Advanced Synthetic Route for Pyrimidine Derivatives Enhancing Pharmaceutical Intermediate Manufacturing Safety
Introduction to Advanced Pyrimidine Synthesis Technology
The pharmaceutical industry continuously seeks robust and safe manufacturing processes for critical active pharmaceutical ingredient (API) intermediates. Patent CN1399632A introduces a significant technological breakthrough in the synthesis of novel pyrimidine derivatives, which serve as vital precursors for potent elastase inhibitors. These compounds are essential in treating conditions such as chronic obstructive pulmonary disease and rheumatoid arthritis. The core innovation lies in a streamlined synthetic pathway that bypasses traditional, hazardous rearrangement reactions, offering a more direct route to high-purity intermediates. This advancement addresses long-standing safety concerns associated with large-scale chemical manufacturing while maintaining rigorous quality standards required for drug substance production.
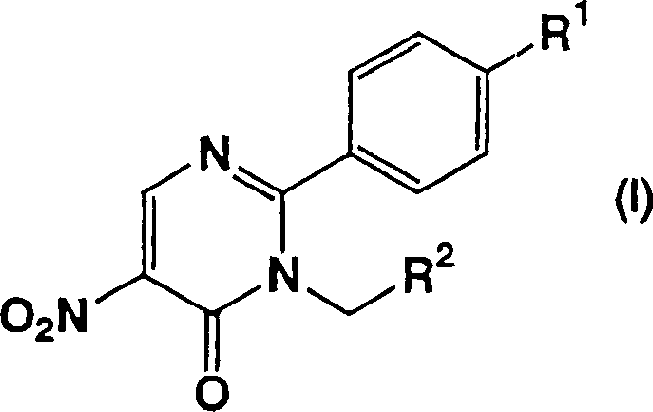
For procurement and supply chain leaders, understanding the underlying chemistry is crucial for assessing long-term vendor viability. The patented method described in CN1399632A utilizes a unique condensation reaction between N-substituted amidine derivatives and 3-methoxy-2-nitroacrylate esters. This approach not only simplifies the molecular construction but also enhances the controllability of the reaction profile. By establishing a reliable pharmaceutical intermediate supplier partnership based on such advanced methodologies, organizations can mitigate risks associated with complex multi-step syntheses. The ability to produce these scaffolds efficiently translates directly into improved cost structures and reduced lead times for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrimidine derivatives useful as serine protease inhibitors relied heavily on methodologies disclosed in earlier patents such as WO98/24806. These conventional routes typically commenced with amidine derivatives and diethyl ethoxymethylene malonate, necessitating a lengthy sequence of 8 to 9 distinct chemical transformations to reach the desired intermediate. A critical bottleneck in these legacy processes was the reliance on the Curtius rearrangement reaction to install specific functional groups. While effective on a laboratory scale, the Curtius rearrangement generates substantial volumes of nitrogen gas as a by-product. In an industrial setting, this rapid gas evolution presents severe safety hazards, including the risk of reactor over-pressurization and potential explosions, thereby complicating process safety management.
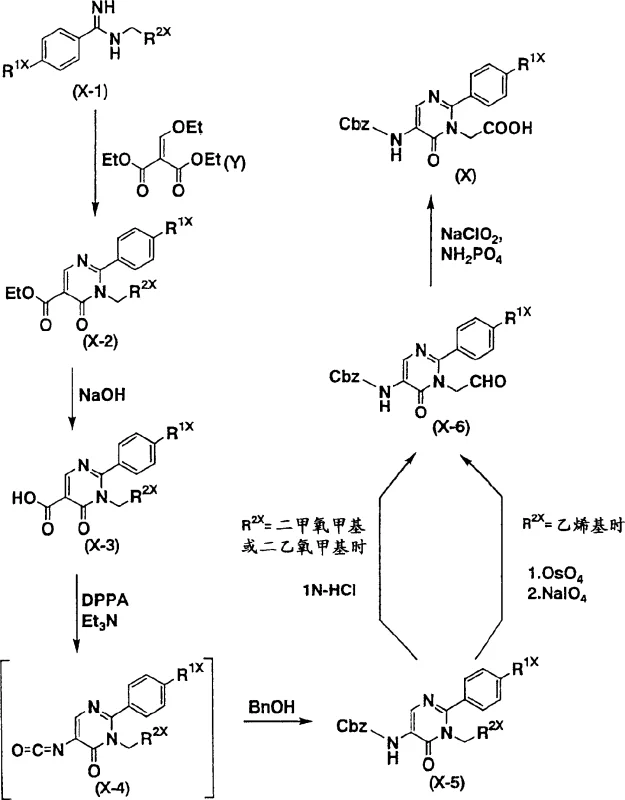
Furthermore, the extended step count inherent in the traditional approach accumulates yield losses at each stage, drastically reducing the overall material throughput. Each additional purification and isolation step increases solvent consumption, waste generation, and operational time, leading to inflated manufacturing costs. For a reliable agrochemical intermediate supplier or pharma partner, managing the environmental footprint of such inefficient processes is increasingly challenging under modern regulatory frameworks. The complexity of handling hazardous reagents like diphenylphosphoryl azide (DPPA) further restricts the number of qualified manufacturers capable of executing these syntheses safely, creating supply chain vulnerabilities for downstream drug developers.
