Advanced Gold-Carborane Catalysis for Efficient Commercial Thiazole Production
Advanced Gold-Carborane Catalysis for Efficient Commercial Thiazole Production
The pharmaceutical and fine chemical industries are constantly seeking robust catalytic systems that can deliver high-purity heterocyclic intermediates under increasingly sustainable conditions. Patent CN111393480A introduces a significant breakthrough in this domain with the development of a novel gold complex containing a bisphosphine ortho-carborane ligand. This technology addresses critical pain points in the synthesis of 2,4-disubstituted thiazoles, which are privileged scaffolds found in numerous bioactive molecules and drug candidates. The core innovation lies in the unique structural stability imparted by the carborane cage, which protects the active gold center while maintaining high catalytic turnover.
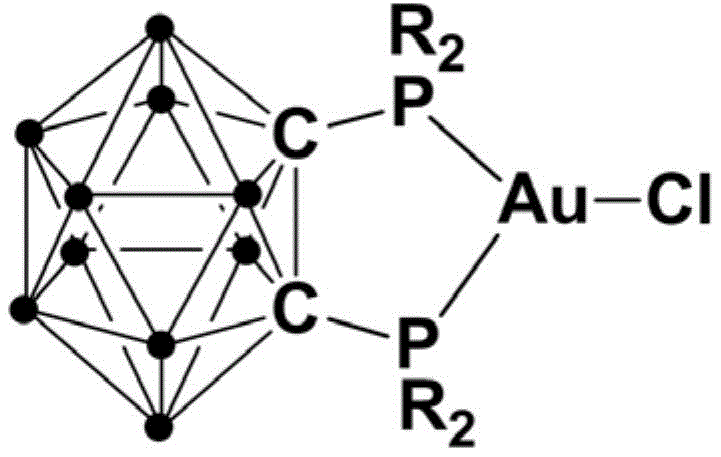
For R&D directors and process chemists, the implications of this patent are profound, offering a pathway to streamline the production of valuable thiazole derivatives. The complex is not only highly active but also remarkably stable against air and moisture, a rarity for monovalent gold species. This stability translates directly into operational simplicity, reducing the need for expensive glovebox techniques during catalyst handling. As a reliable pharmaceutical intermediates supplier, understanding such technological advancements allows us to offer superior process solutions that align with modern green chemistry principles while ensuring consistent product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4-disubstituted thiazoles has relied on methodologies that often suffer from significant operational drawbacks and inefficiencies. Traditional routes frequently necessitate the use of harsh reaction conditions, including elevated temperatures and strong acidic or basic environments, which can degrade sensitive functional groups on the substrate. Furthermore, many existing gold-catalyzed systems require stoichiometric amounts of oxidants or specific additives to activate the catalyst, leading to complex reaction mixtures and difficult purification processes. The instability of conventional Au(I) catalysts towards air and moisture often mandates the use of inert atmosphere techniques, driving up capital expenditure and operational costs in a manufacturing setting.
Another critical limitation is the narrow substrate scope observed in many legacy catalytic systems, where steric hindrance or electronic deactivation leads to poor conversion rates. This often forces process teams to develop custom synthetic routes for different analogs, fragmenting production lines and increasing lead times. The generation of heavy metal waste and the difficulty in removing trace gold residues from the final API intermediate are also persistent challenges that impact both environmental compliance and product safety profiles. These cumulative factors create a bottleneck in the cost reduction in fine chemical manufacturing, particularly for high-volume thiazole-based drugs.
The Novel Approach
The technology disclosed in CN111393480A offers a transformative alternative by utilizing a specifically designed bisphosphine ortho-carborane ligand to stabilize the gold center. This novel approach enables the cyclization of terminal alkynes and thioamides to proceed efficiently at room temperature, eliminating the energy costs associated with heating and the safety risks of high-pressure reactors. The catalyst operates with exceptional efficiency at low loadings, typically around 1.0 mol%, which drastically reduces the consumption of precious metal resources. Moreover, the reaction proceeds without the need for external additives, resulting in cleaner reaction profiles and simplified downstream processing.
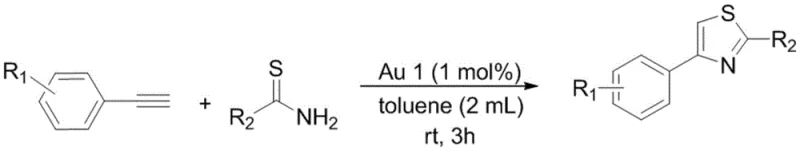
From a supply chain perspective, the robustness of this new catalyst system means that production can be scaled with greater confidence and continuity. The insensitivity to air and water allows for more flexible manufacturing schedules and reduces the risk of batch failures due to minor environmental fluctuations. This reliability is crucial for reducing lead time for high-purity API intermediates, ensuring that downstream drug development timelines are met without delay. The broad substrate tolerance further enhances the versatility of this method, allowing a single catalytic platform to produce a diverse library of thiazole compounds, thereby optimizing inventory management and resource allocation.
