Advanced Decarbonylative Coupling for High-Purity 2-Aryl Benzoxazole Intermediates
Advanced Decarbonylative Coupling for High-Purity 2-Aryl Benzoxazole Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to access privileged scaffolds such as 2-aryl benzoxazoles. These heterocyclic structures are ubiquitous in bioactive molecules, serving as critical cores for kinase inhibitors and antifungal agents. A significant breakthrough in this domain is documented in patent CN115557912A, which discloses a novel synthetic methodology utilizing a palladium and copper dual-catalytic system. This innovative approach transforms simple aryl carboxylic acids and benzoxazoles directly into high-value 2-aryl benzoxazole derivatives through a decarbonylative coupling mechanism. The technical significance of this patent lies not only in its ability to bypass traditional limitations but also in its alignment with green chemistry principles, offering a robust solution for the reliable pharmaceutical intermediate supplier market. By leveraging commodity chemicals as starting materials, this process drastically simplifies the supply chain complexity associated with specialized halide or boronic acid precursors.
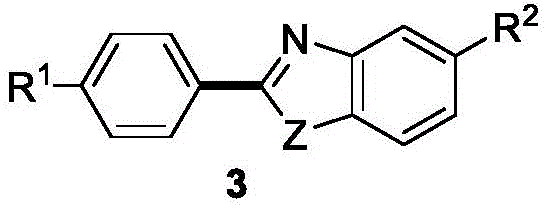
The structural versatility of the target molecules is evident, allowing for extensive derivatization to meet specific drug discovery needs. The core scaffold, as depicted in the patent documentation, supports a wide array of functional groups including halogens, alkyl chains, and electron-withdrawing moieties without compromising reaction efficiency. This flexibility is paramount for R&D teams aiming to optimize structure-activity relationships (SAR) rapidly. Furthermore, the method described in CN115557912A achieves exceptional yields, often exceeding 90% for unsubstituted or electron-rich substrates, which is a critical metric for process chemistry teams evaluating route viability. The combination of high purity output and operational simplicity positions this technology as a cornerstone for next-generation manufacturing of complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aryl benzoxazole compounds has relied heavily on the condensation of o-aminophenols with carboxylic acids or their derivatives, a pathway that frequently suffers from harsh reaction conditions and poor atom economy. Traditional cyclization methods often require high temperatures, strong dehydrating agents, and extended reaction times, leading to significant energy consumption and the generation of substantial chemical waste. Moreover, the substrate scope for these classical methods is notoriously narrow; electron-deficient or sterically hindered aminophenols often fail to react efficiently, limiting the chemical space accessible to medicinal chemists. Alternative transition-metal catalyzed approaches, such as C-H activation using aryl halides, introduce additional burdens related to the cost and availability of halogenated starting materials, as well as the stoichiometric production of salt byproducts. These legacy processes create bottlenecks in cost reduction in pharmaceutical intermediate manufacturing, forcing procurement teams to manage volatile pricing for specialized reagents and complex waste disposal protocols.
The Novel Approach
In stark contrast, the methodology outlined in CN115557912A represents a paradigm shift by utilizing aryl carboxylic acids and benzoxazoles as the primary building blocks, effectively turning a waste-generating decarboxylation step into a constructive bond-forming event. This novel route eliminates the need for pre-functionalized aryl halides or boronic acids, thereby accessing a vastly broader and cheaper pool of raw materials that are abundantly available in the global chemical market. The reaction proceeds smoothly in cyclohexane, a non-polar solvent that facilitates easy product isolation and recycling, further enhancing the environmental profile of the process. As illustrated by the reactant structures, the simplicity of the input materials belies the sophistication of the catalytic transformation occurring within the reactor.
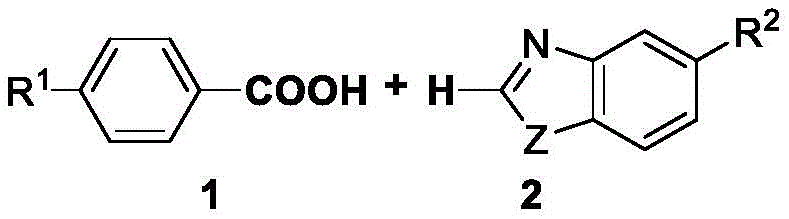
The operational simplicity of this new approach cannot be overstated; it requires no special equipment beyond standard heating and stirring capabilities, making it immediately transferable to existing pilot and production facilities. By avoiding the use of toxic or explosive reagents often associated with older coupling strategies, this method significantly reduces safety risks and regulatory hurdles. For supply chain heads, this translates to reducing lead time for high-purity pharmaceutical intermediates, as the sourcing of carboxylic acids is far more stable and predictable than that of exotic organometallic reagents. The ability to run the reaction at a moderate 120°C for 12 hours ensures that thermal degradation of sensitive functional groups is minimized, preserving the integrity of complex molecular architectures required for advanced therapeutic applications.
Mechanistic Insights into Pd/Cu-Catalyzed Decarbonylative Coupling
The success of this transformation hinges on the synergistic interplay between the palladium and copper catalytic cycles, orchestrated by the bidentate phosphine ligand 1,3-bis(diphenylphosphine)propane (dppp). Mechanistically, the reaction initiates with the oxidative addition of the aryl carboxylic acid derivative to the palladium center, facilitated by the activation of the C-O bond. The presence of di-tert-butyl dicarbonate acts as a crucial dehydrating agent, converting the carboxylic acid into a mixed anhydride in situ, which is a far more reactive electrophile for the metal center. Following oxidative addition, the pivotal decarbonylation step occurs, releasing carbon monoxide and generating an aryl-palladium species. This step is thermodynamically driven and is carefully managed by the reaction temperature of 120°C to ensure complete conversion without promoting catalyst decomposition. The copper co-catalyst plays an essential role in activating the benzoxazole C-H bond, likely through a concerted metalation-deprotonation (CMD) pathway, enabling the subsequent transmetallation or direct insertion into the palladium-aryl bond.
