Advanced Ruthenium-Catalyzed Synthesis of 2,5-Disubstituted Furan Derivatives for Scalable Pharmaceutical Manufacturing
Introduction to Novel Furan Synthesis Technology
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the demand for more efficient and sustainable manufacturing processes. A pivotal advancement in this field is documented in patent CN110143935B, which discloses a highly effective preparation method for 2,5-disubstituted furan derivatives. These structural motifs are ubiquitous in bioactive molecules, serving as critical scaffolds in the development of pharmaceuticals, agrochemicals, and functional materials. The disclosed technology represents a significant leap forward by enabling the construction of new carbon-carbon bonds while simultaneously cleaving existing ones, ultimately leading to cyclization with exceptional regioselectivity. This approach addresses long-standing challenges in the field, offering a pathway that is not only chemically elegant but also practically viable for industrial scale-up. For R&D directors and procurement specialists alike, understanding the nuances of this ruthenium-catalyzed transformation is essential for securing a reliable supply chain of high-purity intermediates.
The significance of this patent lies in its ability to streamline the production of complex furan architectures that were previously difficult to access with high fidelity. Traditional methods often suffered from poor selectivity or required harsh conditions that compromised sensitive functional groups. In contrast, this new methodology operates under relatively mild thermal conditions and, crucially, tolerates an air atmosphere. This feature alone drastically simplifies the operational complexity of the reaction, removing the need for rigorous exclusion of oxygen and moisture that typically inflates production costs. By leveraging a specific combination of a ruthenium catalyst, a carboxylic acid additive, and a sulfur ylide reagent, the process achieves a level of efficiency that makes it highly attractive for the commercial manufacturing of active pharmaceutical ingredients (APIs) and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-disubstituted furans has been plagued by several intrinsic limitations that hindered their widespread adoption in large-scale industrial applications. Conventional routes often relied on multi-step sequences involving unstable intermediates or required the use of stoichiometric amounts of toxic heavy metals, which posed significant environmental and safety hazards. Furthermore, many traditional cyclization protocols demanded strictly anhydrous and anaerobic conditions, necessitating expensive infrastructure for nitrogen blanketing and specialized equipment to maintain inert atmospheres. These requirements not only increased the capital expenditure for manufacturing facilities but also extended the cycle time for each batch due to the meticulous degassing procedures involved. Additionally, older methods frequently exhibited poor regiocontrol, leading to mixtures of isomers that were difficult and costly to separate, thereby reducing the overall atom economy and increasing waste generation.
Another critical drawback of prior art techniques was the limited substrate scope, particularly when dealing with electron-deficient or sterically hindered aromatic rings. Many established protocols failed to deliver acceptable yields when applied to diverse substrates, forcing process chemists to develop custom solutions for each new target molecule. This lack of generality meant that supply chains were fragile and prone to disruption whenever a specific starting material varied in quality or availability. The reliance on hazardous solvents and aggressive reagents further complicated the post-treatment phase, often requiring extensive purification steps to meet the stringent purity specifications demanded by the pharmaceutical industry. Consequently, the cost of goods sold (COGS) for furan-based intermediates remained prohibitively high, limiting their utility in cost-sensitive markets such as generic drugs and agrochemicals.
The Novel Approach
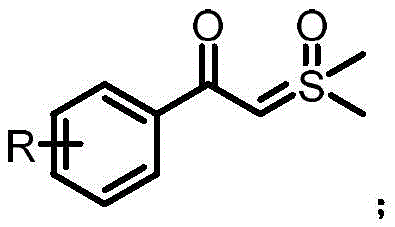
The methodology outlined in patent CN110143935B introduces a paradigm shift by utilizing a ruthenium-catalyzed system that operates effectively under ambient air conditions. This novel approach employs a p-cymene dichloride ruthenium(II) dimer as the catalyst, which works in synergy with 2,4,6-trimethylbenzoic acid as a crucial additive to facilitate the transformation. The reaction involves the interaction between a 1,3-diaryl-1,3-dione and a sulfur ylide, leading to the formation of the furan ring through a mechanism that involves both bond formation and bond cleavage. One of the most compelling aspects of this new route is its operational simplicity; the reaction can be conducted in common organic solvents like toluene at temperatures ranging from 100°C to 120°C without the need for inert gas protection. This elimination of inert atmosphere requirements translates directly into substantial cost reduction in pharmaceutical intermediate manufacturing by lowering energy consumption and equipment complexity.
Moreover, this innovative strategy demonstrates remarkable versatility, accommodating a wide array of substituents on the aromatic rings, including halogens, alkyl groups, and alkoxy moieties. The process exhibits strong reaction specificity, ensuring that the desired 2,5-disubstituted product is formed with high regioselectivity, thereby minimizing the formation of byproducts and simplifying downstream purification. The use of a sulfur ylide as a key reagent allows for the introduction of specific carbon fragments with precision, expanding the chemical space accessible to medicinal chemists. By combining high yields with mild reaction conditions and a green post-treatment protocol involving simple aqueous workups, this method sets a new standard for efficiency. It provides a robust platform for the commercial scale-up of complex heterocycles, ensuring a consistent and reliable supply of high-quality materials for global markets.
Mechanistic Insights into Ruthenium-Catalyzed Cyclization
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic intricacies of the ruthenium-catalyzed cyclization. The reaction initiates with the activation of the 1,3-dicarbonyl compound by the base, generating an enolate species that is poised for nucleophilic attack. The ruthenium catalyst plays a pivotal role in coordinating with the sulfur ylide and the activated diketone, facilitating a unique rearrangement process. Unlike traditional acid-catalyzed dehydrations, this transition metal-mediated pathway allows for the constructive cleavage of a carbon-carbon bond within the precursor framework while simultaneously forging a new carbon-carbon bond to close the furan ring. The presence of the 2,4,6-trimethylbenzoic acid additive is believed to stabilize key intermediates or assist in proton transfer steps, ensuring the catalytic cycle proceeds smoothly without deactivation of the metal center. This delicate balance of electronic and steric factors is what grants the reaction its high fidelity and broad substrate tolerance.
