Advanced Asymmetric Synthesis of Chiral Synthetic Nicotine for Industrial Scale-Up
Advanced Asymmetric Synthesis of Chiral Synthetic Nicotine for Industrial Scale-Up
The rapid expansion of the electronic cigarette industry has precipitated an unprecedented surge in demand for high-purity nicotine, specifically the biologically active S-enantiomer. While traditional extraction from tobacco plants remains a source, it is inherently limited by agricultural variables and the presence of harmful tobacco-specific nitrosamines. Addressing these critical supply chain and safety challenges, patent CN113475740B discloses a groundbreaking preparation method for chiral synthetic nicotine. This novel technical route leverages a sophisticated asymmetric synthesis strategy, utilizing chiral tert-butyl sulfinamide to induce stereochemistry, thereby bypassing the need for costly resolution steps. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards safer, more consistent, and economically viable manufacturing of this high-value alkaloid.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of nicotine has been plagued by significant technical hurdles that impede commercial viability. Early methodologies, such as those reported in the Journal of Organic Chemistry (1990), relied on pyrrolidine starting materials but resulted exclusively in racemic mixtures, necessitating complex and yield-lossing resolution processes. Furthermore, other established routes, such as the one depicted in reaction formula 3 involving 3-bromopyridine, impose severe operational burdens. These legacy processes often require cryogenic conditions, specifically ultra-low temperatures around -78°C, which demand specialized refrigeration infrastructure and drastically increase energy consumption. Additionally, the reliance on Grignard reagents introduces safety risks regarding moisture sensitivity and exothermic control, while the use of expensive chiral catalysts in other patented methods inflates the cost of goods sold (COGS), rendering them unsuitable for mass-market applications.
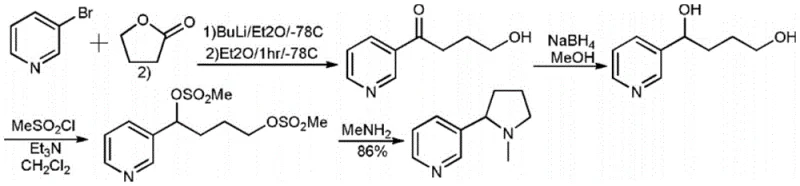
The Novel Approach
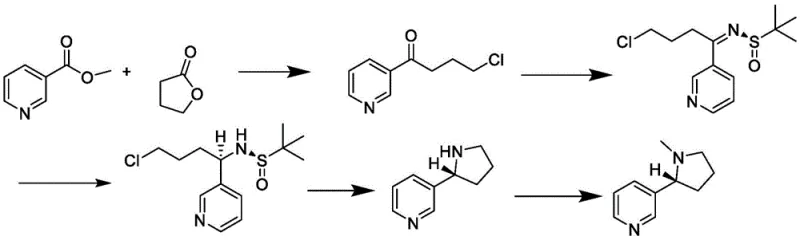
In stark contrast to these cumbersome legacy techniques, the methodology outlined in patent CN113475740B introduces a streamlined, five-step sequence that prioritizes operational simplicity and stereochemical precision. By initiating the synthesis with inexpensive and widely available nicotinate esters and gamma-butyrolactone, the process immediately establishes a lower cost baseline. The core innovation lies in the construction of the chiral center through the steric induction of chiral tert-butyl sulfinamide, rather than relying on post-synthesis resolution. This approach not only eliminates the need for ultra-low temperature reactors but also avoids the use of precious metal catalysts. The resulting pathway, as illustrated in the reaction scheme below, demonstrates a robust capability to produce S-nicotine with high enantiomeric excess and yield under mild reflux conditions, marking a significant advancement in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Chiral Tert-Butyl Sulfinamide Induction
The mechanistic elegance of this synthesis resides in the strategic application of Ellman's chiral auxiliary principles to construct the pyrrolidine ring with precise stereocontrol. In the critical third step of the process, 4-chloro-1-(3-pyridyl)-1-butanone reacts with chiral tert-butyl sulfinamide in the presence of a titanate catalyst, such as tetraethyl titanate. This condensation forms a chiral N-sulfinyl imine intermediate, where the bulky tert-butyl group creates a defined chiral environment. This steric hindrance effectively shields one face of the imine double bond, directing the subsequent nucleophilic attack during the reduction phase. When the reducing agent, preferably sodium borohydride, is introduced in step four, it approaches the imine carbon from the less hindered face, establishing the desired stereocenter with high fidelity. This diastereoselective reduction is the linchpin of the entire operation, ensuring that the resulting amine precursor possesses the correct configuration prior to cyclization.
Following the stereoselective reduction, the process employs an acid-catalyzed cyclization to close the pyrrolidine ring, forming chiral demethylated nicotine. The use of hydrogen halides, such as hydrobromic acid, facilitates the intramolecular nucleophilic substitution where the secondary amine displaces the chloro leaving group. This cyclization step is conducted under reflux conditions, typically around 80°C, which provides sufficient thermal energy to overcome the activation barrier without degrading the sensitive chiral integrity established in the previous step. Finally, the demethylated nicotine undergoes Eschweiler-Clarke methylation using formaldehyde and formic acid. This final transformation installs the N-methyl group to yield the target S-nicotine. The entire sequence is designed to minimize side reactions and racemization, ensuring that the final product maintains an ee value of up to 99%, a critical specification for high-purity OLED material or pharmaceutical grade intermediates where isomeric purity is paramount.
