Advanced HSCCC Purification Technology for Mivacurium Chloride Reference Substance 950U77 Manufacturing
Advanced HSCCC Purification Technology for Mivacurium Chloride Reference Substance 950U77 Manufacturing
The pharmaceutical industry relies heavily on precise reference standards to ensure the safety and efficacy of active pharmaceutical ingredients (APIs), particularly for complex neuromuscular blocking agents. Patent CN111909089B introduces a groundbreaking preparation method for the mikui ammonium chloride reference substance, specifically the trans-hydrolysate known as 950U77. This patent addresses the critical challenge of separating large-polarity quaternary ammonium salt compounds, which are notoriously difficult to purify using conventional solid-phase techniques. By leveraging High-Speed Counter-Current Chromatography (HSCCC), the disclosed method achieves exceptional separation efficiency without the irreversible adsorption losses typical of silica gel columns. This technological advancement is pivotal for manufacturers seeking to establish robust quality control protocols for Mivacurium Chloride production.
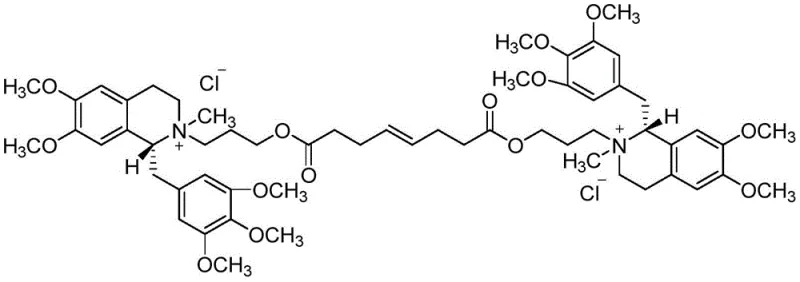
Mivacurium Chloride, also commercially known as Mivacron, is a short-acting non-depolarizing neuromuscular blocker widely used for tracheal intubation and muscle relaxation maintenance during surgical procedures. The molecular architecture of Mivacurium Chloride is highly complex, featuring two chiral carbons and two chiral nitrogens within a symmetrical benzylisoquinoline structure. Due to the presence of these stereocenters, the synthesized drug exists as a mixture of three diastereoisomers: cis-cis, cis-trans, and trans-trans. Accurate quantification of these isomers is essential for regulatory compliance, necessitating a highly pure reference substance like 950U77. The structural integrity and stereochemical purity of this reference standard directly impact the reliability of analytical results used in batch release testing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of polar quaternary ammonium salt reference substances has been plagued by significant technical hurdles associated with solid-phase chromatography. Traditional methods often involve dissolving the crude product in water, freezing to precipitate solids, and utilizing macroporous resins or silica gel columns for further purification. However, the high polarity of compounds like 950U77 leads to strong, often irreversible interactions with the silanol groups on silica gel surfaces. This irreversible adsorption results in substantial sample loss, drastically reducing the overall yield of the valuable reference material. Furthermore, prolonged contact with acidic silica gel can induce partial degradation of the sensitive ester linkages within the molecule, compromising the chemical stability and purity of the final product. These inefficiencies make conventional column chromatography both time-consuming and economically unsustainable for large-scale reference standard production.
The Novel Approach
The innovative methodology presented in patent CN111909089B circumvents these limitations by employing High-Speed Counter-Current Chromatography (HSCCC), a support-free liquid-liquid partition technique. Unlike traditional columns that rely on a solid stationary phase, HSCCC retains the stationary phase purely through centrifugal force within a coiled column. This unique mechanism ensures that the sample never interacts with a solid surface, thereby eliminating irreversible adsorption and preventing sample degradation. The process allows for continuous operation with high loading capacity and rapid separation times. By optimizing the solvent system to create distinct partition coefficients for the target isomer versus impurities, this approach achieves superior resolution. The result is a streamlined purification workflow that maximizes recovery rates and delivers reference substances with purity levels exceeding 99%, setting a new benchmark for the industry.
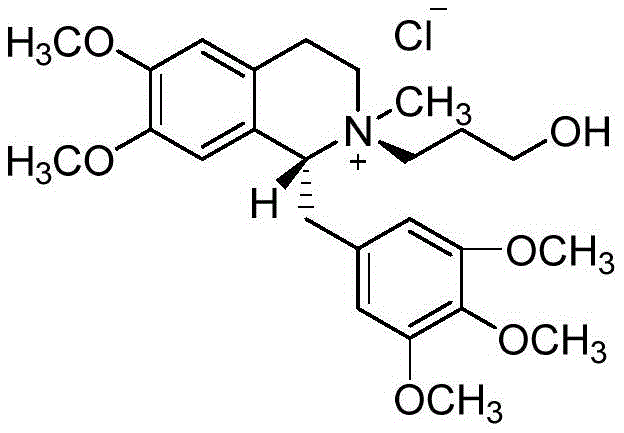
Mechanistic Insights into HSCCC Separation of Quaternary Ammonium Salts
The core of this purification strategy lies in the precise manipulation of partition coefficients (K values) within a carefully selected ternary solvent system. The patent specifies a mixture of Ethyl Acetate (Solvent A), n-Butanol (Solvent B), and Water (Solvent C), with an optimal volume ratio ranging from 1:4:6. In this system, the upper organic phase serves as the stationary phase, while the lower aqueous-rich phase acts as the mobile phase. The separation mechanism is driven by the differential solubility of the 950U77 isomer and its accompanying diastereoisomers between these two immiscible liquid phases. As the mobile phase pumps through the rotating coil, the sample components distribute themselves according to their specific K values. Components with lower K values move faster with the mobile phase, while those with higher K values are retained longer in the stationary phase, resulting in distinct elution peaks.
