Industrial Scale Chiral Amino Acid Production via Novel Hydantoin Resolution Technology
Industrial Scale Chiral Amino Acid Production via Novel Hydantoin Resolution Technology
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methods for the production of high-purity chiral intermediates, a demand that is critically addressed by the technological disclosures found in patent CN1163472C. This seminal intellectual property outlines a groundbreaking process for the preparation of chiral amino acids of formula (I) through the enantiomeric resolution of racemic hydantoin derivatives. Unlike traditional methods that rely on costly chromatographic separations or complex enzymatic pathways, this invention leverages a sophisticated chemical resolution strategy using specific chiral amines in an alkaline medium. The core innovation lies in the ability to transform racemic glycolylurea directly into optically pure amino acids without the need for isolating intermediate chiral hydantoin species, thereby streamlining the manufacturing workflow. This approach not only enhances the overall yield but also significantly reduces the operational complexity associated with multi-step purification processes. For R&D directors and procurement managers alike, understanding the mechanistic depth of this patent is essential for evaluating its potential integration into existing supply chains for API intermediates and specialty chemicals. 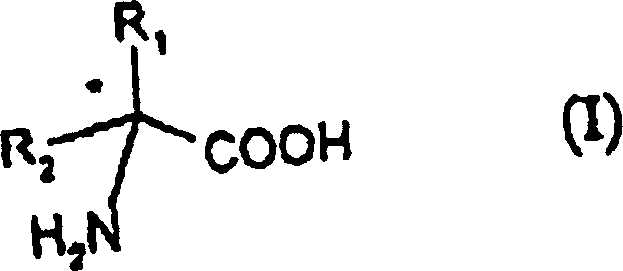
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of chiral amino acids has been plagued by significant technical and economic bottlenecks that hinder efficient commercial scale-up. Conventional methodologies often depend heavily on liquid chromatography (LC) for the resolution of racemic mixtures, a technique that, while effective on a laboratory scale, proves prohibitively expensive and difficult to operate when transferred to large-scale manufacturing environments. Furthermore, enzymatic resolution methods, although highly selective, frequently suffer from the high cost of biocatalysts and the necessity for stringent reaction conditions that limit substrate scope and throughput. These traditional approaches often involve multiple discrete steps, including the separation of diastereomeric salts and the subsequent regeneration of the resolving agent, which cumulatively increase the consumption of solvents, energy, and time. The cumulative effect of these inefficiencies is a substantial escalation in the cost of goods sold (COGS) and a prolonged lead time for product delivery, creating vulnerabilities in the supply chain for critical pharmaceutical intermediates. Consequently, there is an urgent industry need for a method that bypasses these chromatographic and enzymatic constraints while maintaining high stereochemical integrity.
The Novel Approach
The methodology disclosed in CN1163472C represents a paradigm shift by introducing a direct, one-pot chemical resolution process that effectively circumvents the drawbacks of legacy technologies. This novel approach utilizes a specific class of enantiomer resolution agents, such as chiral amines like alpha-methylbenzylamine, which interact with the racemic hydantoin in an alkaline environment to form separable diastereomeric complexes. A distinguishing feature of this invention is the elimination of the intermediate isolation step; the process allows for the direct hydrolysis of the reaction mixture to release the desired chiral amino acid without first isolating the chiral hydantoin intermediate. This consolidation of steps not only simplifies the reactor configuration but also minimizes material loss typically associated with transfer and purification operations between stages. By enabling the quantitative recovery and recycling of the chiral resolution agent through distillation, the process ensures a closed-loop material flow that drastically reduces raw material consumption. This strategic design directly translates to enhanced process economics and a more resilient supply chain capable of meeting the rigorous demands of global pharmaceutical manufacturing.
Mechanistic Insights into Alkaline Hydrolysis and Chiral Resolution
At the heart of this technological advancement is a meticulously engineered reaction mechanism that balances stereochemical selectivity with chemical efficiency. The process initiates with the dissolution of racemic glycolylurea of formula (II) in a solvent system, preferably a mixture of water and a cosolvent such as ethanol or acetone, under alkaline conditions provided by alkali metal hydroxides. The introduction of the chiral resolution agent triggers a selective interaction where one enantiomer of the hydantoin forms a less soluble complex or precipitate, allowing for physical separation from the reaction matrix. Crucially, the patent specifies that the amount of resolution agent required can be as low as 0.2 to 1 equivalent relative to the hydantoin, which is significantly lower than the stoichiometric amounts often required in classical resolution techniques. Following the separation of the resolution agent, which can be recovered via distillation under reduced pressure, the remaining alkaline solution containing the hydantoin salt undergoes hydrolysis. This hydrolysis step is driven by heating the mixture to temperatures ranging from 100°C to 200°C, effectively cleaving the hydantoin ring to liberate the free chiral amino acid. The entire sequence is designed to maximize the enantiomeric excess, with data showing values exceeding 90% and reaching up to 98% in optimized embodiments, ensuring the final product meets the stringent purity specifications required for downstream drug synthesis.
