Advanced Synthesis of Organic Boron Difluoride Complexes for Next-Generation Optoelectronic Applications
The landscape of advanced optoelectronic materials is undergoing a significant transformation, driven by the demand for high-efficiency fluorescent compounds with superior thermal and chemical stability. A pivotal development in this sector is documented in Chinese patent CN102040617B, which discloses a novel class of organic boron difluoride complexes characterized by their exceptional fluorescence quantum yields and robust structural integrity. Unlike traditional boron complexes that often rely on N,N or O,O bidentate ligands, this innovation utilizes a unique N,O-bidentate coordination framework derived from 2-(2-quinolyl)phenol derivatives. This structural modification addresses critical limitations in prior art, specifically regarding solid-state fluorescence quenching and environmental stability. For R&D directors and procurement specialists in the electronic chemicals sector, this patent represents a viable pathway to sourcing high-performance luminescent materials that can be integrated into OLED displays, fluorescent probes, and laser dyes with greater reliability and cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organoboron fluorescent materials has been plagued by several intrinsic challenges that hinder their widespread commercial adoption. Traditional methods often employ N,N-bidentate ligands, such as pyrromethenes, which, while effective in solution, frequently suffer from aggregation-caused quenching (ACQ) in the solid state, drastically reducing their utility in solid-state devices. Furthermore, many conventional synthetic routes require harsh reaction conditions, including elevated temperatures, prolonged reaction times, and the use of moisture-sensitive reagents that demand rigorous anhydrous environments throughout the entire process. These factors not only increase the operational expenditure due to energy consumption but also complicate the supply chain by necessitating specialized storage and handling protocols. Additionally, the purification of these conventional complexes often involves labor-intensive column chromatography, which generates significant solvent waste and limits the scalability of the production process, making it difficult to achieve the consistent purity required for high-end electronic applications.
The Novel Approach
In stark contrast, the methodology outlined in patent CN102040617B introduces a streamlined and highly efficient synthetic strategy that overcomes these historical bottlenecks. By leveraging 2-(2-quinolyl)phenol derivatives as N,O-bidentate ligands, the new approach capitalizes on the rigidification of the molecular structure, which effectively suppresses non-radiative decay pathways and enhances fluorescence intensity in both solution and solid phases. The synthesis is remarkably mild, proceeding efficiently at room temperature (20°C-30°C) or with slight heating (55°C-65°C), thereby eliminating the need for energy-intensive reflux conditions. The reaction kinetics are rapid, typically completing within 15 to 50 minutes, and the product isolation is simplified to a straightforward filtration and washing step using anhydrous ether, bypassing the need for complex chromatographic separation. This drastic simplification of the workflow not only accelerates the time-to-market for new material batches but also aligns perfectly with green chemistry principles by reducing solvent usage and waste generation.
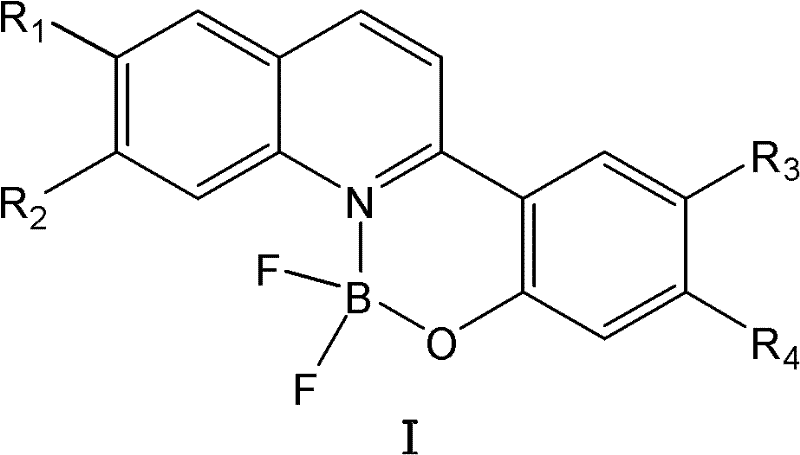
Mechanistic Insights into N,O-Bidentate Coordination Chemistry
The superior performance of these organic boron difluoride complexes can be attributed to the specific electronic and steric properties imparted by the N,O-bidentate coordination environment. In this system, the boron atom acts as a Lewis acid, coordinating simultaneously with the nitrogen atom of the quinoline ring and the oxygen atom of the phenolic group. This chelation creates a rigid, planar six-membered ring structure that restricts the intramolecular rotation of the ligand, a phenomenon known as Restriction of Intramolecular Motion (RIM). By locking the molecular conformation, the complex minimizes energy loss through vibrational relaxation, thereby channeling a higher proportion of absorbed energy into radiative fluorescence emission. Furthermore, the introduction of substituents on the phenolic or quinoline rings allows for fine-tuning of the electronic density, enabling precise modulation of the absorption and emission wavelengths across the visible spectrum. This tunability is crucial for applications requiring specific color coordinates, such as in full-color display technologies.
