Advanced Enzymatic Synthesis of Chiral Carbamate Intermediates for CNS Therapeutics Commercialization
Introduction to Novel Carbamate Synthesis Technology
The pharmaceutical industry's relentless pursuit of safer and more effective Central Nervous System (CNS) therapeutics has driven significant innovation in chiral intermediate synthesis. Patent CN1216858C represents a pivotal advancement in this domain, detailing a robust methodology for producing enantiomerically pure carbamate compounds. These compounds, specifically (S)-2-aryl-1,3-propanediol monocarbamates, exhibit potent pharmacological activity against conditions such as epilepsy, nervous myalgia, and cerebral apoplexy. Unlike traditional racemic mixtures which often require complex and yield-lossing resolution steps, this patented approach leverages a sophisticated combination of chemical phosgenation and biocatalytic hydrolysis. By integrating these distinct chemical disciplines, the process ensures not only high stereochemical fidelity but also operational efficiency that is critical for modern drug manufacturing. The technology addresses the growing regulatory demand for single-enantiomer drugs by providing a pathway that consistently delivers optical purities exceeding 99%, thereby reducing the toxicological burden associated with unwanted isomers.
Furthermore, the versatility of this synthetic route allows for the incorporation of diverse aryl substituents, ranging from simple phenyl groups to complex heterocyclic systems like pyridines. This structural flexibility is essential for medicinal chemists aiming to optimize the pharmacokinetic profiles of lead candidates. The patent underscores the importance of maintaining strict control over reaction parameters, particularly temperature and pH, to preserve the integrity of the chiral center throughout the synthesis. For global procurement teams and R&D directors, understanding the nuances of this process is vital, as it offers a reliable alternative to older, less efficient methods. The ability to scale this chemistry from laboratory benchtops to multi-ton commercial production without compromising purity makes it a highly attractive asset for any organization focused on CNS drug development. Consequently, this technology stands as a benchmark for quality and efficiency in the synthesis of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbamate derivatives for CNS applications relied heavily on non-selective chemical reactions that produced racemic mixtures. These conventional pathways often necessitated subsequent resolution steps, such as chiral chromatography or diastereomeric salt formation, which are inherently inefficient and costly. The reliance on harsh chemical reagents and extreme conditions in traditional methods frequently led to the degradation of sensitive functional groups, resulting in lower overall yields and a complex impurity profile. Moreover, the use of heavy metal catalysts in some older protocols introduced significant challenges regarding residual metal contamination, requiring extensive and expensive purification processes to meet stringent regulatory standards. The environmental footprint of these legacy methods was also substantial, generating large volumes of hazardous waste that complicated disposal and compliance. For supply chain managers, these inefficiencies translated into longer lead times and higher volatility in raw material costs, making it difficult to guarantee consistent supply for clinical and commercial needs.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a strategic enzymatic resolution that fundamentally alters the economic and technical landscape of carbamate production. By employing hydrolases such as Porcine Liver Esterase (PLE), the process achieves exquisite stereoselectivity under mild, aqueous conditions, effectively bypassing the need for aggressive chemical resolution. This biocatalytic step not only enhances the optical purity of the final product but also simplifies the workup procedure, as the enzyme can be easily removed via filtration. The integration of a phosgenation step at controlled low temperatures ensures the efficient formation of the carbamate linkage without compromising the chiral integrity established in previous steps. This hybrid chemical-biological strategy significantly reduces the number of unit operations required, leading to a more streamlined and cost-effective manufacturing process. For stakeholders focused on sustainability and cost reduction, this method offers a compelling value proposition by minimizing waste generation and maximizing atom economy. The result is a high-purity intermediate that is ready for downstream formulation with minimal additional processing.