Controlling the impurity profile in chiral synthesis is paramount, and this process incorporates inherent mechanisms to minimize byproduct formation and racemization. The use of a one-pot system reduces the exposure of the chiral intermediate to potentially racemizing conditions that might occur during isolation and drying steps in multi-step processes. Furthermore, the specific selection of alkaline conditions and the controlled addition of the resolution agent help to suppress side reactions that could lead to the formation of undesired isomers or degradation products. The ability to recycle the chiral amine without extensive purification also prevents the accumulation of impurities that could otherwise catalyze unwanted reactions in subsequent batches. From a quality control perspective, the final neutralization and crystallization steps allow for further purification of the amino acid, ensuring that the enantiomeric purity is maintained or even enhanced in the final solid state. This robust control over the reaction environment and impurity trajectory makes the process highly reliable for the production of high-purity pharmaceutical intermediates where regulatory compliance is non-negotiable. 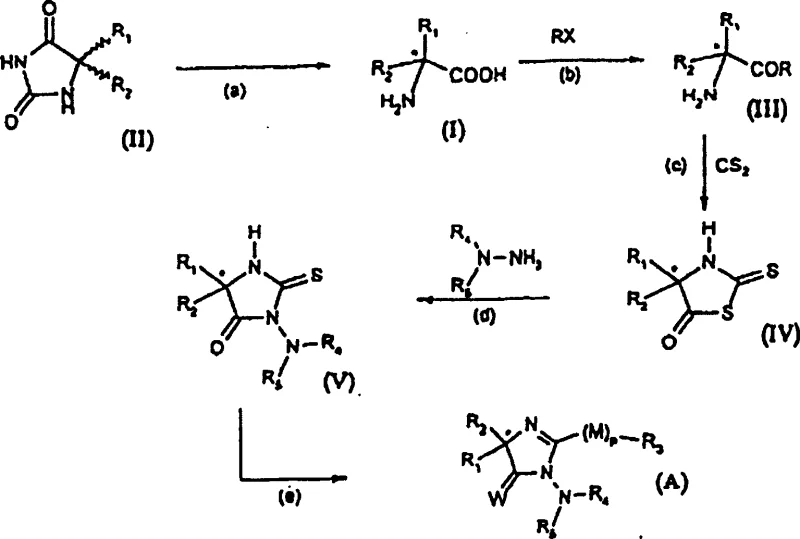
How to Synthesize Chiral Amino Acids Efficiently
The implementation of this synthesis route requires a precise understanding of the operational parameters to ensure optimal yield and stereochemical purity. The process is designed to be adaptable to various substrates defined by the R groups in the hydantoin structure, allowing for the production of a diverse range of chiral amino acids suitable for different therapeutic areas. The standardized protocol involves dissolving the racemic starting material, adding the chiral selector under controlled temperature conditions, separating the precipitate, and then subjecting the filtrate to thermal hydrolysis. Detailed standard operating procedures regarding specific molar ratios, temperature ramps, and distillation pressures are critical for successful technology transfer from laboratory to pilot plant. For technical teams looking to adopt this methodology, adherence to the specific solvent ratios and base concentrations outlined in the patent examples is essential to replicate the high enantiomeric excess reported. The following section outlines the structured steps required to execute this synthesis effectively.
- Dissolve racemic glycolylurea in an alkaline medium containing a chiral resolution agent such as alpha-methylbenzylamine.
- Separate the precipitated resolution agent salt and recover the chiral amine via distillation for reuse.
