Advanced Manufacturing of 6-(Perfluoroalkyl)Uracil Compounds via Novel Carbamate Cyclization
Advanced Manufacturing of 6-(Perfluoroalkyl)Uracil Compounds via Novel Carbamate Cyclization
The chemical industry is constantly seeking more efficient and safer pathways for synthesizing complex heterocyclic structures, particularly those serving as critical intermediates in agrochemical applications. Patent CN1344258A introduces a significant technological breakthrough in the preparation of 6-(perfluoroalkyl)uracil compounds, which are valuable precursors for herbicidal agents. This innovation shifts the synthetic paradigm away from traditional, hazardous methodologies towards a more robust carbamate-based cyclization strategy. By leveraging specific carbamate compounds of Formula II, manufacturers can achieve high-purity uracil derivatives while circumventing the logistical and safety challenges associated with previous generations of synthesis. This report analyzes the technical merits and commercial implications of this improved process for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-(perfluoroalkyl)uracil compounds relied heavily on the formation of 2-(N,N-disubstituted)amino-4-(perfluoroalkyl)-1,3-oxazin-6-one intermediates. As documented in prior art such as Bull. Soc. Chem. Belg., this conventional pathway necessitates the use of phosgene iminium chloride as a key reagent. From an industrial safety and procurement perspective, phosgene iminium chloride presents substantial drawbacks; it is notoriously difficult to handle due to its reactivity and toxicity, requiring stringent containment protocols that drive up operational expenditures. Furthermore, the relative expense of this reagent compared to alternative starting materials creates a persistent cost burden in the manufacturing of herbicide intermediates. These factors collectively limit the scalability and economic viability of the traditional oxazinone route for large-volume production.
The Novel Approach
The methodology disclosed in CN1344258A offers a transformative alternative by utilizing carbamate compounds of Formula II as the primary building blocks. This novel approach completely avoids the use of the problematic 2-(N,N-disubstituted)amino-4-(perfluoroalkyl)-1,3-oxazin-6-one intermediates. Instead, the process involves the direct reaction of the Formula II carbamate with an amine compound of Formula III in the presence of a base. 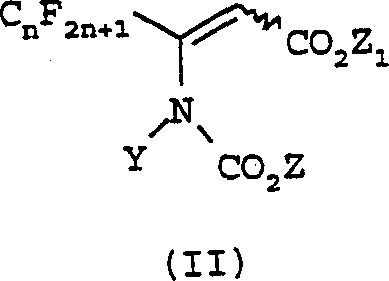 This structural shift allows for the use of more stable and commercially accessible reagents. The reaction proceeds efficiently under moderate thermal conditions, typically ranging from 20°C to 150°C, in common organic solvents. By replacing the hazardous phosgene derivative with a stable carbamate precursor, the new method significantly enhances the safety profile of the synthesis while simultaneously opening avenues for cost optimization through simpler raw material sourcing and handling procedures.
This structural shift allows for the use of more stable and commercially accessible reagents. The reaction proceeds efficiently under moderate thermal conditions, typically ranging from 20°C to 150°C, in common organic solvents. By replacing the hazardous phosgene derivative with a stable carbamate precursor, the new method significantly enhances the safety profile of the synthesis while simultaneously opening avenues for cost optimization through simpler raw material sourcing and handling procedures.
Mechanistic Insights into Base-Catalyzed Heterocyclic Cyclization
The core of this innovative synthesis lies in the base-catalyzed condensation and cyclization mechanism that transforms the linear carbamate precursor into the cyclic uracil structure. When the carbamate compound of Formula II is mixed with the amine compound of Formula III, the addition of a strong organic base, such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) or 1,5-diazabicyclo[4.3.0]non-5-ene (DBN), initiates a nucleophilic attack. The base deprotonates the amine or activates the carbamate carbonyl, facilitating the formation of a new carbon-nitrogen bond. This is followed by an intramolecular cyclization step that closes the pyrimidine ring, expelling the alcohol leaving group from the ester moiety. The choice of solvent plays a critical role in stabilizing the transition states; polar aprotic solvents like N,N-dimethylformamide (DMF) or aromatic hydrocarbons like xylene are preferred to ensure optimal solubility and reaction kinetics. The preference for the (Z)-configuration in the double bond of the Formula II starting material further enhances the stereochemical outcome, leading to higher yields of the desired isomer.
Impurity control is another vital aspect of this mechanistic pathway, particularly for pharmaceutical and agrochemical grade intermediates where strict purity specifications are mandatory. The reaction conditions described allow for precise control over side reactions that might lead to polymeric byproducts or incomplete cyclization. By maintaining the reaction temperature within the specified 20°C to 150°C range and utilizing stoichiometric amounts of the base, the formation of undesired urea derivatives or hydrolysis products is minimized. Post-reaction workup involves standard extraction techniques using solvents like ethyl acetate or dichloromethane, followed by washing with aqueous acid to remove residual amine and base. This straightforward purification protocol ensures that the final 6-(perfluoroalkyl)uracil product meets high purity standards without the need for complex chromatographic separations, which is essential for maintaining cost efficiency in commercial scale-up operations.
