Advanced Synthesis of Fluorinated Butadiene Intermediates for Scalable Agrochemical Production
The chemical landscape for advanced agrochemical intermediates is constantly evolving, driven by the need for higher purity and more sustainable manufacturing processes. Patent CN1150151C introduces a transformative methodology for the preparation of 1,4-diaryl-2-fluoro-1,3-butadiene compounds and their corresponding 1,4-diaryl-2-fluoro-2-butene derivatives. These fluorinated structures serve as critical precursors in the synthesis of next-generation insecticides and acaricides, where the introduction of fluorine atoms significantly enhances metabolic stability and biological activity. The core innovation lies in a novel coupling strategy that completely circumvents the use of traditional phosphonium halide reagents, which have long been a bottleneck in terms of cost and purification complexity. By shifting towards aryl methanesulfonyl fluorides or aryl methanephosphonates, this technology offers a streamlined pathway that aligns perfectly with the rigorous demands of modern pharmaceutical and agrochemical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in EP811593-A1, relied heavily on Wittig-type reactions utilizing phosphonium halide compounds. While chemically feasible, these traditional routes present severe disadvantages for industrial scale-up. The primary issue is the generation of stoichiometric quantities of phosphine oxide by-products during the olefination step. These by-products possess physical properties often similar to the desired fluorinated butadiene product, making their removal via standard crystallization or distillation extremely difficult and costly. Furthermore, phosphonium salts themselves are expensive reagents that require multi-step synthesis, adding significant raw material costs to the overall process. The difficulty in separating impurities not only reduces the final yield but also compromises the purity profile, which is unacceptable for high-performance agrochemical active ingredients where trace impurities can affect efficacy and regulatory approval.
The Novel Approach
The process disclosed in CN1150151C fundamentally reengineers the carbon-carbon bond formation step by employing aryl methanesulfonyl fluorides or aryl methanephosphonates as the nucleophilic partners. This strategic substitution eliminates the formation of stubborn phosphine oxide waste. The reaction involves condensing these activated sulfones or phosphonates with 3-aryl-2-fluoroacrolein derivatives under basic conditions. This approach not only simplifies the downstream purification workup—often requiring only simple aqueous extraction and solvent washes—but also utilizes significantly cheaper and more commercially available starting materials. The versatility of this method allows for the synthesis of a broad range of substituted derivatives, accommodating various halogen and alkoxy groups on the aromatic rings, thereby providing a robust platform for generating diverse libraries of bioactive intermediates.
Mechanistic Insights into Base-Catalyzed Fluorinated Olefination
The mechanistic pathway for this transformation involves the deprotonation of the alpha-carbon of the aryl methanesulfonyl fluoride or phosphonate by a suitable base, generating a reactive carbanion species. This nucleophile then attacks the carbonyl carbon of the 3-aryl-2-fluoroacrolein. Unlike traditional Wittig reactions that form a four-membered oxaphosphetane ring, this mechanism likely proceeds through a beta-hydroxy sulfone or phosphonate intermediate, which subsequently undergoes elimination to form the conjugated double bond system. The presence of the fluorine atom at the alpha-position of the aldehyde adds a layer of electronic complexity, stabilizing the intermediate and influencing the E/Z stereoselectivity of the resulting butadiene. The use of phase transfer catalysts, such as 18-crown-6, further enhances the reaction kinetics by solubilizing the inorganic base cations in organic solvents, ensuring efficient contact between the reactants in heterogeneous mixtures.
Impurity control is inherently superior in this new route due to the nature of the leaving groups. When using sulfones, the leaving group is a sulfinate salt, which is water-soluble and easily removed during the aqueous workup phase. Similarly, phosphonate by-products are generally more polar and easier to separate than triphenylphosphine oxide. The reaction conditions are mild, typically operating between -20°C and 100°C, which minimizes thermal degradation of the sensitive fluorinated intermediates. This thermal stability is crucial for maintaining the integrity of the fluorine-carbon bond, preventing defluorination side reactions that could lead to complex impurity profiles. The ability to tune the base strength, from mild carbonates to strong alkoxides, allows process chemists to optimize the reaction for specific substrate combinations, ensuring high conversion rates and minimal formation of oligomeric by-products.
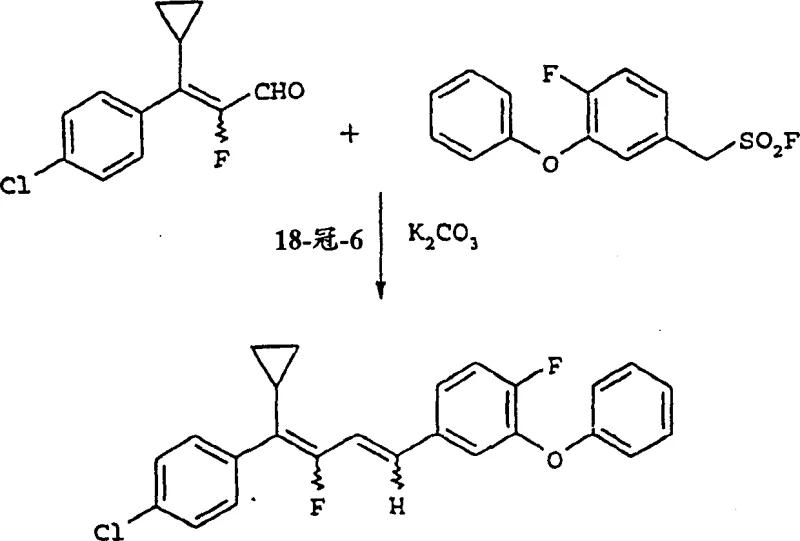
How to Synthesize 1,4-Diaryl-2-Fluoro-1,3-Butadiene Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible framework for laboratory and pilot-scale production. The process begins with the preparation of the key sulfone or phosphonate building block, followed by the coupling step with the fluorinated aldehyde. Detailed operational parameters regarding solvent choice, base equivalents, and temperature ramps are critical for maximizing yield. For instance, the use of acetonitrile as a solvent combined with potassium carbonate and a catalytic amount of 18-crown-6 has shown exceptional results in model reactions. The following guide summarizes the standardized synthetic steps derived from the patent examples to assist technical teams in replicating this high-efficiency route.
