Advanced Synthesis of Organic Boron Difluoride Complexes for High-Performance Luminescent Applications
Advanced Synthesis of Organic Boron Difluoride Complexes for High-Performance Luminescent Applications
The rapid evolution of optoelectronic materials and biomedical imaging technologies has created an urgent demand for high-performance fluorophores with tunable emission properties and exceptional stability. Patent CN102993224A introduces a groundbreaking class of organic boron difluoride complexes characterized by a unique N,O-bidentate ligand system, specifically designed to overcome the limitations of traditional pyrromethene or beta-diketone based fluorophores. This technology represents a significant leap forward in the field of functional materials, offering a robust synthetic pathway to generate compounds that exhibit intense solution fluorescence, high quantum yields, and remarkable thermal stability. For R&D directors and procurement specialists in the fine chemical sector, understanding the nuances of this patented methodology is crucial for securing a reliable supply chain of next-generation luminescent intermediates. The core innovation lies in the structural versatility of the quinoxalinone-derived scaffold, which allows for precise modulation of optical properties through simple substituent variations.
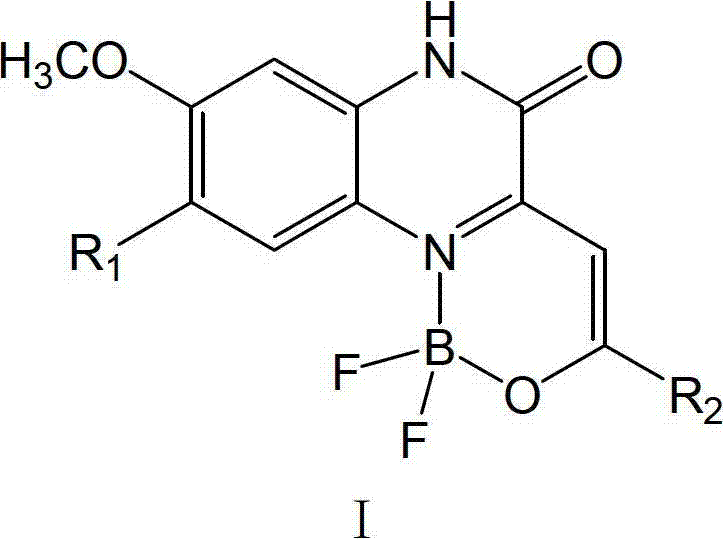
As illustrated in the general structure, the complex features a rigidified planar conformation enforced by the boron coordination, which minimizes non-radiative decay pathways and enhances fluorescence efficiency. The ability to modify the R1 and R2 groups provides a powerful toolkit for chemists to tailor the emission wavelength across the visible spectrum, ranging from yellow-green to orange-red, depending on the electron-donating or withdrawing nature of the substituents. This adaptability makes these compounds highly attractive candidates for diverse applications including laser dyes, information storage media, and photosensitizers for photodynamic cancer therapy. Furthermore, the patent highlights that these complexes maintain their fluorescent integrity in both solution and solid states, a critical attribute for the fabrication of OLED devices and solid-state sensors where aggregation-caused quenching is often a detrimental factor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic boron difluoride complexes has been dominated by N,N-bidentate ligands such as pyrromethenes (BODIPY dyes) and O,O-bidentate ligands like beta-diketonates. While these classes of compounds are well-studied, they suffer from inherent drawbacks that limit their utility in demanding industrial applications. For instance, many traditional BODIPY derivatives require multi-step syntheses involving harsh conditions and expensive reagents, leading to elevated production costs and complex purification protocols. Additionally, the chemical stability of some conventional complexes can be compromised under physiological conditions or upon prolonged exposure to light, resulting in photobleaching that renders them ineffective for long-term bio-imaging or sensing tasks. The reliance on specific substitution patterns to achieve desired optical properties often restricts the chemical space available for optimization, forcing researchers to compromise between stability and brightness. Moreover, the separation of isomers and byproducts in conventional routes frequently necessitates column chromatography, a technique that is notoriously difficult to scale up for commercial manufacturing due to solvent consumption and throughput limitations.
