Revolutionizing Avanafil Production: A Scalable Cytosine-Based Synthetic Strategy for Global API Markets
The pharmaceutical landscape for erectile dysfunction treatments continues to evolve, driven by the demand for rapid-onset agents like Avanafil. A pivotal development in the manufacturing of this high-value Active Pharmaceutical Ingredient (API) is detailed in patent CN103254179A, which discloses a novel preparation method that fundamentally shifts the synthetic paradigm from complex ring construction to efficient functionalization. Unlike legacy approaches that rely on building the pyrimidine core from scratch using costly precursors, this innovation leverages Cytosine—a ubiquitous, inexpensive, and industrially abundant heterocyclic feedstock—as the foundational scaffold. This strategic pivot not only simplifies the synthetic topology but also addresses critical bottlenecks in supply chain stability and process safety. By sequentially executing substitution, condensation, and halogenated addition reactions using readily available side chains such as 3-chloro-4-methoxybenzyl halogen and S-hydroxymethyl pyrrolidine, the patent outlines a robust pathway to the target molecule. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the cost of goods sold (COGS) while ensuring a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
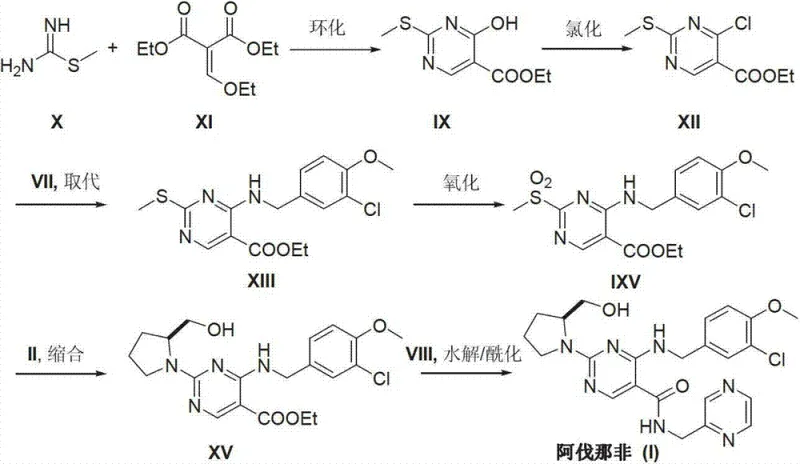
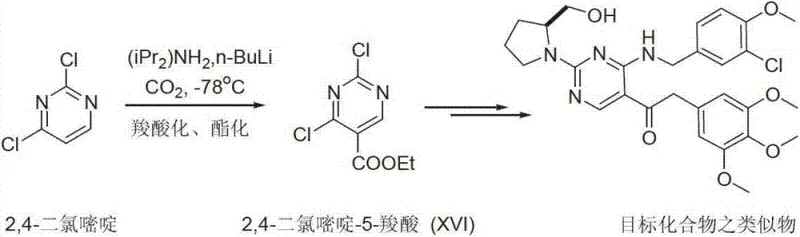
Historically, the synthesis of Avanafil and its analogues has been plagued by inefficient routes that impose severe constraints on commercial scalability. As illustrated in prior art such as WO0183460 and WO0119802, traditional methods often commence with the cyclization of thiourea and ethoxy methyne diethyl malonate to construct the pyrimidine ring, a process that inherently suffers from poor atom economy and generates substantial chemical waste. Furthermore, alternative pathways utilizing 2,4-dichloropyrimidine require the generation of carbanions using n-Butyl Lithium at cryogenic temperatures of -78°C. These ultra-low temperature conditions necessitate specialized refrigeration equipment and rigorous anhydrous protocols, drastically increasing capital expenditure and operational complexity. The reliance on such harsh conditions not only elevates safety risks associated with handling reactive organometallic reagents but also introduces significant challenges in heat transfer and mixing efficiency when attempting to scale from the laboratory to multi-ton reactors. Consequently, these legacy routes often result in inconsistent batch quality and prolonged lead times, creating vulnerabilities in the global supply chain for PDE-5 inhibitors.
