Revolutionizing Amino Acid Derivative Production via Electrochemical Activation and Scale-Up
Revolutionizing Amino Acid Derivative Production via Electrochemical Activation and Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and safe methodologies for synthesizing complex building blocks. Patent CN1768030A introduces a groundbreaking electrochemical process for producing amino acid derivatives, specifically targeting non-natural beta-amino acids and their homologs. This technology represents a significant paradigm shift from traditional chemical oxidation methods, utilizing anodic activation to functionalize the alpha-position of protected amines. By replacing hazardous stoichiometric oxidants with electrons, this method offers a cleaner, more controllable pathway to high-value intermediates used in peptide therapeutics and drug discovery. The process demonstrates exceptional versatility, accommodating various protecting groups and enabling the efficient construction of carbon-carbon bonds adjacent to nitrogen atoms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specific amino acid derivatives, such as 2-pyrrolidineacetic acid analogs, relied heavily on methods described in older literature like US Patent 3,891,616. These conventional routes typically necessitated the use of natural amino acids as starting materials, which were then transformed using dangerous reagents such as diazomethane. The use of diazomethane presents severe safety hazards due to its toxicity and explosive potential, requiring specialized infrastructure and rigorous safety protocols that drive up operational costs. Furthermore, these harsh chemical conditions often carry a high risk of racemization, compromising the stereochemical integrity of the product. The need for cryogenic conditions and the generation of significant chemical waste further diminish the economic and environmental viability of these legacy processes for modern commercial manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in CN1768030A leverages electrochemical oxidation to activate the alpha-position of protected amines under mild conditions. This method eliminates the need for explosive reagents and allows for the direct introduction of functional groups via nucleophilic capture. The process begins with the anodic oxidation of an N-protected amine or amino acid in the presence of a nucleophile, such as methanol, to form an alpha-alkoxy intermediate. This activated species is then subjected to reaction with carbanionic reagents containing unsaturated groups, facilitating chain elongation. Finally, oxidative cleavage of the unsaturated group yields the desired amino acid derivative. This sequence not only improves safety profiles but also enhances overall yield and purity, making it an attractive option for reliable pharmaceutical intermediate supplier networks seeking sustainable solutions.
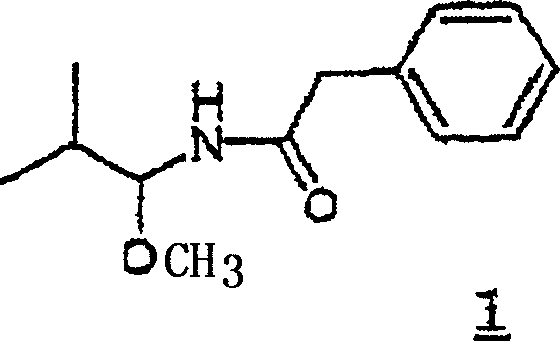
Mechanistic Insights into Electrochemical Alpha-Functionalization
The core of this technology lies in the Shono-type electrochemical oxidation mechanism. In the first step, the protected amine substrate undergoes single-electron oxidation at the anode to form a radical cation. Subsequent deprotonation at the alpha-carbon generates an alpha-amino radical, which is further oxidized to an iminium ion. In the presence of a nucleophile like methanol, this highly reactive iminium species is trapped to form a stable alpha-methoxy amine. This electrochemical activation is remarkably selective and occurs at ambient or slightly cooled temperatures, preserving sensitive functional groups elsewhere in the molecule. The choice of electrode material, typically platinum or graphite, and the supporting electrolyte are critical parameters that influence the efficiency of electron transfer and minimize side reactions.
Following activation, the synthetic route proceeds with a Lewis acid-catalyzed nucleophilic substitution. The alpha-methoxy group acts as a leaving group when activated by catalysts such as titanium tetrachloride or boron trifluoride etherate. This allows for the attack of allyl silanes or other carbon nucleophiles, effectively extending the carbon skeleton by three or more atoms while maintaining the nitrogen protection. The resulting unsaturated amine contains an olefinic double bond strategically positioned for the final transformation. The final step involves the oxidative cleavage of this double bond, typically via ozonolysis followed by an oxidative workup with hydrogen peroxide. This converts the terminal alkene directly into a carboxylic acid moiety, completing the synthesis of the beta-amino acid derivative with high atom economy.