The Novel Approach
The innovative methodology presented in the patent data fundamentally reengineers the synthetic strategy by eliminating the problematic rearrangement steps. Instead of the multi-step degradation and reconstruction of the molecular skeleton, the new process employs a direct condensation reaction. Starting from readily available N-substituted amidine derivatives and 3-methoxy-2-nitroacrylate derivatives, the target 5-nitro-6-oxo-1,6-dihydropyrimidine core is constructed in merely 3 to 5 steps. This drastic reduction in synthetic complexity allows for cost reduction in pharmaceutical intermediate manufacturing by minimizing unit operations and maximizing overall yield. The reaction conditions are mild and adaptable, utilizing common organic solvents and avoiding the generation of hazardous gases.
This novel route offers superior scalability, making it ideal for the commercial scale-up of complex pharmaceutical intermediates. By removing the safety barriers associated with nitrogen gas evolution, manufacturers can operate larger batch sizes with greater confidence and lower insurance overheads. The streamlined process also facilitates tighter control over impurity profiles, as fewer transformation steps mean fewer opportunities for side reactions to occur. Consequently, this approach supports the production of high-purity OLED material or pharmaceutical grades with consistent quality, ensuring that supply chain heads can rely on uninterrupted delivery schedules even during periods of high market demand.
Mechanistic Insights into Amidine-Nitroacrylate Condensation
The heart of this technological advancement is the previously unknown reaction between N-substituted amidine derivatives and 3-methoxy-2-nitroacrylate esters. Unlike reactions involving unsubstituted amidines which are well-documented, the presence of substituents on the nitrogen atom introduces steric and electronic challenges that this new method successfully overcomes. The mechanism involves a nucleophilic attack by the amidine nitrogen on the electron-deficient double bond of the nitroacrylate, followed by cyclization and elimination of methanol. This cascade occurs efficiently within a temperature range of -20°C to 150°C, depending on the specific solvent system employed, such as methanol, toluene, or dimethylformamide. The versatility of this mechanism allows for the incorporation of diverse functional groups at the N1 position of the pyrimidine ring.
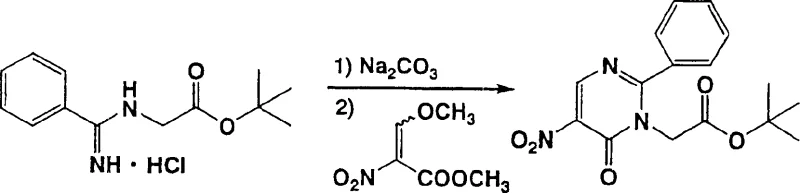
Impurity control is meticulously managed through the precise addition rate of the nitroacrylate reagent. The patent specifies that slowly dropping the 3-methoxy-2-nitroacrylate derivative into the amidine solution, rather than a bolus addition, significantly suppresses the formation of polymeric by-products and regioisomers. This kinetic control is vital for maintaining high purity specifications without resorting to excessive chromatographic purification, which is often cost-prohibitive at scale. Furthermore, the use of additives like calcium acetate or cesium fluoride can further modulate the reaction environment, enhancing selectivity. For R&D directors, understanding these mechanistic nuances is key to troubleshooting potential scale-up issues and ensuring that the final API meets stringent regulatory impurity limits.