The Novel Approach
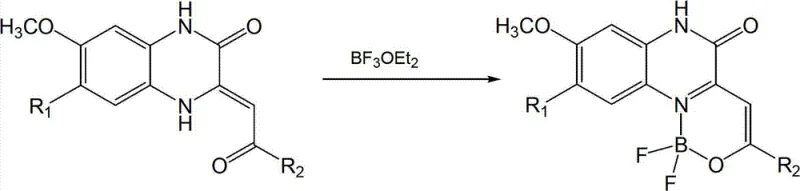
In stark contrast, the methodology disclosed in CN102993224A leverages a beta-ketimine structural motif within a quinoxalinone framework to create a new family of N,O-bidentate boron complexes. This novel approach fundamentally simplifies the synthetic landscape by utilizing readily available starting materials such as methoxy-substituted o-phenylenediamines and substituted aroyl acetoacetates. The reaction conditions are exceptionally mild, typically proceeding at room temperature or under gentle reflux, which significantly reduces energy consumption and minimizes the risk of thermal degradation of sensitive functional groups. The patent data indicates that the complexation with boron trifluoride diethyl ether occurs rapidly, often completing within 30 to 50 minutes, thereby drastically increasing throughput compared to legacy methods that may require hours of reaction time. Perhaps most importantly for supply chain managers, the workup procedure is remarkably straightforward; the product often precipitates directly from the reaction mixture or can be isolated via simple filtration and washing with anhydrous ether, eliminating the need for resource-intensive chromatographic purification. This streamlining of the process not only lowers the cost of goods sold but also enhances the environmental profile of the manufacturing process by reducing solvent waste.
Mechanistic Insights into BF3-Mediated Cyclization and Coordination
The formation of the target organic boron difluoride complex involves a sophisticated yet efficient coordination mechanism driven by the Lewis acidity of boron trifluoride. The precursor ligand, possessing a beta-ketimine structure, acts as a bidentate chelator where the nitrogen atom of the imine and the oxygen atom of the carbonyl group simultaneously coordinate to the boron center. This N,O-coordination mode creates a stable six-membered ring system that locks the molecular conformation, thereby restricting intramolecular rotation and vibration which are primary causes of fluorescence quenching. The introduction of the difluoroboron moiety further enhances the electron-accepting capability of the core, facilitating intramolecular charge transfer (ICT) processes that are responsible for the observed solvatochromism and large Stokes shifts. The use of an acid-binding agent like triethylamine in basic conditions, or the use of acetic acid in acidic conditions, plays a pivotal role in scavenging the hydrogen fluoride generated during the complexation, driving the equilibrium towards the product side. This mechanistic understanding allows process chemists to fine-tune reaction parameters such as stoichiometry and solvent polarity to maximize yield and minimize impurity formation.
From an impurity control perspective, the robustness of this coordination chemistry ensures a clean reaction profile. The strong affinity between the boron atom and the N,O-ligand prevents ligand exchange or hydrolysis under standard handling conditions, resulting in a product with high chemical purity. The patent specifies that yields are consistently above 80%, suggesting that side reactions such as polymerization or decomposition are effectively suppressed. For R&D teams focused on regulatory compliance, the absence of heavy metal catalysts in this synthesis is a major advantage, as it eliminates the need for rigorous metal scavenging steps and extensive testing for residual metal contaminants, which are strict requirements for pharmaceutical and biomedical applications. The structural rigidity imparted by the boron bridge also contributes to the thermal stability of the final complex, allowing it to withstand processing temperatures required for device fabrication without losing its optical properties.
How to Synthesize Organic Boron Difluoride Complexes Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, making it highly suitable for both laboratory research and industrial production. The process begins with the preparation of the specific ligand precursor, which can be synthesized via a condensation reaction catalyzed by sulfamic acid, followed by the critical complexation step with boron trifluoride diethyl ether. The detailed operational parameters, including precise molar ratios, solvent choices, and temperature controls, are essential for reproducing the high yields and purity reported in the patent literature. Operators must ensure that anhydrous conditions are maintained throughout the reaction to prevent hydrolysis of the boron reagent, and the choice between basic or acidic workup conditions should be dictated by the solubility profile of the specific derivative being produced. For a comprehensive guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized protocol below.
