Revolutionizing 2-Aryl Benzoxazole Production: A Cost-Effective FeCl3 Catalytic Strategy for Global Pharmaceutical Supply Chains
Revolutionizing 2-Aryl Benzoxazole Production: A Cost-Effective FeCl3 Catalytic Strategy for Global Pharmaceutical Supply Chains
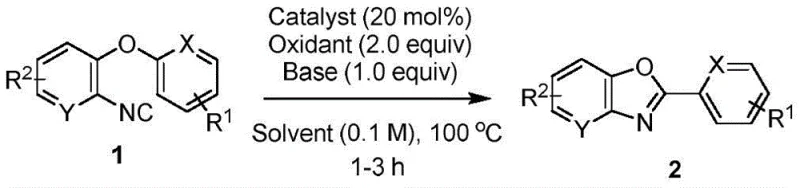
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more sustainable, cost-effective, and efficient synthetic methodologies. A groundbreaking development in this sector is detailed in patent CN114149380A, which discloses a novel preparation method for 2-aryl benzoxazole series compounds. These heterocyclic scaffolds are ubiquitous in medicinal chemistry, serving as critical cores for drugs exhibiting antibacterial, anti-inflammatory, and anticancer activities. The patent introduces a transformative approach that utilizes o-isocyano diaryl ether as a substrate, facilitating an efficient aryl migration from oxygen to imine carbon under the catalysis of ferric trichloride. This innovation not only addresses the limitations of traditional synthesis routes but also aligns perfectly with the modern industry's demand for green chemistry and high atom utilization, presenting a compelling opportunity for reliable pharmaceutical intermediate suppliers to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-aryl benzoxazole skeleton has relied heavily on two primary strategies, both of which suffer from significant drawbacks in a commercial setting. The most common traditional method involves the condensation of 2-aminophenol with carboxylic acid derivatives. While conceptually straightforward, this pathway often necessitates harsh reaction conditions, high temperatures, and the use of strong dehydrating agents, leading to poor atom economy and the generation of substantial chemical waste. Furthermore, the purification of the resulting products can be cumbersome due to the formation of side products and the difficulty in removing unreacted starting materials. Alternatively, C-H functionalization strategies based on pre-formed benzoxazole substrates have been explored; however, these typically rely on expensive transition metal catalysts such as palladium, rhodium, or ruthenium, often accompanied by sophisticated and costly ligand systems. The reliance on noble metals not only inflates the raw material costs drastically but also introduces stringent regulatory hurdles regarding heavy metal residues in the final active pharmaceutical ingredients (APIs), thereby complicating the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN114149380A offers a paradigm shift by employing an oxidative cyclization strategy mediated by inexpensive iron salts. The core of this innovation lies in the use of o-isocyano diaryl ether substrates, which undergo a unique rearrangement where the aryl group attached to the oxygen atom migrates to the isocyano carbon. This process is catalyzed by ferric chloride (FeCl3) in the presence of tert-butyl peroxide as an oxidant and triethylamine as a base, all conducted in a DMF solvent at a moderate temperature of 100 °C. This novel route effectively bypasses the need for precious metal catalysts and complex ligand architectures. Moreover, the reaction demonstrates exceptional regioselectivity and broad substrate applicability, accommodating various electron-donating and electron-withdrawing groups on the aromatic rings. By reconstructing the aryl connection directly within the substrate, the method achieves high atom utilization, avoiding the release of extraneous atoms that typically complicate post-reaction processing. This streamlined approach represents a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
To fully appreciate the robustness of this synthetic platform, one must delve into the mechanistic intricacies of the FeCl3/TBHP/Et3N catalytic system. The reaction initiates with the generation of radicals, likely facilitated by the interaction between the iron catalyst and the tert-butyl peroxide oxidant. In this specific solvent system, the DMF plays a dual role not just as a medium but potentially as a participant in radical generation, where N,N-dimethylformyl radicals may add to the isocyano group of the substrate. This radical addition activates the isocyano functionality, rendering it susceptible to nucleophilic attack. Subsequently, an ipso-attack cyclization occurs, targeting the aryl ring connected to the oxygen atom. This critical step triggers the migration of the aryl group from the oxygen to the imine carbon, forming the new C-C bond that defines the 2-aryl benzoxazole structure. The elegance of this mechanism lies in its ability to forge difficult C-C bonds under relatively mild oxidative conditions without requiring pre-functionalized halides or organometallic reagents.
Furthermore, the impurity profile of this reaction is notably clean, which is a paramount concern for R&D directors focusing on purity specifications. The high regioselectivity ensures that the aryl migration occurs specifically at the desired position, minimizing the formation of structural isomers that are notoriously difficult to separate. The use of a cheap iron catalyst also mitigates the risk of toxic heavy metal contamination, a common issue with palladium-catalyzed cross-couplings. The catalytic cycle is closed efficiently as the released radicals abstract hydrogen to regenerate the solvent or terminate harmlessly, ensuring that the reaction proceeds to completion within a short timeframe of 1 to 3 hours. This mechanistic efficiency translates directly to operational simplicity, allowing for easier scale-up and more predictable batch-to-batch consistency, which are essential attributes for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Aryl Benzoxazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and industrial production. The protocol involves mixing the o-isocyano diaryl ether substrate with the catalytic system in a standard reaction vessel under an air atmosphere, eliminating the need for rigorous inert gas protection which can be a bottleneck in large-scale reactors. The reaction conditions are forgiving, tolerating a range of substituents including halogens, alkyl groups, and trifluoromethyl groups, as evidenced by the successful synthesis of diverse derivatives like 2-phenylbenzo[d]oxazole and its substituted analogs. For detailed standard operating procedures and specific stoichiometric adjustments for different substrates, please refer to the technical guide below.