How to Synthesize 5-Nitro-6-oxo-1,6-dihydropyrimidine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize the benefits of the novel condensation pathway. The process begins with the preparation of the N-substituted amidine, which can be derived from corresponding nitriles or imidates. Once the amidine is ready, it is dissolved in a suitable solvent under an inert atmosphere to prevent oxidation. The critical step involves the controlled addition of the nitroacrylate component, where temperature monitoring is essential to prevent exothermic runaway. Following the reaction, standard workup procedures such as acid-base extraction and recrystallization are employed to isolate the intermediate. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this efficient protocol.
- Prepare the N-substituted amidine derivative solution in an appropriate organic solvent such as methanol or toluene under inert atmosphere.
- Slowly add 3-methoxy-2-nitroacrylate derivative to the amidine solution while maintaining temperature between -20°C and 150°C to suppress by-product formation.
- Purify the resulting 5-nitro-6-oxo-1,6-dihydropyrimidine intermediate via crystallization or column chromatography for subsequent amidation or reduction steps.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers profound strategic advantages for procurement managers and supply chain executives focused on resilience and efficiency. The primary benefit stems from the significant simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By cutting the number of synthetic steps from nearly nine down to as few as three, the consumption of raw materials, solvents, and energy is drastically lowered. This efficiency gain allows for substantial cost savings in the final price of the intermediate, providing a competitive edge in pricing negotiations. Moreover, the elimination of hazardous reagents like DPPA removes the need for specialized containment equipment and expensive waste disposal protocols associated with azide chemistry.
- Cost Reduction in Manufacturing: The streamlined 3-to-5-step process inherently lowers the cost of goods sold by reducing labor hours and utility consumption per kilogram of product. Eliminating the Curtius rearrangement removes the safety costs associated with handling explosive nitrogen gas generation, allowing for simpler reactor designs and lower capital investment. Additionally, higher overall yields mean less starting material is required to produce the same amount of final product, optimizing raw material spend. These factors combine to create a leaner manufacturing model that is less susceptible to fluctuations in commodity prices.
- Enhanced Supply Chain Reliability: Shorter synthesis cycles translate directly into faster turnaround times, enabling suppliers to respond more agilely to market demands. The robustness of the condensation reaction ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. With fewer critical process parameters to monitor compared to the multi-step legacy route, the likelihood of human error is minimized. This reliability is crucial for maintaining continuous API production lines and avoiding costly stockouts that can impact downstream drug availability.
- Scalability and Environmental Compliance: The absence of gas-generating steps makes this process exceptionally scalable, allowing for seamless transition from pilot plant to multi-ton commercial production. The reduced solvent usage and waste generation align with green chemistry principles, helping manufacturers meet increasingly strict environmental regulations. Lower E-factor values contribute to a smaller carbon footprint, which is becoming a key criterion for supplier selection in the global pharmaceutical market. This environmental stewardship enhances the brand reputation of both the supplier and the end-user.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrimidine derivative synthesis technology. These answers are derived directly from the experimental data and claims within patent CN1399632A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply chains. The responses cover safety profiles, scalability potential, and compatibility with downstream processing requirements.
Q: Why is the new synthesis method safer than the conventional Curtius rearrangement route?
A: The conventional method involves a Curtius rearrangement which generates large volumes of nitrogen gas, posing explosion risks during industrial scale-up. The new method utilizes a direct condensation reaction that avoids gas generation entirely.
Q: What is the step reduction achieved in this novel pyrimidine derivative preparation?
A: The prior art methods required 8 to 9 steps to reach the target intermediate depending on the protecting group. The patented process reduces this to only 3 to 5 steps, significantly improving overall efficiency.
Q: Can this process be scaled for commercial production of elastase inhibitors?
A: Yes, the elimination of hazardous gas-generating steps and the use of standard condensation reagents make this route highly suitable for commercial scale-up, ensuring better supply chain reliability and safety compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such cutting-edge synthetic methodologies to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of pyrimidine derivative meets the highest international standards. Our facility is equipped to handle the specific solvent systems and temperature controls required for this novel condensation reaction safely and efficiently.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your elastase inhibitor development program. Together, we can build a more resilient and cost-effective supply chain for next-generation therapeutics.