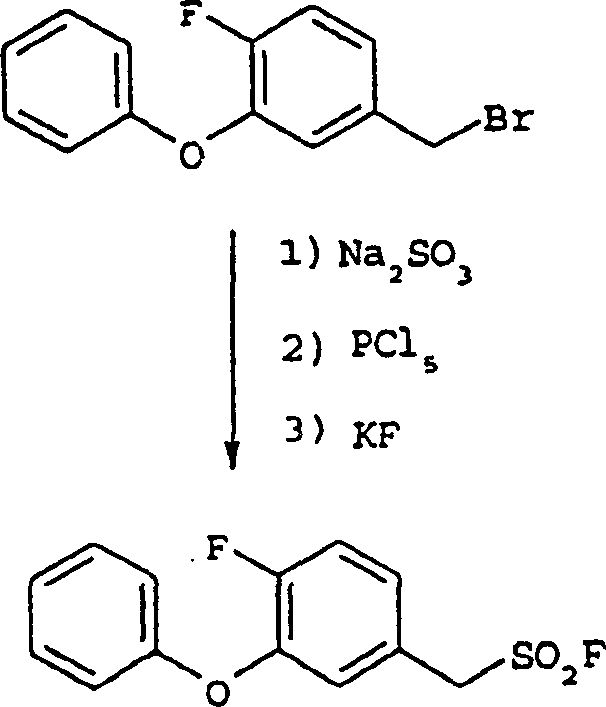
- Preparation of Aryl Methanesulfonyl Fluoride: React arylmethyl bromide with sodium sulfite, followed by chlorination with PCl5 and fluorination with KF.
- Coupling Reaction: Mix the sulfonyl fluoride or phosphonate with 3-aryl-2-fluoroacrolein in the presence of a base like potassium carbonate and a phase transfer catalyst.
- Isolation and Purification: Quench the reaction with water, extract with organic solvents like ethyl acetate, and purify via chromatography or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates directly into tangible operational improvements and risk mitigation. The elimination of phosphonium salts removes a major cost driver and a source of supply chain volatility, as these reagents often have longer lead times and higher price points compared to simple sulfones or phosphonates. Furthermore, the simplified purification process reduces the consumption of solvents and silica gel for chromatography, leading to substantial cost savings in waste disposal and raw material usage. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification purity results.
- Cost Reduction in Manufacturing: The replacement of expensive phosphonium halides with readily available aryl methyl bromides and sulfites drastically lowers the raw material cost basis. Additionally, the avoidance of difficult-to-remove phosphine oxide by-products means that expensive preparative HPLC or multiple recrystallization steps are no longer necessary, significantly reducing processing time and utility costs. The use of inexpensive inorganic bases like potassium carbonate instead of strong, hazardous organolithium reagents further contributes to a safer and more cost-effective manufacturing environment.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as benzyl bromides and sodium sulfite, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The synthetic route is short and convergent, allowing for the stockpiling of key intermediates like the sulfonyl fluorides, which are stable and can be produced in large campaigns. This modularity enables flexible production scheduling, ensuring that delivery timelines for critical agrochemical intermediates can be met even during periods of high market demand.
- Scalability and Environmental Compliance: The process generates aqueous waste streams containing inorganic salts that are easier to treat compared to the organic phosphorus waste associated with traditional Wittig reactions. This simplifies environmental compliance and reduces the burden on wastewater treatment facilities. The reaction is exothermic but manageable within standard reactor setups, and the workup involves simple liquid-liquid extractions, making the technology highly scalable from kilogram to multi-ton production without requiring specialized equipment or hazardous high-pressure conditions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorinated butadiene synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable reference for R&D and procurement teams evaluating this route for potential integration into their supply chains. Understanding these nuances is essential for assessing the feasibility of adopting this method for specific target molecules.
Q: Why is avoiding phosphonium halides significant in this synthesis?
A: Traditional methods using phosphonium halides generate stoichiometric amounts of phosphine oxide by-products, which are notoriously difficult and expensive to separate from the final product, lowering overall yield and purity.
Q: What are the preferred reaction conditions for this coupling?
A: The reaction typically proceeds in solvents like acetonitrile or THF at temperatures ranging from -20°C to 100°C, using bases such as potassium carbonate or sodium methoxide, often enhanced by phase transfer catalysts like 18-crown-6.
Q: Can this process be scaled for industrial insecticide production?
A: Yes, the use of inexpensive inorganic bases and readily available sulfone or phosphonate precursors makes this route highly amenable to large-scale commercial manufacturing with simplified waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Diaryl-2-Fluoro-1,3-Butadiene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the competitive agrochemical sector. Our technical team has extensively analyzed the process described in CN1150151C and possesses the expertise to implement this phosphonium-free route at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of 1,4-diaryl-2-fluoro-1,3-butadiene meets the highest international standards for downstream pesticide synthesis.
We invite you to collaborate with us to optimize your supply chain for fluorinated intermediates. By leveraging this advanced synthetic methodology, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a reliable, cost-effective, and high-quality supply of these critical building blocks for your next-generation insecticide formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
