Advanced Rhodium-Catalyzed Synthesis of High-Purity Chiral Amino Acids for Pharmaceutical Applications
The pharmaceutical industry's relentless pursuit of biologically active molecules places chiral amino acids at the forefront of synthetic targets, where optical purity is not merely a specification but a critical determinant of therapeutic efficacy and safety. Patent CN103429567A introduces a transformative methodology for the preparation of these vital building blocks, addressing the longstanding challenges associated with achieving high enantiomeric excess in industrial settings. This innovation leverages a sophisticated rhodium-catalyzed asymmetric conjugate addition strategy, enabling the synthesis of chiral alpha- and beta-amino acids with exceptional stereocontrol. By utilizing electron-deficient bisphosphine ligands in conjunction with organoboron derivatives, the process circumvents the harsh conditions and moderate selectivity often plaguing conventional routes. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process efficiency and product quality, ensuring that complex chiral centers are established with precision under remarkably mild reaction parameters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chiral amino acids has relied heavily on the asymmetric hydrogenation of dehydroalanine derivatives or enzymatic resolution of racemic mixtures, both of which present distinct operational bottlenecks. While enzymatic resolution is viable, it is often constrained by substrate specificity and the inherent 50% yield limitation of resolving racemates. Furthermore, prior art regarding enantioselective conjugate additions, such as those described by Sibi et al. involving radical pathways, frequently struggle to exceed enantiomeric excess values of 85%, which is insufficient for high-value API manufacturing. Other methods involving organoboron additions, as noted in literature by Reetz and Frost, typically necessitate heating reaction mixtures to temperatures exceeding 100°C and utilize weak bases or specific proton donors that complicate the workup procedure. These thermal demands not only increase energy consumption but also raise safety concerns regarding scalability, while the modest stereochemical outcomes necessitate costly downstream purification steps to meet stringent regulatory standards for chiral drugs.
The Novel Approach
In stark contrast, the methodology disclosed in CN103429567A establishes a robust 'one-pot' protocol that operates efficiently at mild temperatures ranging from -20°C to 70°C, with many embodiments proceeding optimally at ambient conditions around 25°C. This novel approach utilizes a transition metal catalyst, specifically a rhodium complex paired with a chiral, electron-deficient bisphosphine ligand, to facilitate the 1,4-addition of an organoboron compound to an alpha-amino acrylate. The brilliance of this system lies in its dual functionality where the solvent, often a simple alcohol like isopropanol, acts simultaneously as the reaction medium and the proton donor component. This integration eliminates the need for exotic additives and allows for the direct formation of protected chiral amino acids with enantiomeric excesses consistently surpassing 98.5%. Such a streamlined process drastically reduces the number of unit operations, offering a clear pathway for cost reduction in API manufacturing by minimizing waste generation and energy usage while maximizing chemical yield and optical purity.
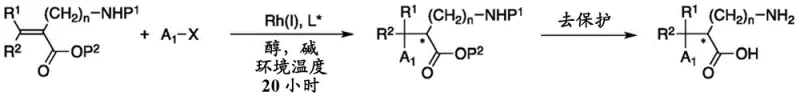
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Conjugate Addition
The core of this technological breakthrough resides in the precise orchestration of the catalytic cycle, driven by the synergy between the rhodium center and the chiral ligand environment. The reaction initiates with the formation of an active rhodium species from precursors such as [RhCl(C2H4)2]2, which then coordinates with the electron-deficient bisphosphine ligand. These ligands, characterized by a phosphorus-selenium coupling constant (JP-Se) greater than 720 Hz, create a sterically demanding and electronically tuned pocket that dictates the facial selectivity of the incoming organoboron reagent. During the critical 1,4-addition step, the carbon-containing group from the boron species is transferred to the beta-position of the alpha-amino acrylate, generating a chiral oxa-pi-allylmetal intermediate. The subsequent protonation step, mediated by the alcoholic solvent, is diastereoselective and serves as the final determinant of the absolute configuration at the newly formed chiral center. This mechanism ensures that even with diverse substituents on the aromatic ring of the boronic acid, the stereochemical integrity is maintained with high fidelity.
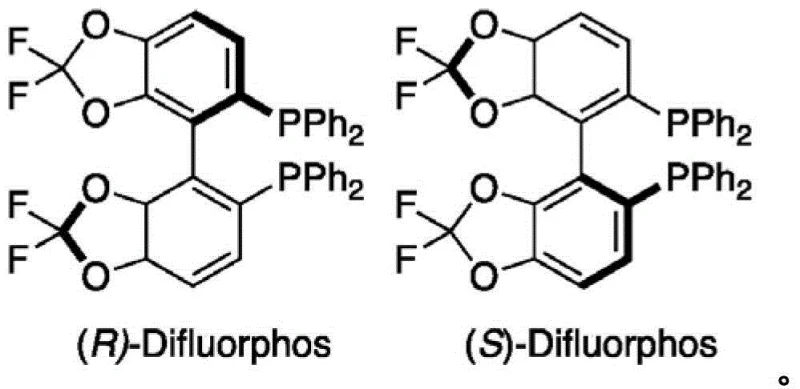
Furthermore, the choice of ligand plays a pivotal role in suppressing side reactions and enhancing the reaction rate. The patent explicitly identifies ligands such as (S)-Difluorphos, (R)-Binap, and (S)-Segphos as particularly effective, with (S)-Difluorphos demonstrating superior performance in many examples, yielding products with up to 99.9% enantiomeric excess. The electron-deficient nature of these ligands accelerates the transmetallation step and stabilizes the catalytic intermediate against decomposition. From an impurity control perspective, the mild basic conditions provided by additives like sodium bicarbonate prevent the racemization of the sensitive alpha-amino ester products, a common issue in stronger alkaline environments. This mechanistic robustness allows for the tolerance of various functional groups on the organoboron partner, including halogens, ethers, and esters, thereby expanding the chemical space accessible for drug discovery without compromising the purity profile of the final pharmaceutical intermediate.
