Advanced Palladium-Catalyzed Cyclization for Commercial Scale-Up of Complex Heterocycles and High-Purity Furan Derivatives
Advanced Palladium-Catalyzed Cyclization for Commercial Scale-Up of Complex Heterocycles and High-Purity Furan Derivatives
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient and sustainable pathways to access critical heterocyclic scaffolds. A pivotal advancement in this domain is documented in patent CN102120735B, which discloses a highly efficient method for preparing substituted furans containing 2,5-di-substituents. This technology represents a significant leap forward for the industry, offering a robust alternative to classical methodologies by utilizing a palladium-catalyzed cyclization of 1,3-conjugated diynes with water. For R&D directors and procurement specialists alike, understanding the nuances of this patent is crucial, as it unlocks new possibilities for cost reduction in fine chemical manufacturing while ensuring the delivery of high-purity furan derivatives. The method operates under inert atmospheres using specific catalyst systems, ligands, and inorganic bases, providing a versatile platform for synthesizing a wide array of aromatic and aliphatic furan compounds that are essential building blocks in pharmaceuticals and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of furan rings has relied heavily on the Paal-Knorr synthesis, a century-old methodology that involves the acid-catalyzed cyclocondensation of 1,4-dicarbonyl compounds. While foundational, this traditional approach suffers from several inherent drawbacks that limit its utility in modern high-throughput manufacturing environments. The requirement for strongly acidic conditions often leads to compatibility issues with acid-sensitive functional groups, necessitating additional protection and deprotection steps that inflate both time and material costs. Furthermore, the preparation of the requisite 1,4-dicarbonyl precursors can be synthetically demanding, often involving multiple steps and generating significant amounts of chemical waste. Recent modifications involving microwave assistance or alternative acid catalysts have attempted to mitigate some issues, yet they still fundamentally rely on the availability of specific dicarbonyl starting materials, creating a bottleneck in the supply chain for complex substituted furans needed in advanced drug discovery programs.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN102120735B introduces a streamlined, one-step catalytic system that directly converts 1,3-conjugated diynes into 2,5-disubstituted furans. This novel approach leverages the power of transition metal catalysis to activate the alkyne moieties, facilitating a cyclization reaction with water that proceeds with remarkable efficiency and selectivity. By bypassing the need for pre-oxidized dicarbonyl precursors, this method drastically simplifies the synthetic route, reducing the overall step count and minimizing the accumulation of impurities. The reaction conditions are notably mild, typically operating between 80°C and 140°C, which preserves the integrity of sensitive substituents on the aromatic rings. This shift in paradigm not only enhances the chemical feasibility of producing diverse furan libraries but also aligns perfectly with the goals of green chemistry by reducing solvent usage and waste generation, making it an attractive option for reliable pharmaceutical intermediates supplier networks seeking sustainable solutions.
Mechanistic Insights into Pd-Catalyzed Cyclization
The core of this transformative synthesis lies in the sophisticated interplay between the palladium catalyst, the ligand environment, and the substrate. The reaction initiates with the coordination of the palladium species, preferably Pd(PPh3)4, to the electron-rich triple bonds of the 1,3-conjugated diyne. This coordination activates the alkyne towards nucleophilic attack, a critical step that is further enhanced by the presence of unsaturated ligands such as norbornadiene. The norbornadiene ligand plays a pivotal role in stabilizing the active catalytic species and facilitating the necessary electronic rearrangements required for ring closure. As the reaction progresses, water acts as the oxygen source, attacking the activated alkyne complex to form a vinyl-palladium intermediate. This step is crucial as it determines the regioselectivity of the oxygen incorporation, ensuring that the furan ring is formed exclusively at the 2,5-positions rather than other potential isomers.
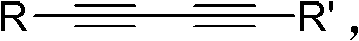
Following the nucleophilic attack, the mechanism proceeds through a series of proton transfers and reductive elimination steps that ultimately release the furan product and regenerate the active palladium catalyst. The use of strong inorganic bases, such as potassium hydroxide or sodium hydroxide, is essential to neutralize the protons generated during the cyclization and to maintain the catalytic cycle's momentum. The choice of solvent, particularly polar aprotic solvents like 1,4-dioxane or non-polar solvents like toluene, influences the solubility of the inorganic base and the stability of the transition states. This delicate balance of components ensures that the reaction proceeds with high turnover numbers, even for sterically hindered substrates. The structural versatility of the starting 1,3-diynes, as depicted in the general formula, allows for the introduction of various aryl, alkyl, or heteroaryl groups, enabling the synthesis of a vast chemical space of furan derivatives tailored for specific biological activities.
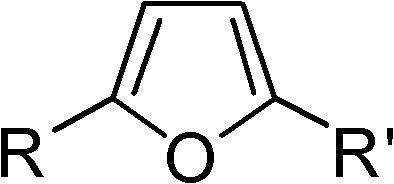
Furthermore, the impurity profile of this reaction is exceptionally clean due to the high selectivity of the palladium system. Unlike acid-catalyzed methods that often produce polymeric byproducts or dehydration side-products, this catalytic cycle is tightly controlled by the ligand sphere around the metal center. The inert atmosphere, typically maintained using nitrogen, prevents the oxidation of the phosphine ligands and the palladium metal, which could otherwise lead to catalyst deactivation and the formation of palladium black. This stability is paramount for commercial operations where batch-to-batch consistency is non-negotiable. By understanding these mechanistic details, process chemists can fine-tune reaction parameters such as temperature and catalyst loading to optimize yields, which have been demonstrated to reach as high as 96% in specific embodiments. This level of control translates directly into reduced purification burdens and higher overall process efficiency.