- Prepare the ligand precursor (Structure II) via condensation of methoxy-substituted o-phenylenediamine with substituted aroyl acetoacetate.
- React the ligand with excess boron trifluoride diethyl ether (BF3·OEt2) in an anhydrous organic solvent such as dichloromethane or toluene/acetic acid.
- Maintain reaction temperature between 15°C-35°C or under reflux for 30-50 minutes, followed by filtration and washing with anhydrous ether to isolate the high-purity complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical performance. The primary value proposition lies in the substantial cost reduction in fluorescent material manufacturing achieved through process intensification and simplification. By eliminating the need for expensive transition metal catalysts and complex purification techniques like column chromatography, the overall cost of production is significantly lowered, allowing for more competitive pricing in the global market. The use of commodity chemicals such as dichloromethane, toluene, and triethylamine ensures that raw material sourcing is straightforward and resilient against supply shocks, unlike specialized reagents that may have limited suppliers. Furthermore, the short reaction times and mild conditions translate to higher equipment utilization rates, enabling manufacturers to produce larger batches in less time without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of costly purification steps and the use of inexpensive, commercially available starting materials drive down the variable costs associated with production. The high yield of the reaction means less raw material is wasted, directly improving the material balance and reducing the cost per kilogram of the final active ingredient. Additionally, the energy savings from running reactions at lower temperatures or for shorter durations contribute to a leaner operational expenditure model.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with downstream users in the electronics and pharma sectors. The simplicity of the isolation process reduces the risk of production delays caused by equipment bottlenecks or purification failures. Sourcing the key intermediates is facilitated by their widespread availability in the fine chemical market, mitigating the risk of single-source dependency and ensuring business continuity.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production without the need for fundamental process redesign. The reduced solvent usage and absence of heavy metals simplify waste treatment protocols, helping companies meet increasingly stringent environmental regulations and sustainability goals. This eco-friendly profile enhances the brand reputation of suppliers and aligns with the green chemistry initiatives of major multinational corporations.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and commercial viability of these organic boron difluoride complexes, we have compiled a list of questions based on the patent data and industry standards. These answers provide clarity on the stability, application scope, and customization options available for clients looking to integrate these materials into their product lines. Understanding these details is essential for making informed purchasing decisions and optimizing the integration of these advanced fluorophores into existing workflows.
Q: What are the primary advantages of the N,O-bidentate boron difluoride complexes described in CN102993224A?
A: These complexes exhibit superior chemical and thermal stability compared to traditional N,N or O,O types, with high fluorescence quantum yields (>0.22) and large Stokes shifts up to 68nm, making them ideal for bio-imaging and laser dyes.
Q: How does the synthesis process ensure high purity for electronic applications?
A: The process utilizes mild reaction conditions (room temperature to reflux) and a simple workup involving filtration and ether washing, which effectively removes unreacted starting materials and byproducts without requiring complex chromatography.
Q: Is this synthesis route scalable for industrial production of fluorescent probes?
A: Yes, the method uses common solvents like dichloromethane and toluene, avoids expensive transition metal catalysts, and achieves yields generally above 80%, indicating excellent potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Boron Difluoride Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN102993224A for the next generation of optoelectronic and biomedical devices. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust, commercial-scale manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles. Whether you require custom derivatives for specific wavelength emissions or bulk quantities for industrial applications, our flexible manufacturing capabilities allow us to tailor our services to your unique requirements.
We invite you to collaborate with us to unlock the full commercial potential of these high-performance fluorescent materials. By leveraging our process optimization expertise, we can help you achieve a Customized Cost-Saving Analysis that identifies further efficiencies in your supply chain. We encourage you to contact our technical procurement team today to request specific COA data for our available grades and to discuss route feasibility assessments for your specific project needs. Let us be your partner in bringing innovative lighting and sensing solutions to the global market.
