Advanced Synthesis of Chiral Synthetic Nicotine for Industrial Scale-up
Advanced Synthesis of Chiral Synthetic Nicotine for Industrial Scale-up
The global demand for high-purity chiral synthetic nicotine has surged in recent years, driven primarily by the rapid expansion of the electronic cigarette industry and the pharmaceutical sector's need for safe, impurity-free active ingredients. Traditional extraction methods from tobacco plants are increasingly scrutinized due to the presence of carcinogenic tobacco-specific nitrosamines and other harmful compounds, alongside supply chain volatility caused by agricultural dependencies. Addressing these critical challenges, the Chinese patent CN113475740B discloses a groundbreaking preparation method that utilizes a novel asymmetric synthesis strategy to produce chiral nicotine with exceptional purity and enantiomeric excess. This technical insight report analyzes the proprietary methodology detailed in the patent, highlighting its potential to redefine the manufacturing landscape for reliable pharmaceutical intermediates suppliers seeking to optimize their production portfolios.
This innovative protocol represents a significant departure from legacy synthesis routes, offering a streamlined pathway that balances chemical efficiency with economic viability. By leveraging inexpensive and commercially abundant raw materials such as methyl nicotinate and gamma-butyrolactone, the process effectively circumvents the high costs associated with traditional chiral resolution techniques. For procurement managers and supply chain heads, this translates into a robust strategy for cost reduction in pharmaceutical intermediates manufacturing, ensuring a stable supply of high-quality material that meets stringent regulatory standards for human consumption without the baggage of plant-derived contaminants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to nicotine synthesis have been plagued by significant technical and economic hurdles that limit their applicability in modern large-scale production. Early methodologies, such as those utilizing pyrrolidine as a starting material, often result in racemic mixtures that require subsequent, costly resolution steps to isolate the biologically active S-enantiomer. Furthermore, alternative routes starting from nicotinic acid frequently necessitate the use of Grignard reagents, which are sensitive to moisture and oxygen, complicating the operational workflow and increasing safety risks in an industrial setting.
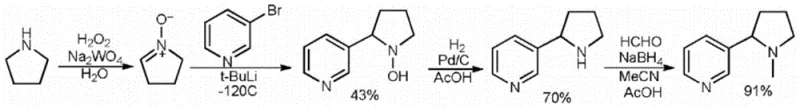
Another prevalent limitation in the prior art involves the use of 3-bromopyridine as a precursor, a route that demands cryogenic reaction conditions as low as -78°C. Such extreme temperatures impose a heavy burden on energy consumption and require specialized refrigeration equipment, drastically inflating capital expenditure and operational costs. Moreover, these conventional pathways often suffer from low overall yields and poor stereocontrol, resulting in products with suboptimal enantiomeric purity that fail to meet the rigorous specifications required for high-end pharmaceutical applications or premium electronic cigarette formulations.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the process outlined in patent CN113475740B introduces a highly efficient and stereoselective route that operates under significantly milder conditions. The core innovation lies in the strategic construction of the chiral center using chiral tert-butyl sulfinamide, which acts as a powerful stereochemical director to induce the formation of the desired configuration directly during the synthesis. This eliminates the need for post-synthesis resolution, thereby simplifying the workflow and enhancing the overall atom economy of the process.
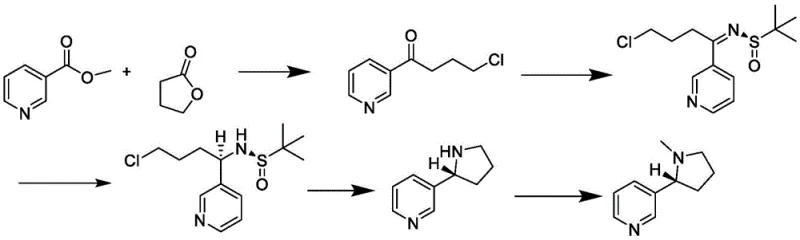
The new methodology utilizes a condensation reaction between methyl nicotinate and gamma-butyrolactone, followed by a ring-opening step to generate a key chloro-ketone intermediate. This intermediate is then subjected to chiral induction and cyclization, ultimately yielding chiral demethylated nicotine which is finally methylated to produce the target S-nicotine. The entire sequence is characterized by high yields, excellent enantiomeric excess values reaching up to 99%, and the use of standard laboratory equipment, making it exceptionally suitable for the commercial scale-up of complex organic molecules in a GMP-compliant environment.
Mechanistic Insights into Chiral Tert-Butyl Sulfinamide Induction
The cornerstone of this synthesis is the sophisticated application of chiral tert-butyl sulfinamide in Step S3, which serves as the pivotal moment for establishing stereochemical integrity. In this stage, the 4-chloro-1-(3-pyridine)-1-butanone intermediate reacts with the chiral sulfinamide in the presence of a titanate catalyst, such as tetraethyl titanate. The bulky tert-butyl group on the sulfinamide creates a specific steric environment that directs the nucleophilic attack or subsequent reduction in a highly facial-selective manner, ensuring that the newly formed chiral center possesses the correct absolute configuration required for biological activity.
