellulose triacetate or polyacrylamide resin enhances its stability against the low-temperature shock and allows for repeated reuse, further driving down the cost per kilogram of the product. The enzyme remains heterogeneous, facilitating easy filtration and recovery from the reaction mixture without the need for complex protein precipitation steps.
From an impurity control perspective, the low-temperature operation is the single most critical factor in defining the purity profile of the final API intermediate. At conventional temperatures (e.g., 37°C), water molecules compete effectively with the 7-amino cephalosporanic acid for the acyl-enzyme intermediate, leading to the regeneration of the free amino acid and the hydrolysis of the product back to the starting nucleus. By operating at 0°C to 5°C, the kinetic barrier for this hydrolytic pathway is raised significantly. Experimental data within the patent indicates that while the reaction rate is slower, the selectivity for amide bond formation over hydrolysis is markedly improved. This results in a reaction mixture where the desired cephalosporin constitutes a much higher percentage of the total organic content, reducing the burden on downstream purification columns. Furthermore, the absence of buffering salts prevents the formation of inorganic impurities that can be difficult to remove to the stringent levels required for parenteral antibiotics. This mechanistic understanding assures quality assurance teams that the process is inherently robust against the formation of hard-to-remove genotoxic or structurally related impurities.
How to Synthesize 7-ACCA Derivatives Efficiently
The practical implementation of this patented technology involves a straightforward sequence of unit operations that are easily adaptable to existing stainless steel reactor trains. The process begins with the dissolution of the 7-amino cephalosporanic acid substrate, such as 7-ACCA, in water or a water-miscible solvent system. The pH is initially adjusted to neutrality using ammonium hydroxide. Following this, the alpha-amino acid ester, such as D-phenylglycine methyl ester, is added in a substantial molar excess. The mixture is then cooled to the target range of 0°C to 5°C using standard glycol jackets or ice baths. Once thermal equilibrium is reached, the immobilized enzyme is introduced. Unlike traditional methods, no further pH adjustment is required; the system is allowed to self-regulate as the reaction progresses. Monitoring is typically performed via HPLC to determine the point of maximum conversion, which usually occurs within a few hours depending on the enzyme loading. The specific reaction pathway for a preferred embodiment involving 7-ACCA and D-phenylglycine methyl ester is shown below:
- Prepare an aqueous solution of the 7-amino cephalosporanic acid substrate (Formula II) and adjust the initial pH using ammonium hydroxide.
- Add the alpha-amino acid derivative (Formula III) in a high molar ratio (3: 1 to 6:1) relative to the cephalosporin nucleus.
- Cool the mixture to 0°C to 5°C, add immobilized penicillin acylase, and maintain ambient pH without buffering agents until maximum conversion is reached.
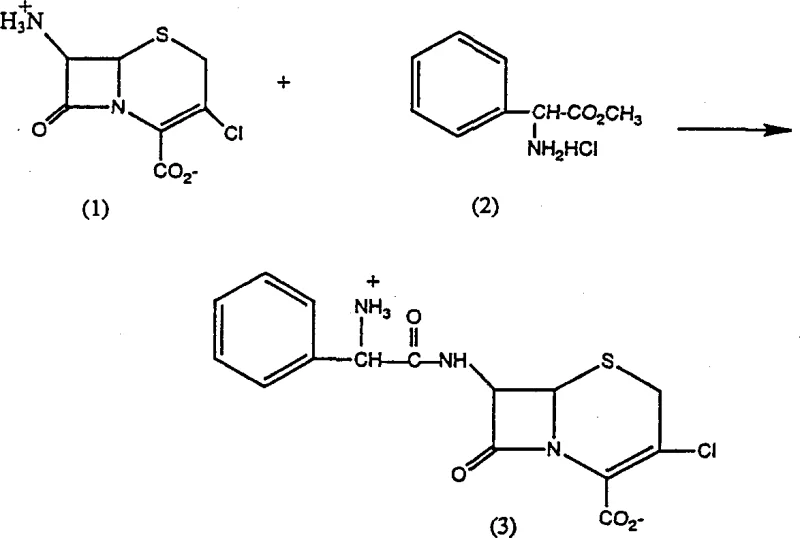
Upon reaching maximum yield, the immobilized enzyme is simply filtered off, ready for regeneration and reuse, while the filtrate containing the product can be directly subjected to crystallization or extraction. Detailed standardized synthesis steps are provided in the guide below.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this low-temperature enzymatic process offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary value driver is the drastic simplification of the raw material list and the reduction in processing time. By eliminating the need for complex organic solvents like pyridine and expensive buffering agents, the direct material costs are significantly lowered. Furthermore, the ability to recover and reuse the immobilized enzyme multiple times transforms the enzyme from a consumable reagent into a durable capital asset, amortizing its cost over tons of production. This shift fundamentally alters the cost structure of cephalosporin intermediate manufacturing, making the enzymatic route commercially competitive with, and often superior to, traditional chemical synthesis. The reduced formation of byproducts also translates to higher throughput in purification units, effectively increasing the capacity of existing manufacturing facilities without the need for capital expansion.