Impurity control is a critical aspect of this mechanism, particularly given the potential for homocoupling of the aryl acid or polymerization of the benzoxazole. The specific choice of dppp as a ligand provides the optimal bite angle and electronic environment to stabilize the active palladium species while suppressing off-cycle reactions that lead to tar formation. Furthermore, the use of sodium bicarbonate as a mild base ensures that the reaction medium remains buffered, preventing acid-catalyzed degradation of the sensitive benzoxazole ring. The high selectivity observed, with yields reaching up to 95% for model substrates, indicates that the catalytic cycle is highly efficient and that side reactions are kinetically disfavored. For R&D directors, understanding this mechanistic robustness provides confidence in scaling the process, as the risk of runaway exotherms or unpredictable byproduct profiles is significantly mitigated by the well-defined coordination chemistry involved.
How to Synthesize 2-Aryl Benzoxazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process is designed to be operationally straightforward, involving the premixing of catalysts and ligands before the introduction of substrates to ensure homogeneous distribution of the active species. Detailed standard operating procedures would dictate the precise order of addition to prevent localized high concentrations of reagents that could lead to hot spots or precipitation. The following guide summarizes the critical operational phases derived from the experimental examples provided in the intellectual property documentation.
- Charge a reaction vessel with aryl carboxylic acid (0.75 mmol), benzoxazole (0.5 mmol), palladium acetate (1 mol%), copper chloride (10 mol%), and 1,3-bis(diphenylphosphine)propane ligand (10 mol%).
- Add di-tert-butyl dicarbonate (150 mol%) as the anhydride source and sodium bicarbonate (150 mol%) as the base into cyclohexane solvent.
- Heat the mixture to 120°C and maintain effective stirring for 12 hours, followed by column chromatography purification to isolate the target 2-aryl benzoxazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this decarbonylative coupling technology offers profound advantages for organizations focused on margin improvement and supply chain resilience. The shift from expensive, specialized coupling partners to commodity carboxylic acids fundamentally alters the cost structure of the manufacturing process. Since aryl carboxylic acids are produced on a massive scale for various industrial applications, their market price is stable and significantly lower than that of aryl iodides or boronic acids typically used in cross-coupling. This raw material substitution drives substantial cost savings in fine chemical manufacturing, allowing procurement managers to negotiate better terms with suppliers due to the high fungibility of the inputs. Additionally, the elimination of stoichiometric activators and the use of a recyclable solvent system like cyclohexane reduce the overall material intensity of the process, directly impacting the cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The economic benefits extend beyond raw material costs to include downstream processing efficiencies. The high conversion rates and clean reaction profiles mean that purification steps, such as column chromatography or crystallization, are less burdensome and consume fewer resources. By removing the need for expensive transition metal scavengers often required to meet strict residual metal limits in APIs, the process simplifies the workup procedure. This reduction in unit operations translates to lower utility consumption and reduced labor hours per kilogram of product. Consequently, the overall manufacturing footprint is minimized, allowing for higher throughput in existing facilities without the need for capital-intensive infrastructure upgrades.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available starting materials that are not subject to the geopolitical or logistical constraints often associated with specialized organometallic reagents. Aryl carboxylic acids and benzoxazoles are stocked by numerous global chemical distributors, ensuring that production schedules are not disrupted by single-source dependencies. This diversification of the supply base mitigates the risk of stockouts and price spikes, providing a stable foundation for long-term production planning. For supply chain heads, this reliability is crucial for maintaining just-in-time delivery commitments to downstream pharmaceutical clients who operate on tight development timelines.
- Scalability and Environmental Compliance: The environmental profile of this method aligns perfectly with increasingly stringent global regulations regarding chemical manufacturing emissions and waste. The absence of halogenated byproducts and the use of a non-chlorinated solvent simplify wastewater treatment and solvent recovery processes. The reaction operates at atmospheric pressure and moderate temperatures, reducing the safety risks associated with high-pressure hydrogenation or cryogenic conditions. This inherent safety and environmental compatibility facilitate easier regulatory approval for commercial scale-up of complex pharmaceutical intermediates, accelerating the time-to-market for new drug candidates. The process is inherently green, supporting corporate sustainability goals while maintaining economic viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, based on the detailed specifications and experimental data provided in the patent literature. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for large-scale production. Understanding these nuances is essential for technical teams evaluating the integration of this method into their existing manufacturing portfolios.
Q: What are the primary advantages of this decarbonylative coupling method over traditional cyclization?
A: Unlike traditional cyclization which often requires harsh conditions and has limited substrate scope, this Pd/Cu catalyzed method utilizes readily available aryl carboxylic acids and benzoxazoles under mild conditions (120°C) with high yields up to 95%.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process uses common solvents like cyclohexane and avoids special equipment requirements. The robust catalytic system and high conversion rates make it highly feasible for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of the 1,3-bis(diphenylphosphine)propane ligand in this reaction?
A: The dppp ligand coordinates with the palladium catalyst to stabilize the active species and facilitate the oxidative addition and decarbonylation steps, ensuring high selectivity and minimizing side reactions during the coupling process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Benzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the decarbonylative coupling technology described in CN115557912A for the production of high-value heterocyclic intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into robust, commercial-grade manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage supply is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-aryl benzoxazole meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective and sustainable synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a competitive advantage through superior supply chain management and advanced chemical manufacturing capabilities.
