Advanced Manufacturing of Cardiovascular Intermediates via Optimized Pyridone Cyclization
Advanced Manufacturing of Cardiovascular Intermediates via Optimized Pyridone Cyclization
The pharmaceutical industry continuously seeks robust synthetic routes for complex cardiovascular agents, particularly those targeting hypertension and ischemic conditions. Patent CN1660842A presents a significant technological advancement in the preparation of [1S-[1a,2b,3b,4a(S*)]]-4-[7-[[1-(3-chloro-2-thienyl)methyl]propyl]amino]-3H-imidazo[4,5-b]pyridin-3-yl]-N-ethyl-2,3-dihydroxycyclopentanecarboxamide, widely recognized as Compound I. This molecule serves as a potent heart protective agent and lipotropic fat decomposition agent, making its efficient synthesis critical for global supply chains. The disclosed methodology addresses historical challenges in heterocyclic chemistry by optimizing the construction of the imidazo[4,5-b]pyridine core, a structural motif essential for biological activity. By refining reaction conditions and solvent systems, this patent provides a blueprint for high-purity intermediate production that aligns with modern Good Manufacturing Practice (GMP) standards.
![Chemical structure of Compound I, a cardiovascular agent intermediate featuring an imidazo[4,5-b]pyridine core linked to a chlorothiophene moiety](/insights/img/cardiovascular-intermediate-synthesis-pharma-supplier-20260305171150-01.webp)
For R&D directors evaluating process feasibility, the structural integrity of Compound I is paramount. The molecule integrates a chiral cyclopentane ring with a substituted thiophene and a fused nitrogenous heterocycle, presenting multiple stereochemical challenges. The patent details a convergent strategy where the complexity is built up through carefully controlled coupling reactions. Understanding the precise stereochemistry at the 1S, 2b, 3b, and 4a positions is vital for ensuring the final API meets stringent pharmacological profiles. The synthesis described avoids racemization risks often associated with harsh acidic or basic conditions in late-stage functionalization, thereby preserving the optical purity required for therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of related pyridone derivatives and their subsequent conversion into imidazo-fused systems has been plagued by low yields and difficult purification protocols. Previous methods, such as those disclosed in U.S.P. 5364862 and U.S.P. 5561134, often relied on multi-step sequences requiring the isolation of unstable intermediates. These conventional approaches frequently utilized volatile or hazardous solvents that complicated waste management and increased operational costs. Furthermore, the nitration and halogenation steps on the pyridine ring often resulted in complex impurity profiles, necessitating extensive chromatographic purification which is impractical at a commercial scale. The inability to telescope these reactions efficiently led to prolonged cycle times and reduced overall throughput, creating bottlenecks for procurement teams managing tight production schedules.
The Novel Approach
The methodology outlined in CN1660842A introduces a streamlined pathway that significantly enhances operational efficiency and product quality. A key innovation lies in the preparation of the 4-hydroxy-3-nitro-2(1H)-pyridone precursor (Compound XII) and its conversion to the chloro-derivative (Compound II) using phosphoryl chloride under controlled conditions. This sequence allows for the direct introduction of the necessary functionality without excessive degradation. 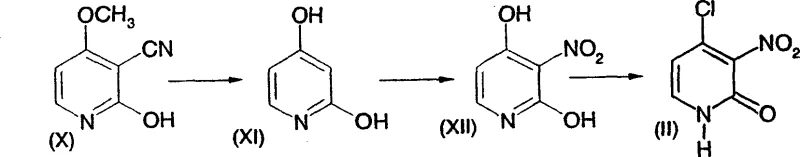
Moreover, the patent describes a telescoped process for the final cyclization step, where the diamine intermediate reacts directly with formamidine acetate or orthoformates in n-butyl acetate. This eliminates the need to isolate the free base of the amine precursor, reducing material loss and solvent consumption. The use of n-butyl acetate is particularly strategic, as it facilitates excellent crystallization properties for the final product, allowing for high-purity isolation via simple filtration rather than complex chromatography. This shift from isolation-heavy workflows to continuous or semi-continuous processing represents a substantial improvement in manufacturing logic, directly addressing the pain points of scalability and cost-efficiency identified in earlier art.
Mechanistic Insights into Formamidine-Mediated Cyclization
The core chemical transformation in this patent involves the cyclodehydration of a 3,4-diaminopyridine derivative to form the imidazo[4,5-b]pyridine ring system. Mechanistically, this reaction proceeds through the nucleophilic attack of the 3-amino group on the electrophilic carbon of the formamidine acetate or triethyl orthoformate. The choice of reagent dictates the kinetics and byproduct profile; formamidine acetate offers a direct route with acetate as a leaving group, while orthoformates require acid catalysis to generate the reactive imidate species. The patent specifies that carrying out this reaction in n-butyl acetate at elevated temperatures (around 85°C) promotes the elimination of water or alcohol, driving the equilibrium toward the cyclic product. This solvent choice is critical because it maintains the reactants in solution at high temperatures while allowing the product to precipitate upon cooling, effectively shifting the equilibrium and simplifying downstream processing.
