Scaling High-Purity Esomeprazole Sodium Production via Optimized Asymmetric Oxidation
The pharmaceutical industry continuously seeks robust manufacturing routes for proton pump inhibitors, specifically focusing on the levoisomer known as Esomeprazole Sodium. Patent CN103570686A introduces a significant technological advancement in the synthesis and refinement of this critical active pharmaceutical ingredient. Unlike traditional methods that rely on the resolution of racemic mixtures or immature enzymatic processes, this patent details a highly efficient asymmetric oxidation pathway. The core innovation lies in the utilization of a chiral titanium-tartrate catalytic system combined with cumene hydroperoxide, which dramatically improves reaction selectivity and yield. This approach addresses the longstanding challenge of controlling over-oxidation to sulfone impurities while maintaining an enantiomeric excess (ee value) exceeding 99.8%. For global supply chains, this represents a pivotal shift towards more sustainable and cost-effective API manufacturing.
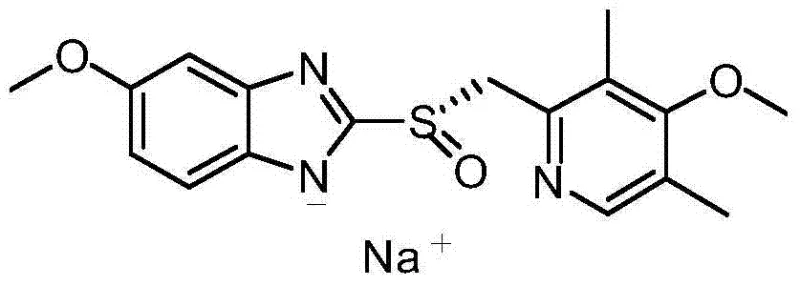
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Esomeprazole has been hindered by the inherent inefficiencies of resolution methodologies. Conventional techniques often involve synthesizing the racemic omeprazole first and then separating the S-enantiomer using chiral resolving agents or chromatography. This approach is fundamentally flawed from an economic and environmental perspective because it theoretically wastes 50% of the starting material—the unwanted R-enantiomer—which cannot be easily recycled. Furthermore, the resolving agents used, such as S-triphenyl ethylene glycol, are often expensive and toxic, adding significant burden to waste treatment protocols. Enzymatic oxidation, while promising, currently suffers from technological immaturity, complex operational requirements, and high production costs that restrict its application in large-scale industrial settings. These factors collectively result in low total recovery rates, often hovering around 13%, making the final drug substance prohibitively expensive for broader market access.
The Novel Approach
The methodology outlined in the patent circumvents these bottlenecks by employing a direct asymmetric oxidation of the prochiral thioether intermediate. Instead of creating a mixture and separating it later, this route builds chirality directly into the molecule during the oxidation step. By utilizing D-(-)-diethyl tartrate and titanium isopropylate to form a chiral catalyst, the reaction selectively targets the sulfur atom to form the S-sulfoxide bond. This eliminates the 50% material loss associated with resolution. Additionally, the substitution of traditional oxidants with cumene hydroperoxide allows for milder reaction conditions, typically between 0°C and 10°C, which minimizes thermal degradation and side reactions. The result is a streamlined process where the total molar yield can exceed 50%, effectively doubling the efficiency compared to older resolution-based benchmarks while simplifying the downstream purification workload.
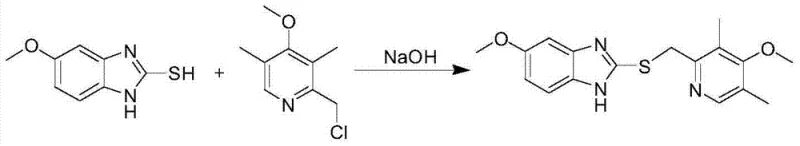
Mechanistic Insights into Titanium-Tartrate Catalyzed Asymmetric Oxidation
The heart of this synthetic strategy is the formation of a chiral titanium complex in situ. When titanium isopropylate reacts with D-(-)-diethyl tartrate and water, it generates a coordinatively unsaturated chiral species that acts as the active catalyst. This complex binds to the sulfur atom of the prochiral thioether substrate, orienting it in a specific spatial configuration. Upon the addition of cumene hydroperoxide, the oxygen transfer occurs stereoselectively, favoring the formation of the S-sulfoxide over the R-sulfoxide. The presence of diisopropylethylamine serves as a base to scavenge protons and stabilize the transition state, ensuring high enantioselectivity. Crucially, the choice of cumene hydroperoxide over stronger oxidants like meta-chloroperbenzoic acid (m-CPBA) is a deliberate mechanistic decision to prevent over-oxidation. Stronger oxidants often push the reaction past the sulfoxide stage to the sulfone, a difficult-to-remove impurity that compromises drug safety. The titanium-tartrate system provides a 'soft' oxidation environment that halts precisely at the sulfoxide stage.
Impurity control is further enhanced through a sophisticated purification protocol designed to handle the specific physicochemical properties of the esomeprazole salt. The patent highlights that while recrystallization is common, it can inadvertently induce polymorphic changes or lead to significant yield loss due to solubility issues. Instead, the process utilizes a targeted slurry purification method using mixed solvent systems, such as acetone and acetonitrile. This technique leverages the differential solubility of the sulfone impurity versus the desired sulfoxide product. By maintaining the temperature between 55°C and 65°C during the dissolution phase and carefully cooling to 15°C-20°C, the process ensures that the sulfone content is reduced to below 0.03% without altering the crystal lattice of the active ingredient. This level of control is essential for meeting the stringent regulatory requirements for generic and branded PPI formulations.