How to Synthesize Chiral Synthetic Nicotine Efficiently
The execution of this synthesis requires careful attention to stoichiometry and reaction conditions, particularly during the chiral induction phase. The patent details a specific protocol where the molar ratios of nicotinate, gamma-butyrolactone, and the base are optimized to maximize the yield of the initial condensation product. Subsequent steps involve precise pH adjustments and solvent selections, such as anhydrous tetrahydrofuran, to ensure the stability of the reactive intermediates. For process chemists looking to implement this technology, understanding the interplay between the titanate catalyst and the sulfinamide is crucial for reproducibility. The detailed standardized synthesis steps, including specific temperatures, reaction times, and workup procedures, are provided in the guide below to facilitate immediate technology transfer and pilot scale-up.
- Condensation of nicotinate and gamma-butyrolactone under alkaline conditions to form a ketone intermediate.
- Acidic ring-opening and separation of 4-chloro-1-(3-pyridyl)-1-butanone.
- Chiral induction reaction with tert-butyl sulfinamide and titanate to form the sulfinamide intermediate.
- Reduction and acid-catalyzed cyclization to generate chiral demethylated nicotine.
- Methylamination using formaldehyde and formic acid to yield final chiral nicotine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers compelling advantages that directly address the pain points of modern supply chain management. Traditional methods often rely on volatile agricultural supply chains or complex synthetic routes requiring specialized cryogenic infrastructure, both of which introduce significant risk and cost variability. By shifting to a fully synthetic route based on commodity chemicals like methyl nicotinate and gamma-butyrolactone, manufacturers can decouple production from seasonal agricultural fluctuations. This transition ensures a more predictable supply continuity, which is essential for maintaining long-term contracts with major e-cigarette and pharmaceutical clients. Furthermore, the elimination of expensive chiral metal catalysts and the avoidance of energy-intensive ultra-low temperature operations fundamentally alter the cost structure of production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost inputs with commodity-grade raw materials. Unlike prior art that necessitates expensive chiral ligands or resolution agents, this method utilizes chiral tert-butyl sulfinamide which, while a specialty chemical, is used in catalytic or stoichiometric amounts that are recoverable or cost-effective relative to the value of the final API. Moreover, the ability to run reactions at ambient or mild reflux temperatures (0°C to 80°C) rather than -78°C drastically reduces utility costs associated with cryogenic cooling. The simplified downstream processing, which avoids complex chiral chromatography, further contributes to substantial cost savings by reducing solvent consumption and processing time.
- Enhanced Supply Chain Reliability: The reliance on easily obtainable starting materials such as nicotinic acid esters and gamma-butyrolactone mitigates the risk of raw material shortages. These precursors are produced on a massive global scale for various industrial applications, ensuring a robust and diversified supply base. This contrasts sharply with methods dependent on niche brominated pyridines or biological extracts subject to harvest yields. By securing a synthesis route based on stable petrochemical derivatives, procurement managers can negotiate better long-term pricing and guarantee delivery schedules, thereby enhancing the overall resilience of the supply chain against market shocks.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations common in fine chemical manufacturing. The absence of pyrophoric reagents like Grignard reagents simplifies safety protocols and reduces the need for specialized containment equipment. Additionally, the high atom economy and the ability to recycle solvents like tetrahydrofuran and ethyl acetate align with green chemistry principles. This reduces the volume of hazardous waste generated, lowering disposal costs and ensuring compliance with increasingly stringent environmental regulations, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield optimization, purity standards, and operational feasibility. Understanding these nuances is vital for technical teams evaluating the integration of this process into existing manufacturing portfolios.
Q: What are the advantages of this chiral synthesis method over traditional extraction?
A: Unlike tobacco extraction which contains carcinogenic impurities and suffers from supply volatility, this chemical synthesis produces high-purity chiral nicotine free from tobacco-specific nitrosamines, ensuring consistent quality for pharmaceutical and vaping applications.
Q: How does this process achieve high enantiomeric excess (ee value)?
A: The process utilizes chiral tert-butyl sulfinamide as a chiral auxiliary. The steric hindrance of the tert-butyl group directs the stereochemistry during the imine formation and subsequent reduction, achieving ee values up to 99% without the need for expensive chiral chromatography resolution.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method avoids ultra-low temperature conditions (such as -78°C) and expensive transition metal catalysts required in prior art. It uses readily available raw materials like methyl nicotinate and operates under mild reflux conditions, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Synthetic Nicotine Supplier
The technological potential of this asymmetric synthesis route is immense, offering a clear pathway to high-volume production of premium S-nicotine. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and GC-MS systems to guarantee that every batch meets the exacting standards required by the global vaping and pharmaceutical industries.
We invite potential partners to engage with our technical procurement team to discuss how this novel synthesis route can optimize your supply chain. By leveraging our manufacturing capabilities, you can secure a stable source of high-purity chiral nicotine while minimizing production costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized process can meet your demand for reducing lead time for high-purity pharmaceutical intermediates.