Mechanistic Insights into Au(I)-Carborane Catalyzed Cyclization
The exceptional performance of this catalytic system can be attributed to the unique electronic and steric properties of the ortho-carborane backbone. The three-dimensional aromaticity of the carborane cage provides a rigid scaffold that enforces a specific geometry around the gold center, preventing the formation of inactive oligomeric species that often plague gold catalysis. The phosphine groups attached to the carbon vertices of the cage act as strong sigma-donors, stabilizing the Au(I) oxidation state and facilitating the activation of the alkyne pi-system. This activation is the critical first step in the catalytic cycle, rendering the terminal alkyne susceptible to nucleophilic attack by the sulfur atom of the thioamide.
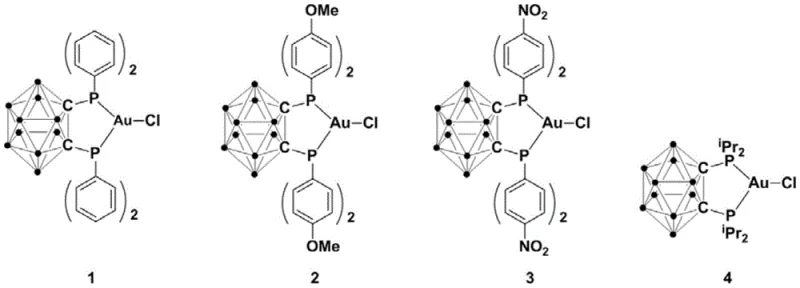
Regarding impurity control, the mechanism favors a direct cyclization pathway that minimizes side reactions such as alkyne polymerization or hydration. The steric bulk of the ligand shields the active site, ensuring high regioselectivity for the 2,4-disubstituted product over potential isomers. This intrinsic selectivity reduces the burden on purification units, as fewer byproducts are generated during the reaction. For quality control teams, this means a more consistent impurity profile across different batches, simplifying the validation process for regulatory filings. The stability of the catalyst also ensures that metal leaching is minimized, aiding in the removal of residual gold to meet stringent pharmaceutical specifications.
How to Synthesize Bisphosphine Ortho-Carborane Gold Complex Efficiently
The preparation of this advanced catalyst is designed for practicality, utilizing a straightforward one-pot synthetic strategy that avoids complex isolation steps for intermediates. The process begins with the lithiation of the ortho-carborane precursor, followed by immediate phosphorylation and subsequent metallation with gold chloride. This telescoped approach maximizes atom economy and minimizes solvent usage, aligning with green chemistry metrics. Detailed standard operating procedures for the synthesis involve precise control of temperature and stoichiometry to ensure high yield and purity of the final complex.
- Dropwise addition of n-BuLi to o-carborane solution at low temperature (-5 to 5°C) followed by stirring.
- Warming to room temperature and adding halogenated phosphine (e.g., ClPPh2) for ligand formation.
- Addition of AuCl to the system to form the final gold complex, followed by solvent removal and washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology represents a strategic opportunity to optimize cost structures and enhance operational resilience. The shift from multi-step, additive-heavy processes to a streamlined, room-temperature protocol fundamentally alters the cost equation for thiazole production. By eliminating the need for specialized anhydrous conditions and expensive reagents, the overall cost of goods sold (COGS) can be significantly reduced without compromising on quality. This efficiency gain is critical in a competitive market where margin pressure is constant.
- Cost Reduction in Manufacturing: The primary driver for cost savings is the drastic reduction in catalyst loading and the elimination of auxiliary additives. Traditional methods often require excess reagents to drive equilibrium, whereas this system achieves high conversion with minimal catalyst input. Furthermore, the ability to run reactions at room temperature removes the energy burden associated with heating large reactor vessels, leading to substantial utility savings. The simplified workup procedure, which often requires only filtration and solvent evaporation, reduces labor hours and solvent consumption, contributing to a leaner manufacturing process.
- Enhanced Supply Chain Reliability: The air and moisture stability of the catalyst mitigates the risk of supply disruptions caused by the degradation of sensitive reagents during storage or transport. This robustness allows for broader sourcing options for raw materials, as strict anhydrous grades are not always necessary for the reaction medium. Consequently, procurement teams can negotiate better terms with suppliers and maintain lower safety stock levels. The consistency of the catalyst performance ensures predictable production cycles, which is vital for meeting just-in-time delivery commitments to pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the mild reaction conditions and the absence of hazardous exotherms. The reduced generation of chemical waste, particularly heavy metal contaminants and acidic byproducts, simplifies wastewater treatment and lowers disposal costs. This environmental advantage supports corporate sustainability goals and ensures compliance with increasingly stringent global regulations. The commercial scale-up of complex heterocycles becomes less risky, enabling faster time-to-market for new drug candidates relying on this thiazole scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold-carborane catalytic system. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios.
Q: What are the stability characteristics of this gold complex compared to traditional catalysts?
A: Unlike many traditional Au(I) catalysts that require inert atmospheres, this bisphosphine carborane complex is insensitive to air and water, allowing for stable storage and handling without rigorous exclusion of moisture.
Q: Does this catalytic system require expensive additives or harsh conditions?
A: No, the process operates under mild conditions at room temperature without the need for additional acidic or basic additives, significantly simplifying the reaction setup and workup procedures.
Q: What is the substrate scope for the thiazole synthesis using this catalyst?
A: The catalyst demonstrates a wide substrate range, effectively converting various terminal alkynes (aryl, alkyl, cycloalkyl) and thioamides into 2,4-disubstituted thiazoles with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gold Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the bisphosphine carborane gold complex in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are successfully translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific thiazole synthesis needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can add value to your supply chain.