- Prepare the reaction mixture by combining o-isocyano diaryl ether substrate (1.0 equiv), ferric chloride catalyst (20 mol%), tert-butyl peroxide oxidant (2.0 equiv), and triethylamine base (1.0 equiv) in a reaction vessel under air atmosphere.
- Add DMF solvent to achieve a substrate concentration of 0.1 M and heat the mixture to 100 °C, maintaining the temperature for 1 to 3 hours while monitoring progress via TLC.
- Upon completion, extract the reaction mixture three times with ethyl acetate, dry the combined organic phases over anhydrous sodium sulfate, and purify the crude product using flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this FeCl3-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical curiosity. The shift from noble metal catalysts to base metal iron represents a fundamental change in the cost structure of producing these valuable heterocycles. By eliminating the dependency on volatile palladium markets and expensive proprietary ligands, manufacturers can achieve substantial cost savings in raw material procurement. Additionally, the simplified workup procedure, which relies on standard extraction and drying techniques rather than complex scavenging processes to remove trace metals, reduces the consumption of auxiliary chemicals and lowers waste disposal costs. This holistic reduction in operational complexity directly contributes to a more resilient and cost-efficient supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with ferric chloride drastically lowers the direct material costs associated with the catalytic system. Since iron salts are commodity chemicals available in bulk quantities at a fraction of the price of noble metals, the overall cost of goods sold (COGS) for the final intermediate is significantly reduced. Furthermore, the absence of specialized ligands removes another layer of cost and supply chain vulnerability, as ligand synthesis and purification can often be as expensive as the metal itself. This economic advantage allows suppliers to offer more competitive pricing without compromising on quality, making the production of bioactive benzoxazole derivatives more financially viable for downstream drug developers.
- Enhanced Supply Chain Reliability: The robustness of this reaction under air atmosphere and its tolerance to various functional groups enhance the reliability of the supply chain. Traditional methods often require strictly anhydrous and anaerobic conditions, necessitating specialized equipment and increasing the risk of batch failure due to minor leaks or moisture ingress. In contrast, this novel method's tolerance to air simplifies the engineering requirements for production facilities, reducing downtime and maintenance costs. The wide substrate scope means that a single standardized protocol can be adapted to produce a library of different benzoxazole derivatives, allowing for greater flexibility in responding to changing market demands and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the high atom economy of this aryl migration strategy aligns well with green chemistry principles. By minimizing the generation of stoichiometric byproducts and avoiding the use of toxic heavy metals, the process reduces the environmental footprint of the manufacturing operation. This is increasingly important as regulatory bodies worldwide tighten restrictions on chemical waste and metal residues in APIs. The short reaction time of 1 to 3 hours also implies higher throughput capabilities, enabling manufacturers to maximize reactor utilization rates. Consequently, this method supports the seamless transition from gram-scale discovery to multi-ton commercial production, ensuring a continuous and compliant supply of critical drug building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of this FeCl3-catalyzed method over traditional palladium-catalyzed routes?
A: The primary advantage is the substitution of expensive noble metal catalysts like palladium with inexpensive and abundant iron salts (FeCl3). Additionally, this method avoids the need for specialized ligands and operates under air atmosphere, significantly reducing raw material costs and simplifying the operational setup compared to inert gas-required Pd-catalysis.
Q: How does the atom economy of this novel aryl migration strategy compare to conventional condensation methods?
A: This method exhibits superior atom economy because it utilizes an intramolecular aryl migration where the aryl group connected to oxygen recombines directly to the imine carbon. Unlike traditional condensation of 2-aminophenol and carboxylic acid derivatives which often release water or other small molecules as waste, this rearrangement minimizes atomic waste and avoids the post-treatment inconveniences associated with leaving groups.
Q: Is this synthesis method scalable for industrial production of bioactive intermediates?
A: Yes, the method is highly scalable due to its mild reaction conditions (100 °C), short reaction time (1-3 hours), and simple workup procedure involving standard extraction and chromatography. The use of common solvents like DMF and stable reagents like tert-butyl peroxide further supports its feasibility for commercial scale-up in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Benzoxazole Supplier
The technological advancements presented in patent CN114149380A underscore the immense potential of iron-catalyzed transformations in modern organic synthesis. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations in driving efficiency and sustainability in the pharmaceutical sector. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality intermediates regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-aryl benzoxazole produced meets the exacting standards required for drug development and manufacturing.
We invite global pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective synthesis route for their projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in advanced catalytic methods can accelerate your drug development timeline and optimize your supply chain economics.