How to Synthesize Chiral Amino Acid Derivatives Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly attractive for process chemistry teams aiming to transition from laboratory scale to pilot production. The general procedure involves charging a reactor with the alpha-amino acrylate substrate, the organoboron derivative (typically 2 equivalents), the rhodium catalyst precursor (1.5 mol%), the chiral ligand (3.3 mol%), and a mild base such as sodium bicarbonate. After establishing an inert atmosphere, a protic solvent like isopropanol is introduced, and the mixture is stirred at controlled temperatures, often between 25°C and 40°C, for approximately 20 hours. The detailed standardized synthesis steps, including specific molar ratios, workup procedures, and purification protocols for various substrates, are outlined in the guide below.
- Prepare the reaction medium by mixing the alpha-amino acrylate starting material, organoboron derivative (e.g., boronic acid), Rhodium catalyst precursor, chiral bisphosphine ligand, and a base in a protic solvent like isopropanol.
- Maintain the reaction mixture under an inert atmosphere (Argon) at mild temperatures ranging from -20°C to 70°C, typically around 25°C for boronic acids, for a duration of approximately 20 hours.
- Upon completion, concentrate the mixture in vacuo and purify the resulting chiral amino acid derivative using standard chromatography techniques such as silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology offers substantial strategic benefits beyond mere chemical elegance. The shift towards a 'one-pot' synthesis significantly simplifies the manufacturing workflow, eliminating the need for isolating unstable intermediates and reducing the overall processing time. This consolidation of steps directly translates to enhanced supply chain reliability, as fewer unit operations mean fewer potential points of failure or delay in the production schedule. Moreover, the use of commercially available and stable organoboron reagents, such as arylboronic acids, ensures a consistent and secure raw material supply, mitigating the risks associated with sourcing exotic or custom-synthesized starting materials that often plague complex API campaigns.
- Cost Reduction in Manufacturing: The process operates under mild thermal conditions, typically avoiding the extreme heating required by legacy methods, which leads to significant energy savings and reduced utility costs. Additionally, the high enantiomeric excess achieved (>98.5%) minimizes the need for expensive chiral resolution or recrystallization steps, thereby lowering the overall cost of goods sold. The elimination of transition metal removal steps often associated with other catalytic systems further streamlines the downstream processing, contributing to substantial cost optimization.
- Enhanced Supply Chain Reliability: By utilizing robust catalyst systems and stable reagents that are tolerant to air and moisture during handling, the process ensures high batch-to-batch consistency. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery windows for downstream API manufacturers. The versatility of the method allows for the rapid adaptation to different amino acid analogs without requiring extensive process re-validation, providing agility in responding to changing market demands.
- Scalability and Environmental Compliance: The use of green solvents like isopropanol and mild inorganic bases aligns with modern environmental, health, and safety (EHS) standards, facilitating easier regulatory approval and waste management. The high atom economy of the conjugate addition reaction reduces chemical waste generation, supporting sustainability goals. Furthermore, the mild reaction conditions enhance process safety, making the scale-up from kilograms to multi-ton quantities safer and more predictable for commercial manufacturing facilities.
Frequently Asked Questions (FAQ)
To assist technical stakeholders in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope, catalyst loading, and product specifications. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the process capabilities.
Q: What is the primary advantage of this Rhodium-catalyzed method over traditional hydrogenation?
A: Unlike traditional asymmetric hydrogenation which often requires high pressure or specific substrates, this method utilizes mild temperatures (-20°C to 70°C) and readily available organoboron reagents to achieve superior enantiomeric excesses exceeding 98.5%.
Q: Which ligands are most effective for this transformation?
A: The patent highlights electron-deficient bisphosphine ligands such as (S)-Difluorphos, (R)-Binap, and (S)-Segphos. Specifically, ligands with a phosphorus-selenium coupling constant (JP-Se) greater than 720 Hz are preferred for optimal stereocontrol.
Q: Can this process be scaled for industrial production?
A: Yes, the process is designed as a 'one-pot' synthesis without isolating intermediates, using stable organoboron compounds and common solvents like isopropanol, which significantly simplifies operational complexity and facilitates commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amino Acid Supplier
The technological potential demonstrated in CN103429567A underscores the importance of having a manufacturing partner capable of executing complex asymmetric syntheses with precision. NINGBO INNO PHARMCHEM stands ready to leverage this advanced rhodium-catalyzed methodology to support your drug development pipeline. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, our team possesses the technical expertise to optimize these reactions for maximum yield and purity. Our rigorous QC labs and stringent purity specifications ensure that every batch of chiral intermediate meets the exacting standards required for global pharmaceutical registration.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this catalytic process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring a seamless transition from bench-scale discovery to commercial supply.