Mechanistic Insights into Enzymatic Hydrolysis and Phosgenation
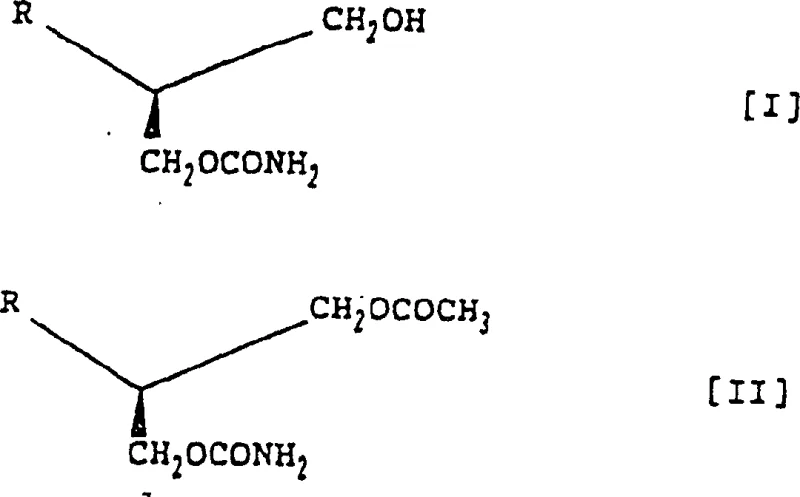
The core of this innovative synthesis lies in the precise orchestration of two distinct reaction mechanisms: low-temperature phosgenation followed by enzyme-catalyzed hydrolysis. The initial step involves the reaction of (R)-3-acetoxy-2-arylpropanol with phosgene in a solvent system comprising aromatic hydrocarbons and halocarbons, typically toluene and dichloromethane. The presence of a sterically hindered amine base, such as triethylamine or antipyrine, is crucial for scavenging the hydrogen chloride generated during the formation of the chloroformate intermediate. Maintaining the reaction temperature between -30°C and 0°C is imperative to prevent side reactions and ensure the stability of the reactive intermediates. Subsequent treatment with anhydrous ammonia at even lower temperatures (-70°C to 0°C) converts the chloroformate into the stable carbamate derivative. This sequence establishes the foundational structure while preserving the stereochemistry of the starting material.
Following the chemical synthesis of the acetoxy intermediate, the process transitions to a biocatalytic phase that is critical for achieving the desired (S)-configuration. The enzymatic hydrolysis is conducted in a phosphate buffer solution maintained at a neutral pH of approximately 7, which is optimal for the activity and stability of the hydrolase. The enzyme selectively cleaves the acetoxy group at the 3-position of the propanol chain, transforming the intermediate into the target (S)-2-aryl-1,3-propanediol monocarbamate. This selectivity is governed by the specific active site geometry of the enzyme, which discriminates between the enantiomers with high fidelity. The reaction is typically completed within 4 to 5 hours at temperatures ranging from 0°C to 35°C, with careful monitoring to prevent thermal denaturation of the biocatalyst. The use of water as the primary reaction medium in this step further enhances the environmental profile of the process, eliminating the need for large volumes of organic solvents during the critical stereo-defining transformation.
How to Synthesize (S)-2-Aryl-1,3-Propanediol Monocarbamate Efficiently
Implementing this synthesis route requires a disciplined approach to process control, particularly regarding temperature management and reagent stoichiometry. The initial phosgenation step demands rigorous exclusion of moisture and precise dosing of phosgene to ensure complete conversion while minimizing safety risks. Operators must be trained to handle the exothermic nature of the reaction and the toxicity of the reagents involved. Once the intermediate is formed, the transition to the enzymatic step involves a change in solvent system, typically requiring the removal of organic solvents and suspension of the substrate in the aqueous buffer. The choice of enzyme is critical; while PLE is preferred for its high selectivity, other lipases like PPL or PSL can be evaluated depending on the specific aryl substituent. Monitoring the reaction progress via HPLC or TLC is essential to determine the endpoint, ensuring that hydrolysis is complete before the enzyme is filtered off. This detailed protocol ensures that the final product meets the stringent purity specifications required for pharmaceutical applications.
- React (R)-3-acetoxy-2-arylpropanol with phosgene in a mixture of aromatic hydrocarbon and halocarbon solvents at -30 to 0°C in the presence of an amine base.
- Treat the resulting chloroformate intermediate with anhydrous ammonia at temperatures between -70 to 0°C to form the (S)-3-acetoxy-2-arylpropanol carbamate.