From an impurity control perspective, the mechanism ensures high product fidelity through the thermodynamic stability of the B-N and B-O bonds formed during the reaction. The use of an organic base, such as triethylamine or N,N-diisopropylethylamine, serves a dual purpose: it deprotonates the phenolic hydroxyl group to generate a more nucleophilic phenoxide anion, which facilitates faster coordination with the boron center, and it neutralizes the hydrogen fluoride byproduct generated during the complexation. This in situ neutralization prevents acid-catalyzed decomposition of the sensitive fluorophore backbone, ensuring that the final product maintains its structural integrity and optical properties. The result is a crude product of such high purity that simple washing suffices for isolation, significantly reducing the risk of trace metal or solvent contaminants that could otherwise act as quenchers in downstream electronic applications.
How to Synthesize Organic Boron Difluoride Complexes Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing fine chemical manufacturing facilities. The process begins with the dissolution of the specific 2-(2-quinolyl)phenol ligand in a common anhydrous organic solvent, followed by the controlled addition of the base and the boron source. The reaction progress is easily monitored, and the precipitation of the product often occurs spontaneously upon completion, facilitating immediate recovery. This operational simplicity makes it an ideal candidate for both laboratory-scale optimization and large-scale commercial production, ensuring that supply chain partners can reliably meet fluctuating demand without compromising on quality standards. For detailed procedural specifics regarding stoichiometry and workup, please refer to the standardized guide below.
- Dissolve the 2-(2-quinolyl)phenol ligand in an anhydrous organic solvent such as toluene or dichloromethane under stirring.
- Add an organic base like triethylamine to deprotonate the phenolic hydroxyl group, enhancing coordination ability.
- Introduce boron trifluoride diethyl etherate at room temperature or with mild heating, followed by filtration and washing to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere technical performance. The shift towards this milder, high-yield process directly addresses the industry's pressing need for cost reduction in electronic chemical manufacturing while simultaneously enhancing supply security. By eliminating the reliance on expensive transition metal catalysts and complex purification infrastructure, manufacturers can achieve substantial operational savings that can be passed down the value chain. Moreover, the robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for long-term supply agreements in the volatile electronics market.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its exceptional atom economy and minimal resource consumption. Since the reaction proceeds with high yields generally exceeding 70 percent and often reaching above 90 percent in optimized examples, the raw material utilization rate is maximized, leaving very little waste. The elimination of column chromatography, a notoriously solvent-intensive and time-consuming step, results in a drastic reduction in solvent procurement costs and waste disposal fees. Furthermore, the ability to run the reaction at or near room temperature significantly lowers energy expenditures associated with heating and cooling, contributing to a leaner overall cost structure that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for downstream manufacturers of displays and sensors, and this synthesis route fortifies the supply chain through its reliance on readily available commodity chemicals. Reagents such as boron trifluoride diethyl etherate, triethylamine, and common solvents like toluene or dichloromethane are widely produced and easily sourced from multiple suppliers, mitigating the risk of single-source bottlenecks. The short reaction time, ranging from merely 15 to 50 minutes, allows for rapid turnover of production batches, enabling manufacturers to respond swiftly to urgent orders or unexpected demand spikes. This agility ensures that lead times for high-purity organic boron difluoride complexes can be kept to a minimum, safeguarding the production schedules of our clients.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this method offers a clear advantage through its inherently greener profile. The simplified workup procedure, which involves filtration and washing rather than extensive solvent evaporation and chromatography, significantly reduces the volume of volatile organic compounds (VOCs) released into the atmosphere. The high thermal stability of the final products also implies safer storage and transportation conditions, reducing the logistical complexities associated with hazardous materials. Consequently, scaling this process from pilot plants to multi-ton commercial production is straightforward and compliant with increasingly stringent environmental regulations, future-proofing the supply chain against potential regulatory hurdles.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries based on the specific data points and embodiments found within the patent literature. These insights are intended to assist technical decision-makers in evaluating the feasibility of integrating these materials into their current product lines. Understanding the nuances of the substitution effects and reaction parameters is essential for optimizing the final application performance.
Q: What are the key advantages of the N,O-bidentate ligand system in this patent?
A: The N,O-bidentate ligand system provides a rigid structure that minimizes non-radiative decay, resulting in high fluorescence quantum yields and strong solid-state emission compared to traditional N,N or O,O systems.
Q: How does the new synthesis method improve production efficiency?
A: The method operates under mild conditions (20°C-65°C) with short reaction times (15-50 minutes) and eliminates the need for complex chromatographic purification, significantly reducing processing time and energy consumption.
Q: Can these complexes be scaled for industrial manufacturing?
A: Yes, the process uses common solvents and reagents with high yields (generally above 70%), making it highly suitable for commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Boron Difluoride Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in patent CN102040617B and are fully equipped to bring these advanced materials to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of major display and optoelectronic manufacturers. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of organic boron difluoride complex meets the exacting standards required for high-performance electronic applications, delivering consistent fluorescence quantum yields and thermal stability.
We invite you to collaborate with us to leverage this cutting-edge technology for your next-generation products. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our optimized synthesis route can lower your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the commercialization of your advanced fluorescent material projects.