Impurity control is inherently superior in this liquid-liquid system because the absence of a solid matrix removes the risk of catalytic degradation or tailing effects caused by active sites on silica. The hydrolysate 950U77, being a mono-ester quaternary ammonium salt, exhibits different polarity characteristics compared to the di-ester parent drug and other byproducts. The HSCCC system effectively resolves these subtle polarity differences, allowing for the collection of the first major peak which corresponds to the desired trans-hydrolysate. Post-separation, the addition of sodium chloride facilitates salting-out, enabling efficient extraction of the purified product into an organic solvent like dichloromethane. This multi-step refinement ensures that the final solid obtained after evaporation and ether precipitation meets the stringent purity requirements necessary for a certified reference material.
How to Synthesize Mivacurium Chloride Reference Substance Efficiently
The synthesis and purification workflow described in the patent offers a reproducible pathway for generating high-quality 950U77. The process begins with the quaternization of R-(+)-5'-methoxy laudanosine using 3-chloro-1-propanol in the presence of sodium iodide and sodium carbonate under reflux conditions. Following the reaction, the crude mixture undergoes a preliminary cleanup involving anion exchange resin to remove excess ions and byproducts. The resulting solid is then subjected to the critical HSCCC separation step. For detailed operational parameters, including flow rates, rotation speeds, and specific solvent ratios required to replicate this high-purity outcome, please refer to the standardized synthesis guide below.
- Perform reflux reaction on R-(+)-5'-methoxy laudanosine with 3-chloro-1-propanol, sodium iodide, and sodium carbonate in an organic solvent to generate the crude quaternary ammonium salt.
- Dissolve the crude solid in water, treat with strong base anion exchange resin to remove impurities, and precipitate the product using sodium chloride followed by organic extraction.
- Purify the extracted crude product using High-Speed Counter-Current Chromatography (HSCCC) with a specific ethyl acetate/n-butanol/water solvent system to isolate high-purity 950U77.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of HSCCC technology for reference substance production translates into tangible operational efficiencies and cost optimizations. The elimination of solid-phase media removes the recurring cost of purchasing and disposing of large quantities of silica gel or specialty resins. Furthermore, the dramatic improvement in recovery rates means that less starting material is required to produce the same amount of final reference standard, directly lowering the cost of goods sold (COGS). The speed of the HSCCC process also reduces the overall cycle time for quality control material production, ensuring that analytical labs have timely access to the standards they need for batch release testing without bottlenecks.
- Cost Reduction in Manufacturing: The transition from solid-phase to liquid-liquid chromatography fundamentally alters the cost structure of purification. By avoiding the irreversible adsorption losses inherent to silica gel columns, the process significantly increases the yield of the final product. This higher yield implies that fewer raw materials are consumed per gram of purified 950U77, leading to substantial cost savings in precursor procurement. Additionally, the solvent system utilized consists of common, relatively inexpensive organic solvents like ethyl acetate and n-butanol, which can be easily recovered and recycled, further driving down operational expenses and waste disposal costs.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the variability and low throughput of traditional purification methods. The HSCCC method described offers a continuous and scalable process that is less prone to the batch-to-batch variations seen in manual column packing and elution. The robustness of the liquid-liquid separation ensures consistent purity profiles, reducing the risk of failed quality control tests that could delay product shipments. This reliability allows manufacturers to maintain leaner inventories of reference standards while still meeting rigorous regulatory demands, thereby optimizing working capital and improving overall supply chain agility.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology offers distinct advantages for industrial application. The absence of solid waste (spent silica gel) simplifies waste management and aligns with green chemistry principles by reducing the environmental footprint of the manufacturing process. The equipment used for HSCCC is readily scalable from preparative to industrial scales without the need for complex re-engineering of the separation chemistry. This scalability ensures that as demand for Mivacurium Chloride grows, the production of its critical reference substances can be expanded seamlessly to support commercial manufacturing volumes without compromising on purity or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel purification technology. These insights are derived directly from the experimental data and technical specifications outlined in patent CN111909089B, providing clarity on how this method outperforms legacy techniques in terms of efficiency and product quality.
Q: Why is High-Speed Counter-Current Chromatography (HSCCC) preferred over silica gel column chromatography for 950U77?
A: Traditional silica gel chromatography causes irreversible adsorption and sample degradation due to the high polarity and sensitivity of quaternary ammonium salts. HSCCC utilizes a liquid-liquid partition system without a solid support, eliminating sample loss and ensuring higher recovery rates and purity.
Q: What is the specific solvent system used for the separation of Mivacurium Chloride reference substance?
A: The patent specifies a ternary solvent system comprising Ethyl Acetate (Solvent A), n-Butanol (Solvent B), and Water (Solvent C). The optimal volume ratio identified is approximately 1:4:6, which provides the necessary partition coefficient for effective separation of the trans-hydrolysate isomer.
Q: What purity levels can be achieved using this HSCCC method?
A: According to the experimental data in patent CN111909089B, the HSCCC method achieves a liquid phase purity of up to 99.67% for the 950U77 reference substance, with isomer content reduced to negligible levels (0.28%), significantly outperforming traditional freezing and resin adsorption methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mivacurium Chloride Reference Substance Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-purity reference standards play in the pharmaceutical value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global API manufacturers. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and potency of every batch. Our expertise in complex separation technologies allows us to replicate and optimize processes like the HSCCC method described, guaranteeing a consistent supply of top-tier intermediates and reference materials.
We invite you to collaborate with us to enhance your supply chain resilience and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced capabilities can support your long-term strategic goals in the competitive pharmaceutical market.