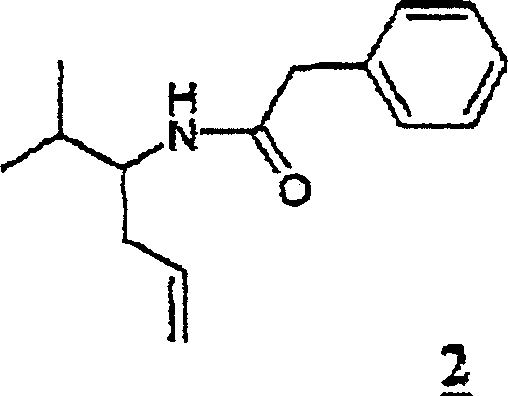
How to Synthesize Beta-Homovaline Derivatives Efficiently
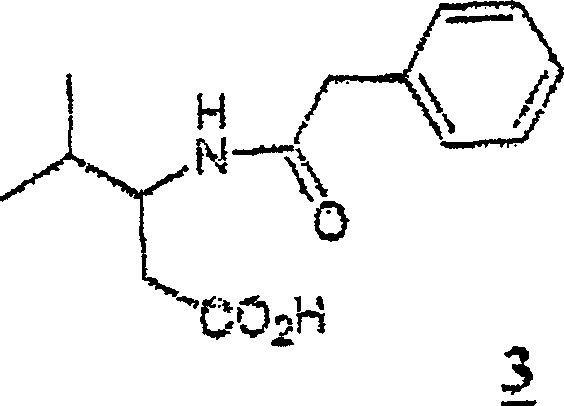
The synthesis of beta-homovaline and related structures serves as a prime example of this platform technology's utility. The process starts with the electrochemical methoxylation of N-phenylacetyl valine in methanol, achieving high conversion rates with minimal byproduct formation. The resulting alpha-methoxy intermediate is then reacted with allyltrimethylsilane in dichloromethane at low temperatures to install the allyl group. Following isolation, the allyl chain is cleaved using ozone at cryogenic temperatures, followed by oxidation to the acid. This streamlined sequence avoids the pitfalls of multi-step protection-deprotection strategies common in classical organic synthesis. For detailed operational parameters, electrode configurations, and workup procedures, please refer to the standardized synthesis guide below.
- Perform anodic oxidation of N-protected amino acids in methanol to generate alpha-methoxy intermediates.
- React the activated intermediate with allyltrimethylsilane using a Lewis acid catalyst to introduce the carbon chain.
- Execute oxidative cleavage of the olefinic bond via ozonolysis followed by oxidative workup to yield the final carboxylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical methodology offers substantial strategic benefits beyond mere technical feasibility. The elimination of hazardous reagents like diazomethane drastically simplifies regulatory compliance and reduces the capital expenditure associated with safety infrastructure. By utilizing electricity as a primary reagent, the process decouples production from the volatility of specialized chemical feedstock markets, leading to more predictable cost structures. Furthermore, the high yields reported in the patent examples, often exceeding 90% for individual steps, translate directly into reduced raw material consumption and lower waste disposal costs. This efficiency is crucial for cost reduction in API manufacturing, where margin pressures are intense.
- Cost Reduction in Manufacturing: The electrochemical process inherently reduces the cost of goods sold by minimizing reagent expenses. Traditional oxidation methods often require expensive stoichiometric oxidants that generate heavy metal waste, necessitating costly remediation. In this novel route, electrons serve as the oxidant, and the supporting electrolytes can often be recycled. Additionally, the high selectivity of the electrochemical step reduces the burden on downstream purification, lowering solvent usage and energy consumption during crystallization and chromatography. These factors combine to create a leaner, more cost-effective production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available starting materials such as natural amino acids and common silanes. Unlike processes dependent on exotic catalysts or unstable intermediates, this method relies on robust chemistry that can be sourced from multiple vendors. The modular nature of electrochemical reactors also allows for flexible production scheduling; capacity can be increased by adding more cell stacks rather than building entirely new reactor vessels. This scalability ensures that suppliers can respond rapidly to fluctuations in demand, reducing lead time for high-purity pharmaceutical intermediates and preventing stockouts.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with green chemistry principles. The absence of heavy metal oxidants and the potential for solvent recycling significantly reduce the environmental footprint. Electrochemical flow cells, which are ideal for scaling this chemistry, offer superior heat and mass transfer compared to batch reactors, ensuring consistent quality at the 100 MT scale. The final enzymatic resolution step using penicillin acylase further underscores the commitment to biocatalysis, offering a renewable and biodegradable alternative to chemical resolution agents. This makes the process highly attractive for companies aiming to meet strict ESG (Environmental, Social, and Governance) targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a realistic view of the technology's capabilities. Understanding these nuances is essential for R&D teams evaluating process transfer and for procurement specialists assessing vendor qualifications.
Q: What are the safety advantages of this electrochemical method over traditional diazomethane routes?
A: Traditional methods often rely on hazardous reagents like diazomethane which pose explosion risks and require specialized containment. This electrochemical process utilizes electricity as a clean reagent, operating under mild temperatures and avoiding toxic heavy metals or explosive precursors, significantly enhancing operational safety.
Q: How does this process ensure high stereochemical purity for pharmaceutical applications?
A: The process allows for the production of racemic mixtures which can subsequently be resolved using highly specific enzymatic methods, such as penicillin acylase hydrolysis. This enzymatic resolution step provides excellent enantiomeric excess (>99% ee), meeting the stringent purity requirements for active pharmaceutical ingredients.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the electrochemical steps are highly amenable to scale-up using modern flow electrolysis cells, which improve mass transfer and heat management. Combined with standard downstream processing like crystallization and chromatography, the route supports production from pilot kilogram scales up to multi-ton annual capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Homovaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of complex amino acid derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art electrochemical flow reactors and rigorous QC labs capable of handling sensitive intermediates with stringent purity specifications. We understand that transitioning from bench-scale chemistry to industrial manufacturing requires deep expertise in process safety and optimization, areas where our technical team excels.
We invite global partners to collaborate with us to leverage this advanced technology for your pipeline projects. Whether you require custom synthesis of beta-amino acids or scale-up of existing electrochemical routes, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable supply of high-quality chemical intermediates.