- Cost Reduction in Manufacturing: The elimination of stoichiometric coupling reagents and protecting group chemistry traditionally used in organic synthesis leads to substantial cost savings. In the enzymatic route, the only reagents consumed are the substrates and a catalytic amount of enzyme, whereas chemical routes often require expensive activating agents and generate stoichiometric amounts of salt waste. The low-temperature operation also reduces the energy load associated with heating and cooling cycles, as the reaction is exothermic and requires only maintenance of a cool environment rather than active heating. Additionally, the simplified workup procedure, which avoids complex extractions to remove buffers, reduces solvent consumption and waste disposal costs. These factors combine to create a leaner, more cost-effective manufacturing process that improves the overall margin profile for the final pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Reliance on fewer and more commodity-grade raw materials enhances supply chain resilience. The key substrates, 7-ACCA and amino acid esters, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the immobilized enzyme means that production campaigns can run for extended periods without the need for frequent catalyst replenishment, ensuring consistent batch-to-batch quality and reliable delivery schedules. The process tolerance to ambient pH fluctuations further reduces the risk of batch failures due to minor deviations in raw material quality or operator error. This operational stability is crucial for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages and ensuring that inventory levels remain optimized to meet market demand without excessive safety stock.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. By avoiding chlorinated solvents and heavy metal catalysts, the process generates a waste stream that is easier and cheaper to treat. The high atom economy of the enzymatic condensation ensures that the majority of the input mass ends up in the desired product, minimizing waste generation at the source. Scalability is straightforward because the heat transfer requirements are modest (cooling only) and the reaction kinetics are not limited by mass transfer issues common in heterogeneous chemical catalysis. This allows for seamless scale-up from pilot plant to multi-ton commercial production, enabling rapid response to market surges in antibiotic demand while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic cephalosporin synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process feasibility. Understanding these nuances is essential for technical teams assessing the integration of this route into their existing manufacturing portfolios. The answers highlight the balance between reaction kinetics, thermodynamic control, and practical operational parameters that define the success of this method.
Q: Why is low temperature critical in this enzymatic cephalosporin synthesis?
A: Operating at 0°C to 20°C significantly suppresses the competitive hydrolysis of the amino acid derivative and the final product, which is a major side reaction at conventional temperatures like 37°C, thereby improving yield and purity.
Q: What are the advantages of using ambient pH conditions?
A: Maintaining ambient pH eliminates the need for expensive and complex buffer systems. The process allows the pH to drift naturally (typically from 7.0 down to 6.0), simplifying the workup and reducing chemical waste.
Q: How does the high molar ratio of amino acid affect the reaction?
A: Using a high molar excess of the alpha-amino acid derivative (3 to 5 equivalents) drives the equilibrium towards the desired amide bond formation, compensating for the reduced enzyme activity at lower temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalosporin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN1063491C to maintain competitiveness in the global pharmaceutical market. Our R&D team has extensively validated similar enzymatic platforms, demonstrating our capability to translate complex patent methodologies into robust, GMP-compliant manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of low-temperature enzymatic synthesis are fully realized at an industrial scale. Our state-of-the-art facilities are equipped with precise temperature control systems and rigorous QC labs capable of monitoring the subtle impurity profiles associated with beta-lactam synthesis. We are committed to delivering high-purity cephalosporin intermediates that meet the stringent specifications required by top-tier generic and innovator drug companies worldwide.
We invite you to collaborate with us to leverage this technology for your next project. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this enzymatic route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our cephalosporin portfolio and to discuss route feasibility assessments for your target molecules. Let us partner with you to drive efficiency and quality in your antibiotic supply chain.