Impurity control is another mechanistic advantage of this specific protocol. In traditional syntheses, over-alkylation or polymerization of the amine groups can occur, leading to difficult-to-remove tars. By strictly controlling the stoichiometry of the cyclizing agent and utilizing a buffered system (often involving the dihydrochloride salt and a weak base like sodium carbonate), the reaction pH is maintained within an optimal window. This prevents the protonation of the nucleophilic amine while avoiding the decomposition of the sensitive nitro or chloro substituents on the pyridine ring. The result is a cleaner reaction profile with fewer side products, which is essential for meeting the rigorous impurity specifications demanded by regulatory bodies for cardiovascular medications. The detailed examples in the patent demonstrate that this mechanistic control translates directly into higher isolated yields and superior crystal morphology.
How to Synthesize Compound I Efficiently
The synthesis of Compound I requires precise adherence to the telescoped protocol described in the patent to maximize yield and purity. The process begins with the preparation of the key diamine intermediate, which is subsequently subjected to cyclization without isolation. This approach minimizes exposure of the sensitive amine to oxidative conditions and reduces handling losses. Operators must ensure that the reaction temperature during the cyclization phase is maintained strictly between 80°C and 90°C to ensure complete conversion while preventing thermal degradation of the thiophene moiety. The following guide outlines the critical operational parameters derived from the preferred embodiments of the patent data.
- Suspend the dihydrochloride salt of the aminopyridine precursor in water and heat to approximately 65°C to form a solution.
- Add n-butyl acetate and an aqueous base solution to liberate the free amine, followed by the addition of formamidine acetate.
- Heat the mixture to 85°C for cyclization, then cool gradually to induce crystallization of the final imidazo[4,5-b]pyridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic route described in CN1660842A offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant reduction of processing steps through telescoping. By combining the neutralization of the amine salt and the subsequent cyclization into a single pot operation, manufacturers can drastically reduce solvent usage and waste generation. This consolidation of steps translates directly into lower utility costs and reduced demand for reactor volume, allowing existing infrastructure to produce greater quantities of the intermediate. Furthermore, the reliance on commodity chemicals such as phosphoryl chloride, zinc powder, and n-butyl acetate ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with specialized or scarce reagents.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps is a major driver for cost optimization in this process. Traditional methods often require drying and purifying the aminopyridine intermediate before cyclization, which consumes significant time and energy. By proceeding directly from the salt form or crude solution to the final cyclized product, the process removes these unit operations entirely. Additionally, the use of n-butyl acetate as a crystallization solvent allows for the recovery and recycling of mother liquors, further enhancing the economic viability of the route. The high selectivity of the reaction also means less material is lost to byproduct formation, improving the overall mass balance and reducing the cost per kilogram of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the reagents used in this synthesis contributes to a more resilient supply chain. Unlike processes that rely on air-sensitive catalysts or cryogenic conditions, this method operates at atmospheric pressure and moderate temperatures using standard industrial equipment. The reduction steps utilizing zinc/ammonium acetate or catalytic hydrogenation are well-understood technologies that can be easily sourced from multiple vendors globally. This flexibility ensures that production schedules are not disrupted by the unavailability of niche materials. Moreover, the stability of the intermediates allows for safer storage and transport if partial campaigns are necessary, providing supply chain planners with greater flexibility in inventory management.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the straightforward workup procedures. The precipitation of the final product from n-butyl acetate avoids the need for large-scale chromatography, which is often a bottleneck in API manufacturing. From an environmental perspective, the process generates less hazardous waste compared to older methods that might utilize chlorinated solvents for purification. The ability to recycle solvents and the use of less toxic reductants align with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This compliance reduces the risk of regulatory shutdowns and ensures long-term sustainability for the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for technical teams evaluating the transfer of this technology to their own manufacturing sites.
Q: What are the critical purity specifications for the pyridone intermediate?
A: The patent emphasizes high purity through telescoped processing, specifically controlling nitro-reduction byproducts and ensuring complete cyclization to avoid open-chain impurities.
Q: Can this process be scaled for commercial API production?
A: Yes, the use of robust solvents like n-butyl acetate and toluene, along with standard reagents like POCl3 and zinc powder, indicates high suitability for multi-kilogram to ton-scale manufacturing.
Q: How does this method improve upon previous US patents?
A: This method offers improved yields and operational simplicity by eliminating intermediate isolation steps, particularly in the conversion of the nitro-pyridine to the final cyclic structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Compound I Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cardiovascular therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry described in CN1660842A can be executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Compound I meets the exacting standards required for clinical and commercial applications. Our commitment to quality assurance means that our clients can rely on us for a steady supply of material that supports their regulatory filings and market launch timelines.
We invite potential partners to engage with our technical procurement team to discuss how we can tailor this manufacturing process to your specific needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need specific COA data to validate our capabilities, we are ready to provide comprehensive support. By leveraging our expertise in heterocyclic chemistry and process optimization, we can help you secure a reliable source of this vital intermediate, reducing your lead time for high-purity pharmaceutical intermediates and accelerating your path to market.