The Novel Approach
In stark contrast to these cumbersome methodologies, the approach disclosed in CN103254179A offers a streamlined solution by bypassing the need for de novo ring synthesis entirely. By selecting Cytosine as the starting material, the process capitalizes on a pre-existing, stable pyrimidine architecture that is both economically viable and chemically robust. The novel route proceeds through a logical sequence of transformations: initial N-alkylation at the N1 position, followed by condensation at the C2 position, and final functionalization at the C5 position. This modular strategy allows for the independent optimization of each step, facilitating tighter control over impurity profiles and reaction kinetics. The elimination of cryogenic steps and the replacement of exotic reagents with commodity chemicals like Cytosine and simple benzyl halides dramatically lowers the barrier to entry for manufacturing. This shift not only enhances the economic feasibility of production but also aligns with green chemistry principles by reducing energy consumption and solvent usage, thereby presenting a compelling value proposition for environmentally conscious pharmaceutical manufacturers seeking a reliable pharmaceutical intermediate supplier.
Mechanistic Insights into Cytosine Functionalization and Coupling
The core of this technological breakthrough lies in the precise manipulation of the electronic properties of the Cytosine ring to achieve regioselective functionalization. The initial step involves a nucleophilic substitution where the nitrogen atom of the Cytosine ring attacks the electrophilic carbon of the 3-chloro-4-methoxybenzyl halide. This reaction is facilitated by the presence of a base, such as triethylamine or potassium carbonate, which deprotonates the N1-H bond, increasing its nucleophilicity without compromising the integrity of the sensitive amide functionality within the ring. Following this, the introduction of the chiral pyrrolidine side chain at the 2-position is achieved through a condensation reaction. This step is critical as it establishes the stereochemistry essential for the biological activity of the final drug product. The use of advanced coupling reagents like BOP or HBTU ensures high conversion rates and minimizes racemization, a common pitfall in peptide-like bond formations. The mechanistic elegance of this sequence ensures that the complex molecular architecture of Avanafil is assembled with high fidelity, reducing the burden on downstream purification processes.
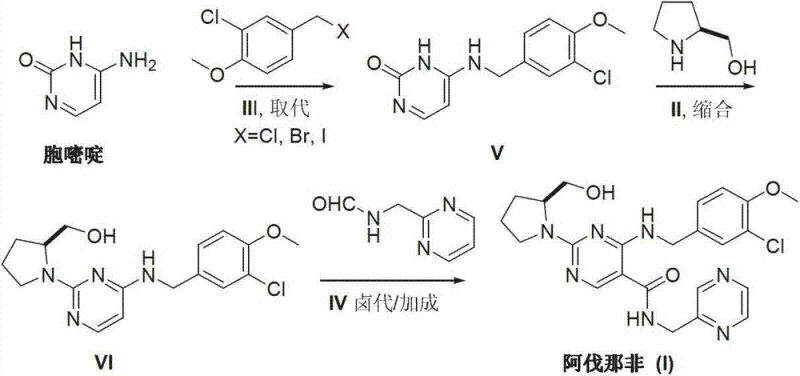
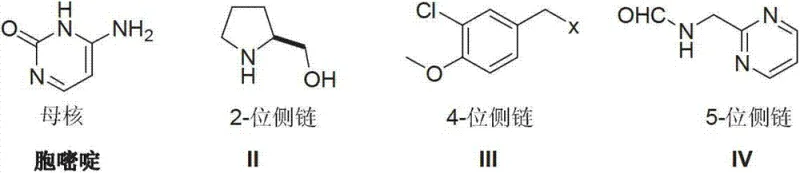
Furthermore, the final stage of the synthesis involves a sophisticated halogenation and addition sequence to install the 5-position side chain. The patent describes a halogenating reaction followed by a catalytic addition with N-(2-methylpyrimidine) methane amide. This transformation likely proceeds via an activated intermediate, potentially a halogenated pyrimidine species, which undergoes nucleophilic attack or metal-catalyzed cross-coupling. The choice of catalyst, such as nickel acetate or palladium complexes, along with phosphine ligands, plays a pivotal role in activating the C-N or C-C bond formation under relatively mild thermal conditions (e.g., 120°C). This mechanistic pathway avoids the use of aggressive reagents that could degrade the sensitive methoxy and chloro substituents on the benzyl ring. By understanding these mechanistic nuances, process chemists can fine-tune reaction parameters to maximize yield and minimize the formation of des-chloro or demethylated byproducts, ensuring the delivery of high-purity Avanafil that meets stringent regulatory specifications for clinical use.
How to Synthesize Avanafil Efficiently
The synthesis of Avanafil via this Cytosine-based route represents a paradigm shift in process chemistry, moving away from hazardous cryogenic operations toward safer, ambient-pressure transformations. The protocol begins with the alkylation of Cytosine, followed by the installation of the chiral pyrrolidine moiety, and concludes with the coupling of the pyrimidine-methyl amine side chain. This sequence is designed to maximize atom economy and operational simplicity, making it an ideal candidate for technology transfer from R&D to pilot plant and eventually to full-scale commercial production. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and temperature profiles optimized for reproducibility, are outlined in the guide below.
