Advanced Quaternary Ammonium Salt Technology for Scalable Pharmaceutical Intermediate Manufacturing
The landscape of fine chemical synthesis is constantly evolving, driven by the need for safer, more efficient, and environmentally compliant processes. A significant breakthrough in this domain is documented in Chinese Patent CN1269776C, which introduces a novel class of condensing agents based on quaternary ammonium salts containing specific triazine rings. This technology addresses long-standing challenges in the preparation of carboxylic acid derivatives, particularly amides and esters, which serve as critical building blocks for pharmaceuticals, agrochemicals, and polymers. Traditional methods often rely on harsh conditions or hazardous reagents, but this innovation enables condensation reactions to proceed under mild conditions with remarkably high yields. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages and scalability of this triazine-based approach is essential for optimizing production pipelines and ensuring supply chain continuity.
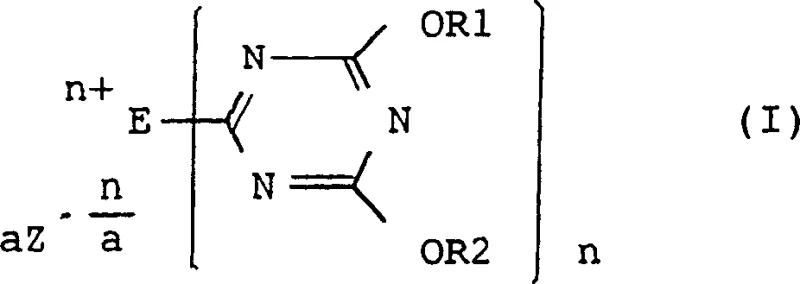
The core of this invention lies in the unique chemical structure of the condensing agent, defined by general formula (I). As illustrated in the structural diagram, the molecule features a 1,3,5-triazine ring substituted with alkoxy or aryloxy groups at the 4 and 6 positions, linked at the 2-position to a quaternary nitrogen atom. This specific architecture is not merely academic; it fundamentally alters the reactivity profile of the condensation process. The quaternary nitrogen, part of a tertiary amine-derived group (E), acts as a potent activator, facilitating the nucleophilic attack on the carboxylic acid without the need for extreme thermal energy. This structural integrity allows the reagent to remain stable yet highly reactive, providing a robust solution for commercial scale-up of complex pharmaceutical intermediates where consistency is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amide and ester bonds has relied heavily on carbodiimide-based condensing agents, such as dicyclohexylcarbodiimide (DCC) or 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC). While effective in certain contexts, these reagents present significant operational and safety liabilities that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. A primary concern is occupational health; carbodiimides are known sensitizers that can cause severe skin rashes and respiratory issues, necessitating expensive containment protocols and protective equipment. Furthermore, their performance is notoriously inconsistent in protic solvents. When water or alcohols are present, carbodiimides often undergo hydrolysis or side reactions, leading to fluctuating yields that can range drastically from 17% to 73%, as noted in prior art. This unpredictability complicates process validation and increases waste generation, making them less ideal for modern, green chemistry initiatives.
The Novel Approach
In stark contrast, the quaternary ammonium salt technology described in CN1269776C offers a paradigm shift in condensation chemistry. By utilizing a triazine ring bonded to a quaternary nitrogen, the new reagents eliminate the safety hazards associated with carbodiimides, creating a safer working environment that reduces regulatory burdens. More importantly, this approach demonstrates exceptional tolerance to solvent conditions. The patent data confirms that high yields are achievable even in protic organic solvents or aqueous organic mixtures, a capability that traditional agents lack. This solvent flexibility is a game-changer for reducing lead time for high-purity pharmaceutical intermediates, as it allows for simpler workup procedures and the potential recycling of solvent streams. The ability to mix the carboxylic acid, the nucleophile, and the condensing agent simultaneously in a one-pot reaction further streamlines the workflow, removing the need for isolating unstable active intermediates.
Mechanistic Insights into Triazine-Mediated Condensation
The efficacy of this technology stems from the electronic properties of the triazine-quaternary ammonium motif. The electron-deficient nature of the triazine ring, enhanced by the electron-withdrawing effect of the adjacent quaternary nitrogen, makes the carbon at the 2-position highly susceptible to nucleophilic attack by the carboxylate anion. This forms a reactive acyl-triazine intermediate in situ. Unlike the O-acylisourea intermediates formed by carbodiimides, which are prone to rearrangement into unreactive N-acylureas, the triazine-based intermediate is more stable and selectively reacts with the incoming amine or alcohol nucleophile. This mechanistic pathway minimizes side reactions and racemization, which is critical when synthesizing chiral high-purity pharmaceutical intermediates like peptides or beta-lactam antibiotics. The presence of the quaternary ammonium moiety also enhances the solubility of the reagent in polar media, facilitating better mass transfer and reaction kinetics.
