Advanced Synthesis of 4-Chloro-3-Nitropyridine for High-Purity Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for key heterocyclic building blocks, and patent CN112707860A presents a significant advancement in the production of 4-chloro-3-nitropyridine. This active intermediate serves as a critical precursor for synthesizing anti-peptic ulcer drugs such as pirenzepine and acts as a vital component in leukotriene biosynthesis inhibitors. Furthermore, its utility extends into the agrochemical sector as a specialized insecticide intermediate, highlighting its dual-market value. The disclosed methodology addresses longstanding challenges associated with product stability and purification efficiency, offering a streamlined pathway that transitions from a fragile nitropyridine derivative to a stable chlorinated final product. By leveraging a controlled nitration followed by a rigorous chlorination protocol, this technology ensures consistent quality metrics essential for downstream API synthesis. For procurement specialists and R&D teams alike, understanding this patented approach is crucial for securing a reliable supply chain of high-purity pharma intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-chloro-3-nitropyridine has relied on direct chlorination strategies starting from 3-nitropyridine, typically employing hydrogen peroxide and hydrochloric acid as the oxidizing and chlorinating agents. While conceptually simple, this conventional route is plagued by severe operational drawbacks that hinder large-scale manufacturing efficiency. The reaction environment is notoriously aggressive, leading to the generation of substantial quantities of unidentified byproducts that complicate the isolation of the target molecule. Moreover, the resulting product exhibits poor chemical stability, often undergoing decomposition during the necessary post-treatment and purification stages. This instability not only drives down the overall yield but also necessitates complex and costly purification protocols to meet the stringent purity specifications required for pharmaceutical applications. Consequently, the traditional method fails to provide a cost-effective solution for the commercial scale-up of complex pyridine derivatives, creating bottlenecks in the supply of critical drug intermediates.
The Novel Approach
In stark contrast, the innovative method detailed in the patent introduces a two-step sequence that fundamentally alters the reaction landscape to favor stability and yield. The process initiates with the nitration of a hydroxypyridine precursor using a concentrated sulfuric acid and potassium nitrate system, carefully managed at temperatures between 70-80°C to maximize regioselectivity. This first stage yields 4-hydroxy-3-nitropyridine with an impressive 80% yield and 98% purity, establishing a high-quality foundation for the subsequent transformation. The second step involves a chlorination reaction utilizing phosphorus oxychloride in a toluene solvent system, refluxed at 110°C to drive the conversion to completion. This approach eliminates the instability issues associated with direct chlorination, as the intermediate is robust enough to withstand the reaction conditions and subsequent workup procedures involving ice water quenching and pH adjustment. 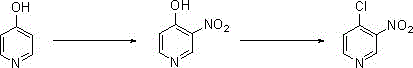
Mechanistic Insights into Nitration-Chlorination Sequence
The core of this synthetic breakthrough lies in the precise control of electrophilic aromatic substitution followed by nucleophilic displacement. In the first step, the hydroxyl group on the pyridine ring acts as a strong activating and ortho/para-directing group, facilitating the introduction of the nitro group at the 3-position relative to the nitrogen atom. The use of concentrated sulfuric acid serves a dual purpose: it acts as a dehydrating agent to generate the active nitronium ion from potassium nitrate and provides a polar medium that stabilizes the transition state. Maintaining the temperature at 70-80°C is critical; lower temperatures result in incomplete conversion, while higher temperatures risk poly-nitration or degradation of the sensitive pyridine ring. The subsequent isolation via pH adjustment to neutrality ensures that the zwitterionic nature of the hydroxy-nitropyridine is exploited for precipitation, effectively removing acidic impurities without the need for chromatographic separation.
The second mechanistic phase involves the conversion of the hydroxyl group to a chlorine atom using phosphorus oxychloride (POCl3), a classic reagent for activating hydroxyl groups in heterocyclic chemistry. The mechanism likely proceeds through the formation of a dichlorophosphate intermediate, which is then displaced by a chloride ion in a nucleophilic aromatic substitution manner, driven by the electron-withdrawing effect of the adjacent nitro group. This electronic activation makes the 4-position highly susceptible to nucleophilic attack, allowing the reaction to proceed efficiently under reflux conditions in toluene. The choice of toluene as a solvent is strategic, as it allows for high-temperature reflux (110°C) which provides the necessary activation energy for the displacement while remaining inert to the harsh chlorinating conditions. The final purification via extraction with petroleum ether leverages the solubility differences between the chlorinated product and polar byproducts, ensuring a final purity of 95% suitable for immediate use in coupling reactions.