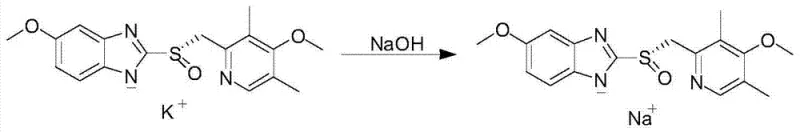
How to Synthesize Esomeprazole Sodium Efficiently
The synthesis of Esomeprazole Sodium via this patented route involves three distinct operational stages that must be tightly controlled to ensure reproducibility and quality. The process begins with the condensation of the benzimidazole and pyridine precursors, followed by the critical chiral oxidation, and concludes with a salt exchange and purification sequence. Each step requires precise management of stoichiometry, temperature, and solvent composition to maximize the yield of the S-enantiomer while minimizing the formation of the sulfone byproduct. The following guide outlines the standardized workflow derived from the patent examples, providing a roadmap for process chemists aiming to implement this high-efficiency route in a pilot or commercial plant setting.
- Condense 2-mercapto-5-methoxybenzimidazole with 2-chloromethyl-3,5-dimethyl-4-methoxypyridine hydrochloride to form the prochiral thioether intermediate.
- Perform asymmetric oxidation using a chiral titanium-tartrate complex and cumene hydroperoxide to generate the esomeprazole potassium salt with high enantiomeric excess.
- Dissolve the potassium salt in water, adjust pH, and perform a salt exchange with sodium hydroxide followed by rigorous purification to obtain the final sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric oxidation technology translates into tangible strategic benefits beyond mere technical specifications. The primary advantage is the drastic improvement in atom economy. By eliminating the resolution step, the process theoretically utilizes 100% of the prochiral thioether input rather than discarding half of it. This fundamental shift in material efficiency leads to a substantial reduction in the cost of goods sold (COGS), as fewer kilograms of expensive starting materials are required to produce the same amount of final API. Furthermore, the avoidance of chiral chromatography columns and expensive resolution reagents removes significant capital and operational expenditures from the manufacturing budget, allowing for more competitive pricing in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of the resolution step inherently removes the cost associated with purchasing, recovering, or disposing of chiral resolving agents. Traditional methods often require stoichiometric amounts of expensive tartaric acid derivatives or specialized resins, whereas this catalytic method uses sub-stoichiometric amounts of titanium and tartrate. Additionally, the use of cumene hydroperoxide, a commodity chemical, replaces more specialized and hazardous oxidants, further driving down raw material costs. The simplified workflow also reduces energy consumption by avoiding the multiple heating and cooling cycles often associated with repetitive recrystallizations needed in resolution processes.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as 2-mercapto-5-methoxybenzimidazole and 2-chloromethyl-3,5-dimethyl-4-methoxypyridine, are well-established intermediates with robust global supply chains. Unlike enzymatic methods that may rely on proprietary biocatalysts with limited suppliers, this chemical synthesis relies on standard fine chemicals available from multiple vendors. This diversification of the supply base mitigates the risk of single-source bottlenecks and ensures continuous production capability even during market fluctuations. The stability of the intermediates also allows for safer storage and transport, reducing logistics complexities.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner profile. The reduction in waste generation—specifically the absence of the 50% unwanted enantiomer waste stream—significantly lowers the burden on wastewater treatment facilities. The reaction conditions are mild, typically operating near ambient or slightly elevated temperatures, which reduces the energy footprint of the reactor farm. Moreover, the high purity of the crude product prior to final refinement means less solvent is consumed during the purification stage, aligning with green chemistry principles and facilitating easier regulatory approval for new drug master files (DMFs).
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Esomeprazole Sodium synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, impurity profiles, and scalability concerns. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer.
Q: Why is asymmetric oxidation preferred over resolution methods for Esomeprazole production?
A: Resolution methods inherently waste 50% of the raw material by discarding the unwanted dextrorotatory isomer, leading to higher costs and lower atom economy. Asymmetric oxidation utilizes the prochiral thioether directly, theoretically allowing for 100% conversion to the desired S-isomer, significantly improving yield and reducing raw material costs.
Q: How does this process control the formation of sulfone impurities?
A: The process utilizes cumene hydroperoxide as the oxidant within a controlled titanium-tartrate catalytic system. This system offers superior selectivity compared to harsher oxidants like m-CPBA. Furthermore, specific purification protocols using mixed solvent systems effectively reduce sulfone content to below 0.03%, ensuring high product safety.
Q: What are the critical parameters for maintaining crystal form during purification?
A: Traditional recrystallization can sometimes induce unwanted polymorphic changes or yield loss. This optimized method employs specific slurry purification techniques using solvents like acetone and acetonitrile at controlled temperatures (55-65°C), which removes impurities effectively without compromising the stable crystal form required for bioavailability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels demonstrated in patent literature are faithfully reproduced in our manufacturing facilities. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Esomeprazole Sodium meets the highest international standards for enantiomeric excess and impurity control.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a clear understanding of the technical and commercial advantages offered by our optimized manufacturing processes.