How to Synthesize 6-(Perfluoroalkyl)Uracil Efficiently
To implement this synthesis effectively, manufacturers must first secure high-quality carbamate precursors, which can be prepared by reacting beta-amino-beta-(perfluoroalkyl)acrylates with chloroformates. The subsequent cyclization step requires careful selection of the amine partner to introduce the desired substituent at the N-3 position of the uracil ring. The process is versatile, accommodating various amines including alkylamines and arylamines, allowing for the generation of a diverse library of herbicidal candidates. Detailed standard operating procedures regarding specific molar ratios, addition rates, and quenching protocols are essential for reproducibility. 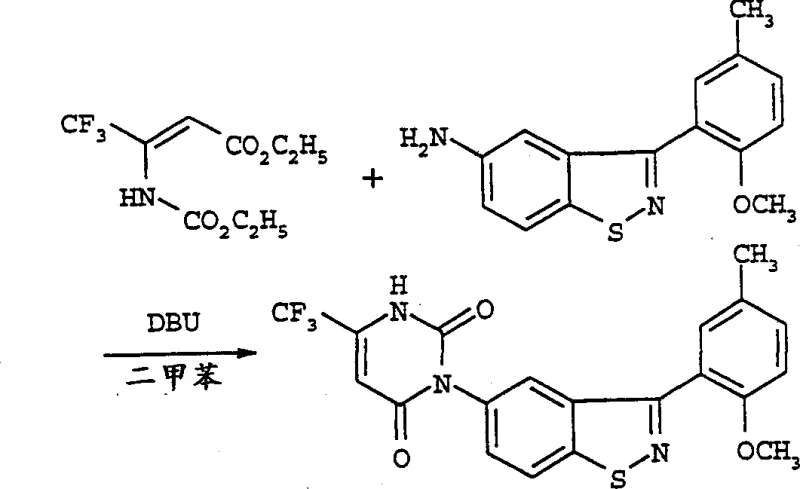 The following guide outlines the standardized synthesis steps derived from the patent examples to ensure consistent quality and yield.
The following guide outlines the standardized synthesis steps derived from the patent examples to ensure consistent quality and yield.
- Prepare the carbamate starting material (Formula II) by reacting a beta-amino-beta-(perfluoroalkyl)acrylate with a chloroformate in the presence of a base.
- React the Formula II carbamate compound with an amine compound (Formula III) in a suitable solvent such as xylene or DMF.
- Add a strong organic base like DBU or DBN and heat the mixture between 20°C to 150°C to facilitate cyclization into the final uracil structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN1344258A process represents a strategic opportunity to optimize the cost structure and reliability of agrochemical intermediate supply. The elimination of phosgene iminium chloride from the supply chain removes a significant bottleneck associated with hazardous material logistics, storage, and regulatory compliance. This shift not only reduces the direct cost of goods sold by substituting expensive reagents with more economical carbamates but also lowers the indirect costs related to safety training, specialized equipment maintenance, and waste disposal. Furthermore, the use of common solvents like xylene and toluene ensures that raw material availability remains high, mitigating the risk of supply disruptions that often plague specialty chemical markets. This stability is crucial for maintaining continuous production schedules in the face of fluctuating global demand for herbicides.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this manufacturing process is the substitution of the costly and hazardous phosgene iminium chloride with stable carbamate compounds. This change eliminates the need for expensive containment systems and specialized handling protocols required for toxic gases or their immediate precursors. Additionally, the reaction proceeds with high efficiency, minimizing raw material waste and maximizing the yield of the final uracil product. The ability to use standard laboratory and plant equipment without modification further contributes to capital expenditure savings, allowing facilities to repurpose existing infrastructure for this new chemistry. Overall, the process delivers substantial cost savings through both raw material optimization and operational simplification.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly enhanced by the reliance on commercially available and stable starting materials. Carbamate compounds and simple amines are produced in large volumes by multiple global suppliers, reducing dependency on single-source vendors for niche reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, adds another layer of resilience, ensuring that production can continue even if specific utility parameters fluctuate slightly. This flexibility allows supply chain managers to diversify their vendor base and negotiate better terms, securing a steady flow of high-purity intermediates necessary for downstream herbicide formulation. Consequently, the risk of production stoppages due to raw material shortages is drastically minimized.
- Scalability and Environmental Compliance: Scalability is a key strength of this process, as it avoids the engineering challenges associated with handling gaseous or highly reactive intermediates on a multi-ton scale. The reaction can be safely scaled from kilogram to metric ton quantities using standard batch reactors, facilitating a smooth transition from pilot plant to commercial production. From an environmental compliance perspective, the process generates less hazardous waste compared to the conventional oxazinone route. The absence of phosgene-derived byproducts simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only ensures compliance with increasingly stringent environmental regulations but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details is critical for evaluating the feasibility of integrating this technology into existing production lines.
Q: What is the primary advantage of the carbamate route over the conventional oxazinone method?
A: The carbamate route eliminates the need for phosgene iminium chloride, a reagent that is difficult to handle, hazardous, and relatively expensive, thereby improving safety and reducing raw material costs.
Q: What types of bases are suitable for this cyclization reaction?
A: The patent specifies several effective bases including heterocyclic tertiary amines like DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) and DBN, as well as trialkylamines and alkali metal alcoholates.
Q: Can this process be scaled for commercial herbicide production?
A: Yes, the process utilizes standard solvents like xylene and toluene and operates at moderate temperatures (20°C to 150°C), making it highly amenable to large-scale commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-(Perfluoroalkyl)Uracil Supplier
As the global demand for high-performance herbicides continues to grow, the need for reliable and efficient supply chains for critical intermediates like 6-(perfluoroalkyl)uracil has never been more pressing. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies such as the carbamate cyclization route to deliver superior quality products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major agrochemical companies. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for final herbicide registration and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this improved manufacturing process can benefit your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your current procurement model, highlighting potential efficiencies gained by switching to this safer, more economical route. We encourage you to request specific COA data and route feasibility assessments to validate the technical superiority of our offerings. Let us help you secure a sustainable and cost-effective supply of high-purity 6-(perfluoroalkyl)uracil compounds for your agrochemical portfolio.