- Hydrolyze the remaining alkaline reaction mixture under heat to release the final chiral amino acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this hydantoin resolution technology offers profound advantages that directly address the pain points of procurement managers and supply chain heads. The primary value driver is the significant reduction in manufacturing costs achieved through the elimination of expensive chromatographic resins and the minimization of solvent usage associated with multi-step purification. By enabling the recovery and reuse of the chiral resolution agent, the process transforms a typically consumable cost center into a recyclable asset, thereby lowering the variable cost per kilogram of the final product. This economic efficiency is further amplified by the one-pot nature of the reaction, which reduces the requirement for multiple reactor vessels and the associated labor and energy costs for heating, cooling, and transferring materials between units. For procurement teams, this translates into a more stable pricing structure and the ability to negotiate better terms with suppliers who utilize such efficient manufacturing platforms. The overall simplification of the process flow also reduces the risk of production delays caused by equipment bottlenecks or complex purification failures.
- Cost Reduction in Manufacturing: The economic model of this synthesis is fundamentally superior to traditional methods due to the drastic simplification of the downstream processing requirements. By avoiding the need for preparative liquid chromatography or expensive enzymatic kits, the capital expenditure and operational expenditure are both significantly curtailed. The ability to recover the chiral amine resolving agent quantitatively means that the cost of this high-value reagent is amortized over many batches rather than being a single-use expense. Furthermore, the use of common alkaline reagents like sodium hydroxide and standard solvents like water and ethanol ensures that raw material sourcing is straightforward and inexpensive. This combination of reagent recycling and simplified processing logic results in substantial cost savings that can be passed down the supply chain, making the final chiral amino acid intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of manufacturing processes that rely on specialized or single-source materials. This technology mitigates such risks by utilizing widely available commodity chemicals for the reaction medium and base, reducing dependency on niche suppliers. The robustness of the one-pot process also means that production timelines are shorter and more predictable, as there are fewer hand-off points where delays can occur. The high yield and purity achieved in a single operational sequence reduce the need for re-work or re-processing, which often disrupts delivery schedules. For supply chain heads, this reliability ensures that inventory levels can be optimized and that customer demand can be met consistently without the volatility associated with more fragile synthetic routes. The scalability of the process from kilograms to metric tons further ensures that supply can be ramped up quickly to meet surges in market demand.
- Scalability and Environmental Compliance: As regulatory pressure on chemical manufacturing increases, the environmental footprint of a synthesis route becomes a critical factor in vendor selection. This method offers a greener alternative by reducing the volume of waste solvents and chemical byproducts generated per unit of product. The aqueous-based solvent system and the ability to recycle the chiral amine minimize the discharge of hazardous organic waste, aligning with modern environmental, social, and governance (ESG) goals. The simplicity of the equipment required for this one-pot reaction also facilitates easier scale-up from pilot to commercial production without the need for complex, specialized infrastructure. This scalability ensures that the process remains efficient and compliant even at high production volumes, providing a sustainable long-term solution for the manufacturing of chiral intermediates. The reduced energy consumption associated with fewer unit operations further contributes to a lower carbon footprint for the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this chiral resolution technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and technical stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing production lines or for sourcing high-quality intermediates from suppliers who utilize this methodology. The answers provided reflect the specific capabilities and constraints defined within the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: How does this method improve upon traditional enzymatic resolution?
A: Unlike enzymatic methods which often require complex separation steps and expensive biocatalysts, this chemical resolution utilizes a one-pot process with recoverable chiral amines, significantly simplifying the operational workflow and reducing material costs.
Q: What is the enantiomeric purity achievable with this process?
A: The patent data indicates that the process can achieve an enantiomeric excess greater than 90%, with specific embodiments demonstrating values as high as 98%, ensuring high-purity standards suitable for pharmaceutical applications.
Q: Is the chiral resolution agent reusable in this synthesis?
A: Yes, a key economic advantage of this method is the ability to quantitatively separate and recover the chiral resolution agent, such as alpha-methylbenzylamine, via distillation, allowing it to be directly recycled into subsequent batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amino Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-purity synthesis routes in the development of next-generation pharmaceuticals and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the hydantoin resolution method are translated into reliable industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of chiral amino acids meets the exacting standards required by global regulatory bodies. Our infrastructure is designed to support the complex chemistry involved in chiral resolution, providing a secure and compliant environment for the manufacture of sensitive intermediates. By partnering with us, clients gain access to a supply chain that is not only cost-effective but also technically robust and capable of adapting to evolving project needs.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this technology can optimize your specific supply chain requirements. We encourage you to request a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this streamlined manufacturing route. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality chiral intermediates consistently. Let us collaborate to enhance your production efficiency and secure a competitive advantage in the market through superior chemical manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