- Perform enzymatic hydrolysis using a hydrolase such as Porcine Liver Esterase (PLE) in a phosphate buffer at pH 7 and 0-35°C to yield the final high-purity monocarbamate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant reduction of manufacturing complexity, which directly correlates to lower operational costs and improved margin potential. By eliminating the need for multiple recrystallization steps or chiral chromatography columns often associated with racemic resolutions, the process drastically reduces the consumption of expensive stationary phases and solvents. This streamlining of the purification workflow not only accelerates the production cycle but also minimizes the physical footprint required for manufacturing, allowing for higher throughput within existing facilities. Furthermore, the high yield and optical purity achieved in each batch reduce the volume of off-spec material that must be reworked or discarded, contributing to a more sustainable and economically viable operation. These efficiencies make the technology highly attractive for companies seeking to optimize their cost of goods sold (COGS) in a competitive market environment.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reliance on readily available biocatalysts significantly lowers the cost of raw materials and waste treatment. Enzymes can often be recovered or used in immobilized forms, further enhancing cost efficiency over multiple batches. The mild reaction conditions reduce energy consumption associated with heating and cooling, leading to substantial utility savings. Additionally, the simplified workup procedures decrease labor hours and equipment utilization time, driving down the overall manufacturing overhead. These cumulative savings create a robust economic model that supports competitive pricing strategies for the final API.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents like toluene and ethyl acetate ensures that raw material sourcing is not subject to the volatility of specialized reagent markets. The robustness of the enzymatic step provides a buffer against minor variations in feedstock quality, ensuring consistent output even when supply chains face disruptions. The ability to produce high-purity intermediates with minimal impurities reduces the risk of batch failures during downstream API synthesis, thereby securing the continuity of supply for finished drug products. This reliability is crucial for maintaining long-term contracts with pharmaceutical partners who prioritize supply security.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram-scale laboratory experiments to multi-kilogram pilot runs without loss of efficiency. The aqueous nature of the key hydrolysis step aligns well with green chemistry principles, reducing the generation of hazardous organic waste streams. This environmental compatibility simplifies regulatory compliance and permitting processes, facilitating faster deployment of new production lines. The ease of scaling ensures that the technology can meet increasing market demand as the drug candidate progresses through clinical trials to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbamate synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers cover aspects ranging from catalyst selection to process safety, offering a comprehensive overview of the technology's capabilities.
Q: What is the primary advantage of using enzymatic hydrolysis in this carbamate synthesis?
A: Enzymatic hydrolysis offers superior stereoselectivity compared to chemical catalysts, ensuring high optical purity (>99% ee) while operating under mild, neutral pH conditions that minimize side reactions and simplify downstream purification.
Q: Which specific hydrolases are recommended for converting the acetoxy intermediate to the final diol monocarbamate?
A: The patent specifically highlights Porcine Liver Esterase (PLE) as the preferred catalyst, although Lipase from Porcine Pancreas (PPL), Candida Lipase (CCL), and Pseudomonas Lipase (PSL) are also viable options for this transformation.
Q: How does this process address the safety concerns associated with phosgene usage?
A: While phosgene is utilized in the initial step, the process employs controlled low-temperature conditions (-30 to 0°C) and immediate conversion to the stable carbamate using excess anhydrous ammonia, mitigating risks and ensuring a safe, scalable workflow for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbamate Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of CNS therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical supply to full-scale manufacturing. We are equipped with state-of-the-art facilities capable of handling sensitive reactions such as phosgenation under strictly controlled conditions, alongside dedicated biocatalysis suites for enzymatic transformations. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify the identity and optical purity of every batch. By partnering with us, you gain access to a supply chain that is both resilient and responsive to the dynamic needs of the pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this patented technology can be tailored to your specific project requirements. Whether you need a Customized Cost-Saving Analysis or detailed route feasibility assessments, our experts are ready to provide the data-driven insights necessary to move your program forward. We encourage potential partners to request specific COA data for our carbamate intermediates to verify their suitability for your synthesis plans. Together, we can accelerate the delivery of life-saving medications to patients while optimizing the economic and environmental performance of your supply chain.