This mechanism effectively bypasses the randomness of non-catalyzed reactions, providing a deterministic path to the S-enantiomer. The use of titanium alkoxides facilitates the formation of a chelated transition state, locking the substrate into a conformation that favors the formation of the desired diastereomer. Following this induction, the subsequent reduction with sodium borohydride and acid-mediated cyclization proceeds with retention of configuration, preserving the high optical purity established in the earlier step. This precise control over the molecular architecture is what allows the process to achieve an ee value of 99%, a critical metric for R&D directors focused on impurity profiles and product consistency.
How to Synthesize S-Nicotine Efficiently
The synthesis of S-nicotine via this patented route involves a carefully orchestrated sequence of five distinct chemical transformations, each optimized for maximum yield and minimal byproduct formation. The process begins with the condensation of nicotinate and gamma-butyrolactone under basic conditions, followed by acid-mediated ring opening to generate the linear ketone precursor. The critical chiral induction step utilizes sulfinamide chemistry to set the stereocenter, which is then locked in place through reductive cyclization and final N-methylation. For detailed operational parameters and safety protocols, the standardized synthesis steps are provided in the guide below.
- Perform condensation reaction between nicotinate and gamma-butyrolactone using an alkaline condensate like NaH in anhydrous tetrahydrofuran.
- Acidify the mixture and reflux to perform a ring-opening reaction, isolating 4-chloro-1-(3-pyridine)-1-butanone.
- React the ketone intermediate with chiral tert-butyl sulfinamide and titanate to induce chirality, forming the sulfinamide derivative.
- Reduce the sulfinamide derivative with a reducing agent like sodium borohydride and cyclize under acidic conditions to form chiral demethylated nicotine.
- Perform methylamination using formaldehyde and formic acid, followed by purification to obtain the final chiral nicotine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers transformative advantages that directly address the pain points of modern chemical procurement and supply chain management. By shifting away from agriculture-dependent extraction and towards a fully synthetic, petrochemical-derived pathway, manufacturers can decouple their production schedules from seasonal harvest fluctuations and geopolitical instability affecting tobacco crops. This transition ensures a continuous, predictable supply of raw materials, which is essential for maintaining the production velocity required by the fast-moving consumer goods sector, particularly in the volatile electronic cigarette market.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the substitution of expensive, specialized reagents with commodity chemicals. The use of methyl nicotinate and gamma-butyrolactone as starting materials significantly lowers the input cost compared to routes requiring precious metal catalysts or high-pressure hydrogenation equipment. Furthermore, the elimination of cryogenic conditions (-78°C) removes the need for energy-intensive cooling systems, leading to drastic reductions in utility costs and allowing for the use of standard glass-lined reactors, thereby optimizing capital allocation.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals enhances the resilience of the supply chain against disruptions. Unlike niche chiral catalysts that may have long lead times or single-source dependencies, the reagents used in this protocol, such as sodium hydride and sodium borohydride, are produced globally in massive quantities. This abundance ensures that production bottlenecks are minimized, reducing lead time for high-purity active ingredients and allowing suppliers to respond rapidly to spikes in market demand without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedures facilitate seamless scale-up from pilot plant to multi-ton production. The process avoids the generation of heavy metal waste streams associated with traditional catalytic hydrogenation, simplifying wastewater treatment and reducing the environmental footprint. This alignment with green chemistry principles not only lowers disposal costs but also ensures compliance with increasingly stringent environmental regulations, safeguarding the facility's operating license and corporate reputation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters and partners.
Q: Why is synthetic chiral nicotine preferred over tobacco-extracted nicotine?
A: Tobacco-extracted nicotine often contains carcinogenic tobacco-specific impurities that are harmful to human health. Synthetic routes, particularly the one described in CN113475740B, eliminate these impurities, ensuring a safer profile for pharmaceutical and electronic cigarette applications while allowing for consistent industrial production unaffected by climate or crop yields.
Q: How does this new method improve upon conventional racemic synthesis?
A: Conventional methods often produce racemic mixtures requiring expensive resolution steps or utilize harsh conditions like cryogenic temperatures (-78°C). This novel approach uses chiral tert-butyl sulfinamide to directly induce the desired stereochemistry, achieving high enantiomeric excess (ee value) without the need for complex resolution or extreme reaction conditions.
Q: What are the key cost drivers reduced in this synthesis pathway?
A: The process utilizes cheap and readily available starting materials such as methyl nicotinate and gamma-butyrolactone, avoiding expensive chiral metal catalysts or high-pressure hydrogenation equipment. Additionally, the mild reaction conditions reduce energy consumption and equipment maintenance costs associated with ultra-low temperature operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this advanced synthesis route to revolutionize the supply of chiral synthetic nicotine. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of S-nicotine meets the highest standards for enantiomeric excess and chemical purity required by global regulatory bodies.
We invite forward-thinking enterprises to collaborate with us to leverage this cutting-edge technology for their product lines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity pharmaceutical intermediates and specialty chemicals.