How to Synthesize 4-Chloro-3-Nitropyridine Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful addition of raw materials to cooled concentrated sulfuric acid to manage the exotherm, followed by a controlled heating ramp to facilitate nitration. Operators must monitor the reaction progress via HPLC to confirm the complete consumption of the starting material before proceeding to the quenching phase, which involves pouring the mixture into ice water to precipitate the intermediate. The second stage demands anhydrous conditions for the POCl3 reaction, followed by a meticulous workup involving dichloromethane extraction and a critical cold wash with saturated sodium bicarbonate to neutralize residual acids without degrading the product.
- Nitration: React 4-hydroxypyridine with concentrated sulfuric acid and potassium nitrate at 70-80°C to form 4-hydroxy-3-nitropyridine (80% yield).
- Workup: Quench reaction in ice water, adjust pH to 7, and filter to obtain the light yellow solid intermediate.
- Chlorination: Reflux the intermediate with phosphorus oxychloride in toluene at 110°C, followed by extraction and purification to yield 4-chloro-3-nitropyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield, directly impacting the bottom line through operational efficiency and risk mitigation. The elimination of unstable reaction intermediates significantly reduces the risk of batch failures, which is a common cost driver in the manufacturing of specialty chemicals. By stabilizing the process through the isolation of a high-purity intermediate, manufacturers can decouple the two reaction steps, allowing for better inventory management and flexible production scheduling. This modularity means that the intermediate can be stockpiled during periods of low demand and converted to the final chlorinated product as needed, smoothing out supply fluctuations and ensuring continuity for downstream API production lines. Furthermore, the use of commodity chemicals like sulfuric acid and toluene reduces dependency on exotic or supply-constrained reagents, enhancing the resilience of the supply chain against market volatility.
- Cost Reduction in Manufacturing: The economic advantages of this process are primarily derived from the simplification of the purification workflow and the high efficiency of the reagents used. Unlike traditional methods that may require expensive chromatography or multiple recrystallizations to remove stubborn byproducts, this method relies on straightforward pH-controlled precipitation and liquid-liquid extraction. The high yield of the first step (80%) minimizes the amount of raw material wasted, directly lowering the cost of goods sold (COGS). Additionally, the ability to recover and potentially recycle solvents like toluene and dichloromethane further contributes to substantial cost savings in large-scale operations. By avoiding the use of precious metal catalysts or hazardous oxidants that require specialized disposal, the process also reduces environmental compliance costs and waste treatment fees.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the robustness of the reaction conditions and the availability of raw materials. The reagents employed, including concentrated sulfuric acid, potassium nitrate, and phosphorus oxychloride, are bulk industrial chemicals with stable global supply networks, reducing the risk of procurement delays. The process tolerance, demonstrated by the ability to achieve high purity even with slight variations in workup, ensures consistent output quality regardless of minor operational fluctuations. This consistency is vital for maintaining long-term contracts with pharmaceutical clients who require certified batches of intermediates. Moreover, the scalability of the process from 10L laboratory reactors to 50L pilot vessels indicates a clear path to multi-ton production without the need for re-engineering the core chemistry, guaranteeing that supply can meet surging demand.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern green chemistry principles by minimizing waste generation and maximizing atom economy in the key transformation steps. The aqueous workup in the first step generates a salt solution that is easier to treat than the organic sludge produced by oxidative chlorination methods. The second step, while using POCl3, is contained within a closed reflux system, minimizing VOC emissions and operator exposure. The final purification via extraction avoids the use of large volumes of silica gel or other solid adsorbents, reducing solid waste disposal requirements. These factors collectively make the process easier to permit and operate in regulated jurisdictions, facilitating faster scale-up and reducing the administrative burden on the EHS (Environment, Health, and Safety) departments of manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative analysis. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the nitration-chlorination sequence over legacy methods, focusing on purity, yield, and operational safety.
Q: Why is the new synthesis method superior to the traditional H2O2/HCl route?
A: The traditional method suffers from low yields, significant byproduct formation, and product instability during post-treatment. The novel nitration-chlorination sequence offers higher stability, simpler purification via pH adjustment, and improved overall purity (up to 98% for the intermediate).
Q: What are the critical control points for ensuring high purity in this process?
A: Critical control points include maintaining the nitration temperature between 70-80°C to prevent over-nitration, precise pH adjustment to 7 during quenching to isolate the intermediate effectively, and careful temperature control (0°C) during the bicarbonate wash in the second step to prevent decomposition.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the process utilizes common industrial reagents like concentrated sulfuric acid and phosphorus oxychloride. The workup involves standard operations like filtration and extraction, making it highly suitable for scale-up from kilogram to multi-ton production without requiring specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-3-Nitropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications and advanced agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering 4-chloro-3-nitropyridine with stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific thermal and corrosive requirements of the nitration and chlorination steps described in CN112707860A, guaranteeing a supply that is both consistent and compliant with global regulatory frameworks.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can drive value in your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies translate into competitive pricing for your projects. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your supply chain is built on a foundation of chemical excellence and reliability.