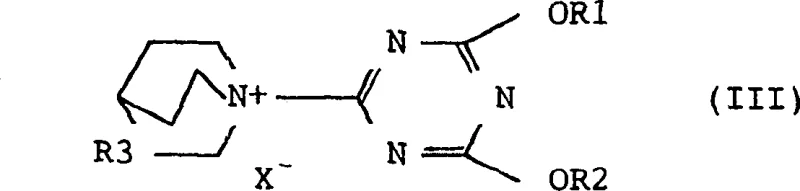
A particularly preferred embodiment of this technology involves the use of quinuclidine-derived quaternary ammonium salts, as depicted in general formula (III). The rigid bicyclic structure of quinuclidine provides steric bulk that can influence the selectivity of the reaction, while the quaternary nitrogen ensures strong activation of the triazine ring. The patent highlights that salts with chloride, perchlorate, or tetrafluoroborate counter-anions are particularly effective. For instance, the tetrafluoroborate salts offer superior stability and lack explosion risks, while chloride salts are economically advantageous due to their ease of preparation. This versatility allows process chemists to tailor the reagent to specific substrate requirements, ensuring optimal performance whether the goal is synthesizing a simple ester or a complex cephalosporin derivative. The robustness of this mechanism supports the production of high-purity OLED material precursors or specialty chemicals where impurity profiles must be tightly controlled.
How to Synthesize Quaternary Ammonium Salt Derivatives Efficiently
Implementing this technology involves a straightforward two-stage process: first, the preparation of the quaternary ammonium salt itself, and second, its application in the condensation reaction. The salt is typically synthesized by reacting a 2-chloro-4,6-dialkoxy-1,3,5-triazine with a tertiary amine in a solvent like tetrahydrofuran or acetonitrile. The resulting precipitate is filtered and dried, yielding a stable solid reagent. In the subsequent condensation step, the carboxylic acid and the nucleophile are dissolved in a suitable solvent, and the salt is added. The reaction proceeds smoothly at temperatures ranging from -20°C to 70°C. Detailed standardized synthetic steps see the guide below.
- Prepare the condensing agent by reacting a 2-chloro-4,6-dialkoxy-1,3,5-triazine with a tertiary amine such as quinuclidine or N-methylmorpholine in an organic solvent.
- Mix the carboxylic acid compound and the nucleophilic compound (amine or alcohol) in a solvent system, which can include protic solvents or aqueous mixtures.
- Add the quaternary ammonium salt condensing agent to the mixture at a temperature between -20°C and 70°C, allowing the reaction to proceed to high yield without isolating active intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this quaternary ammonium salt technology translates into tangible operational improvements beyond mere chemical yield. The primary advantage lies in the drastic simplification of the manufacturing process. By enabling reactions in aqueous or protic solvents, the need for rigorous drying of solvents and substrates is eliminated, significantly reducing energy consumption and processing time. This directly contributes to cost reduction in pharmaceutical intermediates manufacturing by lowering utility costs and increasing batch throughput. Furthermore, the elimination of hazardous carbodiimides reduces the costs associated with waste disposal and worker safety compliance, creating a leaner and more sustainable production model.
Supply chain reliability is another critical benefit. The reagents described in the patent are synthesized from readily available industrial raw materials, such as cyanuric chloride and common tertiary amines. This ensures a stable supply of the condensing agent, mitigating the risk of production delays caused by raw material shortages. The high yields reported in the patent examples, often exceeding 90% for various substrates, mean that less starting material is required to produce the same amount of product. This efficiency enhances the overall Enhanced Supply Chain Reliability by maximizing output from existing reactor capacity. Additionally, the ability to recycle solvent streams, particularly in aqueous systems, reduces the volume of organic waste generated, aligning with increasingly stringent environmental regulations and reducing disposal fees.
Scalability is inherently supported by the mild reaction conditions and the stability of the reagents. The process does not require cryogenic temperatures or high-pressure equipment, making it compatible with standard glass-lined or stainless-steel reactors found in most multipurpose chemical plants. This ease of scale-up facilitates the commercial scale-up of complex pharmaceutical intermediates from pilot plant to full commercial production without significant process re-engineering. The robustness of the chemistry ensures that quality remains consistent across different batch sizes, a key factor for maintaining regulatory approval and customer trust. Moreover, the reduced formation of by-products simplifies purification, often allowing for crystallization rather than chromatography, which is far more economical at large scales.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology, we have compiled answers to common inquiries regarding its application and performance. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is vital for integrating this method into existing workflows and maximizing its potential for cost reduction in pharmaceutical intermediates manufacturing.
Q: How does this quaternary ammonium salt compare to traditional carbodiimide condensing agents?
A: Unlike carbodiimides which can cause skin rashes and require strict anhydrous conditions, this triazine-based quaternary ammonium salt is safer to handle and maintains high reaction yields even in protic solvents or aqueous organic mixtures.
Q: Can this method be used for synthesizing cephalosporin antibiotics?
A: Yes, the patent specifically highlights the successful application of this method for coupling 2-aminothiazolylacetic acid derivatives with 7-aminocephalosporanic acid derivatives to produce cephalosporin intermediates with yields often exceeding 80%.
Q: What are the solvent requirements for this condensation reaction?
A: The process is highly flexible and supports a wide range of solvents including ethers, esters, halogenated hydrocarbons, and notably, it allows for the use of water-containing organic solvents, which simplifies downstream processing and cost reduction in manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quaternary Ammonium Salt Supplier
The transition to advanced condensation technologies requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive support for the production of carboxylic acid derivatives using these novel quaternary ammonium salts. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from laboratory concept to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical and specialty chemical applications.
We invite you to collaborate with us to explore how this technology can optimize your specific synthesis routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current production challenges. Contact us today to request specific COA data and route feasibility assessments, and let us help you achieve greater efficiency and reliability in your supply chain.