How to Synthesize 2,5-Disubstituted Furan Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational protocols to maximize yield and safety. The general procedure involves charging a sealable reactor, such as a glass pressure tube, with the 1,3-conjugated diyne substrate, the palladium catalyst, the chosen ligand, and the inorganic base. It is imperative to establish an inert environment prior to heating; this is typically achieved by purging the vessel with nitrogen gas multiple times to displace atmospheric oxygen, which acts as a poison for the low-valent palladium species. Once the atmosphere is secured, the organic solvent is added, and the mixture is sealed tightly to prevent solvent loss at elevated temperatures. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometric ratios and thermal profiles required for optimal performance across different substrate classes.
- Prepare the reaction mixture by combining the 1,3-conjugated diyne substrate, palladium catalyst (such as Pd(PPh3)4), ligand, and inorganic base in a suitable organic solvent like 1,4-dioxane.
- Ensure an inert atmosphere by purging the reaction vessel with nitrogen gas to prevent catalyst oxidation and maintain high catalytic activity throughout the process.
- Heat the sealed reaction mixture to temperatures between 80°C and 140°C for 4 to 24 hours, then cool and isolate the high-purity furan product via standard workup procedures.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed methodology offers profound advantages that extend beyond mere chemical elegance. For procurement managers and supply chain heads, the primary value proposition lies in the drastic simplification of the raw material supply chain. Traditional routes often depend on specialized 1,4-dicarbonyl compounds that may have limited suppliers and long lead times. In contrast, 1,3-conjugated diynes are increasingly accessible through established coupling chemistries, and the use of water as a reagent eliminates the need for expensive oxidants or dehydrating agents. This shift significantly reduces the complexity of sourcing, thereby enhancing supply chain reliability and mitigating the risk of production stoppages due to material shortages. Moreover, the one-step nature of the reaction consolidates multiple processing stages into a single unit operation, which inherently lowers capital expenditure and operational overheads associated with intermediate isolation and handling.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of costly precursor synthesis and the reduction of waste disposal costs. By avoiding the use of strong mineral acids and the subsequent neutralization steps required in Paal-Knorr synthesis, the process generates significantly less saline waste, leading to substantial cost savings in wastewater treatment. Additionally, the high catalytic efficiency means that lower loadings of palladium can be utilized while maintaining high yields, optimizing the consumption of this precious metal. The ability to run the reaction in common solvents like toluene or dioxane further contributes to cost reduction in fine chemical manufacturing, as these solvents are readily available in bulk quantities and can often be recovered and recycled efficiently.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system ensures consistent production output, which is critical for maintaining uninterrupted supply to downstream pharmaceutical clients. The tolerance of the reaction to various functional groups means that a single set of conditions can be applied to a wide range of substrates, reducing the need for extensive process re-validation when switching between different furan derivatives. This flexibility allows manufacturers to respond rapidly to changing market demands without the delays associated with developing new synthetic routes for each new compound. Consequently, reducing lead time for high-purity furan derivatives becomes a tangible reality, enabling faster time-to-market for new drug candidates that rely on these heterocyclic cores.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is straightforward due to the absence of hazardous reagents and the use of standard heating equipment. The reaction does not require extreme pressures or cryogenic temperatures, making it compatible with existing multipurpose reactors in most fine chemical facilities. From an environmental standpoint, the atom economy of incorporating water into the final product is excellent, and the lack of stoichiometric oxidants aligns with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on the manufacturer and enhances the sustainability profile of the final API intermediate, a factor that is becoming increasingly important for major pharmaceutical buyers evaluating their supplier base.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical implementation and commercial viability of this synthesis, we have compiled a set of questions based on the specific details found in the patent literature. These answers provide clarity on catalyst selection, reaction conditions, and the scope of substrates that can be successfully converted. Understanding these facets is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The following responses are derived directly from the experimental data and claims presented in the intellectual property documentation.
Q: What are the primary advantages of this Pd-catalyzed method over traditional Paal-Knorr synthesis?
A: Unlike the traditional Paal-Knorr synthesis which requires harsh acidic conditions and pre-functionalized 1,4-dicarbonyl precursors, this novel method utilizes readily available 1,3-conjugated diynes and water in a one-step process. It operates under milder conditions with higher selectivity and avoids the generation of excessive acidic waste, significantly simplifying the downstream purification process for pharmaceutical applications.
Q: Which catalyst systems demonstrate the highest efficiency for this cyclization reaction?
A: Experimental data indicates that tetrakis(triphenylphosphine)palladium(0), or Pd(PPh3)4, exhibits superior catalytic activity compared to other palladium sources like Pd(OAc)2. When combined with norbornadiene as a ligand and strong inorganic bases such as potassium hydroxide, the system achieves isolated yields exceeding 90% for various aromatic substrates, ensuring robust performance for commercial manufacturing.
Q: Is this synthetic route scalable for industrial production of furan intermediates?
A: Yes, the process is highly amenable to scale-up due to its operational simplicity and the use of common organic solvents like 1,4-dioxane or toluene. The reaction tolerates a broad range of functional groups on the diyne substrate, allowing for the versatile production of diverse furan derivatives without requiring complex protection-deprotection strategies, thereby enhancing supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Disubstituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced heterocyclic intermediates in the development of next-generation therapeutics and agrochemicals. Our technical team has thoroughly analyzed the potential of the Pd-catalyzed cyclization route described in CN102120735B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless and efficient. Our facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2,5-disubstituted furan meets the exacting standards required for GMP manufacturing environments. We are committed to delivering high-purity furan derivatives that empower your research and production pipelines.
We invite you to collaborate with us to explore how this innovative synthesis can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and target specifications. By partnering with us, you gain access to specific COA data and route feasibility assessments that validate the commercial viability of this approach for your supply chain. Contact us today to discuss how we can support your demand for reliable pharmaceutical intermediates supplier services and drive value through chemical innovation.