From an impurity control perspective, the mechanism inherently favors the formation of the thermodynamically stable furan product, suppressing competing side reactions that often plague non-catalytic methods. The regioselectivity is governed by the electronic nature of the substituents on the starting materials, with the catalyst directing the cyclization to occur at the most favorable positions. This precise control minimizes the generation of regioisomers, which are notoriously difficult to separate and often constitute major impurities in crude products. Furthermore, the use of an air atmosphere suggests that the catalytic cycle is robust against oxidation, likely due to the stability of the ruthenium(II) species under these conditions. Understanding these mechanistic details empowers process engineers to optimize reaction parameters such as temperature and concentration to maximize throughput while maintaining the stringent purity profiles required for regulatory compliance in drug substance manufacturing.
How to Synthesize 2,5-Disubstituted Furan Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to ensure optimal outcomes. The process begins with the careful selection of high-purity reagents, specifically the 1,3-diaryl-1,3-dione and the corresponding sulfur ylide, as their quality directly impacts the final yield. The reaction mixture is assembled by combining the base, typically lithium tert-butoxide, with the diketone, catalyst, additive, and ylide in a suitable solvent like toluene. It is critical to maintain the reaction temperature within the specified range of 100-120°C to drive the cyclization to completion without inducing thermal decomposition of sensitive components. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are provided in the guide below to assist technical teams in replicating this high-performance protocol.
- Combine lithium tert-butoxide, 1,3-diaryl-1,3-dione, ruthenium catalyst, additive, sulfur ylide, and toluene in a reaction vessel.
- Heat the mixture to 100-120°C under an air atmosphere for 12-24 hours to facilitate cyclization.
- Dilute with ethyl acetate, wash with water, dry the organic phase, and purify via chromatography to isolate the target furan derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic advantages that extend beyond mere chemical efficiency. The primary benefit lies in the drastic simplification of the manufacturing infrastructure required. By eliminating the necessity for inert gas systems, facilities can reduce their dependency on nitrogen or argon supplies, which are subject to market volatility and logistical constraints. This operational flexibility enhances supply chain reliability, allowing for continuous production runs without the interruptions associated with gas cylinder changes or pipeline maintenance. Furthermore, the use of toluene as a preferred solvent aligns with established recovery and recycling protocols in most chemical plants, minimizing waste disposal costs and environmental impact. The robustness of the reaction under air conditions also reduces the risk of batch failures due to accidental oxygen ingress, a common issue in sensitive organometallic processes.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the removal of expensive operational overheads. Since the reaction does not require an inert atmosphere, the capital investment for specialized reactors and gas handling systems is significantly lowered. Additionally, the high regioselectivity reduces the burden on purification units, meaning less solvent and stationary phase are consumed during chromatography or crystallization steps. The catalyst loading is relatively low, and the potential for catalyst recovery or the use of cost-effective ruthenium sources further drives down the raw material costs. These factors collectively contribute to a leaner cost structure, making the final furan derivatives more competitive in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: The wide substrate scope of this methodology ensures that supply chains remain resilient even when specific starting materials face shortages. Because the reaction tolerates various functional groups including halogens and alkoxy groups, manufacturers can source alternative precursors if primary suppliers encounter disruptions. The mild reaction conditions also mean that the process is less prone to runaway exotherms or safety incidents, ensuring consistent delivery schedules. This reliability is crucial for downstream customers who depend on just-in-time delivery of intermediates for their own production lines. By mitigating the risks associated with complex reaction setups, this technology fosters a more stable and predictable supply environment for critical pharmaceutical building blocks.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by its inherent safety and simplicity. The absence of pyrophoric reagents or extreme pressures makes it ideal for large-scale batch reactors commonly found in CDMO facilities. Moreover, the green nature of the post-treatment, which involves simple aqueous washes and standard drying agents, simplifies wastewater treatment and reduces the generation of hazardous waste streams. This alignment with green chemistry principles helps manufacturers meet increasingly stringent environmental regulations and sustainability goals. The ability to produce high volumes of material with a reduced environmental footprint positions this technology as a future-proof solution for the sustainable manufacturing of fine chemicals and active ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this furan synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their operations. Understanding these details is vital for assessing the feasibility of adopting this method for specific project requirements and for engaging in informed discussions with technical service providers.
Q: What catalyst system is employed in this furan synthesis?
A: The process utilizes a p-cymene dichloride ruthenium(II) dimer as the catalyst,配合 with 2,4,6-trimethylbenzoic acid as an additive and lithium tert-butoxide as the base.
Q: Does this reaction require an inert atmosphere?
A: No, a significant advantage of this patented method is that it proceeds efficiently under an air atmosphere, eliminating the need for costly nitrogen or argon protection.
Q: What is the typical yield range for these derivatives?
A: Depending on the substrate substituents, isolated yields typically range from 40% to 73%, with excellent regioselectivity observed across various aryl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Disubstituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN110143935B for the production of high-value heterocycles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the highest industry standards. Our state-of-the-art facilities are equipped to handle ruthenium-catalyzed reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications for every batch released. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process development team is ready to optimize this specific furan synthesis to match your unique volume and quality needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this efficient route can lower your overall production expenses. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your target molecules. Let us help you secure a stable, cost-effective, and high-quality supply of 2,5-disubstituted furan derivatives to accelerate your drug development timelines.