- Perform nucleophilic substitution of Cytosine with 3-chloro-4-methoxybenzyl halide to form the N-alkylated intermediate.
- Execute a condensation reaction between the N-alkylated cytosine and S-hydroxymethyl pyrrolidine to construct the 2-position side chain.
- Finalize the synthesis via halogenation followed by a catalytic addition reaction with N-(2-methylpyrimidine) methane amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic route described in CN103254179A offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By anchoring the synthesis on Cytosine, a bulk chemical produced globally in massive quantities, manufacturers can insulate themselves from the volatility associated with niche, custom-synthesized intermediates. This shift significantly mitigates the risk of supply disruptions caused by the limited capacity of specialized chemical vendors. Moreover, the elimination of ultra-low temperature requirements (-78°C) removes a major bottleneck in batch processing times and energy costs. Cryogenic reactions are notoriously slow to cool and warm, limiting the number of batches a reactor can turn over in a given month. By operating at elevated but manageable temperatures (50-120°C), the throughput of existing manufacturing infrastructure can be substantially increased without the need for capital-intensive retrofitting.
- Cost Reduction in Manufacturing: The economic implications of this route are profound, primarily driven by the replacement of expensive, multi-step precursors with commodity-grade starting materials. Traditional routes often require the synthesis of complex pyrimidine esters or the use of stoichiometric amounts of costly organolithium reagents, both of which contribute heavily to the raw material cost. In contrast, the Cytosine route utilizes inexpensive alkyl halides and simple amines. Additionally, the reduction in synthetic steps—from cyclization to final coupling—directly correlates to lower labor costs, reduced solvent consumption, and decreased waste disposal fees. The qualitative improvement in process efficiency means that the overall cost of goods sold (COGS) can be optimized significantly, allowing for more competitive pricing in the generic API market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of key inputs. The reliance on 2,4-dichloropyrimidine or custom malonate derivatives in older patents creates a single point of failure if a specific supplier encounters production issues. Cytosine, however, is a fundamental building block in the nucleobase industry, sourced from multiple global producers with established quality systems. This diversification of supply sources ensures continuity of operations even during regional disruptions. Furthermore, the robustness of the reaction conditions—avoiding moisture-sensitive and air-sensitive reagents—reduces the likelihood of batch failures due to environmental factors. This reliability translates to shorter lead times for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology offers a cleaner profile that aligns with increasingly stringent global regulations. The avoidance of heavy metal catalysts in early stages and the reduction of hazardous waste streams associated with lithiation byproducts simplify the wastewater treatment process. The process is inherently safer, removing the risks associated with handling pyrophoric reagents like n-BuLi on a multi-ton scale. This safety profile facilitates easier regulatory approval for new manufacturing sites and reduces insurance premiums. The scalability is further enhanced by the use of common solvents like ethanol and acetonitrile, which are easily recovered and recycled, supporting a circular economy approach within the chemical plant and ensuring long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams assessing the quality capabilities of potential partners.
Q: What are the primary advantages of the Cytosine-based route over traditional lithiation methods?
A: The Cytosine-based route eliminates the need for hazardous cryogenic conditions (-78°C) and expensive organolithium reagents required in traditional dichloropyrimidine pathways, significantly enhancing operational safety and reducing energy consumption.
Q: How does this synthetic method impact the purity profile of the final API?
A: By utilizing highly selective nucleophilic substitutions on the pre-formed pyrimidine ring of Cytosine, the method minimizes the formation of regio-isomers often seen in non-selective cyclization reactions, leading to a cleaner impurity profile.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes inexpensive, industrially available raw materials like Cytosine and avoids complex multi-step ring constructions, making it highly amenable to scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avanafil Supplier
The transition to a Cytosine-based synthesis for Avanafil represents a significant leap forward in process efficiency, yet realizing its full potential requires a partner with deep expertise in heterocyclic chemistry and scale-up engineering. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and mixing requirements of the condensation and alkylation steps described in CN103254179A, ensuring that the theoretical benefits of the patent are translated into real-world yield and purity. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor impurity profiles at every stage of the synthesis, guaranteeing a product that meets the highest international pharmacopoeia standards.
We invite pharmaceutical companies and contract manufacturers to leverage our technical prowess to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of Avanafil intermediates, positioning your organization for success in the competitive global marketplace.
