Optimizing Pantoprazole Intermediate Production: A Deep Dive into Green Chlorination Technology
The pharmaceutical industry continuously seeks robust synthetic routes for Proton Pump Inhibitors (PPIs), with Pantoprazole remaining a cornerstone therapy for gastric disorders. Central to this value chain is the critical intermediate 4-chloro-3-methoxy-2-methyl-4-pyridine, whose efficient production dictates the cost and availability of the final Active Pharmaceutical Ingredient (API). A pivotal advancement in this domain is detailed in patent CN103483248A, which introduces a novel chlorination protocol that fundamentally alters the waste profile of traditional manufacturing. Unlike conventional approaches that struggle with the disposal of hazardous reagents, this method leverages a strategic DMF-mediated scavenging technique to transform waste into value. By integrating this technology, manufacturers can achieve superior yield consistency while adhering to increasingly stringent environmental regulations regarding acidic effluent. This report analyzes the technical merits of this synthesis, offering R&D and procurement leaders a clear pathway toward sustainable cost optimization in pharmaceutical intermediates manufacturing.
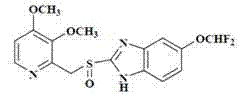
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chlorinated pyridine derivatives has relied heavily on the use of phosphorus oxychloride (POCl3) as both a solvent and a chlorinating agent. While chemically effective, the traditional workup procedures present severe logistical and environmental challenges for large-scale operations. In standard protocols, the reaction mixture containing a vast excess of POCl3 is typically quenched directly with ice water or neutralized with alkaline solutions. This indiscriminate hydrolysis generates substantial quantities of phosphoric acid and hydrochloric acid wastewater, creating a heavy burden on effluent treatment plants. Furthermore, the exothermic nature of quenching POCl3 poses significant safety risks, requiring specialized cooling infrastructure to prevent thermal runaways. The resulting acidic sludge not only increases disposal costs but also complicates the isolation of the organic product, often leading to lower recovery rates due to emulsification or product degradation in harsh acidic conditions. These factors collectively inflate the operational expenditure (OPEX) and extend the production cycle time, rendering older methods economically unsustainable in modern green chemistry contexts.
The Novel Approach
The methodology outlined in patent CN103483248A offers a transformative solution by reimagining the post-reaction treatment phase. Instead of destructive quenching, the process introduces dimethylformamide (DMF) to the reaction mixture after the primary chlorination is complete. This strategic addition reacts with the residual phosphorus oxychloride to form the Vilsmeier reagent, a stable and valuable complex that can be separated via phase differentiation. This innovation achieves two critical objectives simultaneously: it eliminates the generation of immediate acidic wastewater and recovers a useful chemical byproduct that can be utilized in other Vilsmeier-Haack formylation reactions. The upper organic layer, now free of excessive corrosive reagents, undergoes a mild hydrolysis in ice water under controlled pH conditions. This gentle workup preserves the integrity of the sensitive pyridine ring, resulting in significantly higher purity and mass yield compared to brute-force neutralization methods. For a reliable pharmaceutical intermediates supplier, adopting this route means transitioning from a waste-intensive operation to a circular, value-retaining process.
Mechanistic Insights into POCl3-Mediated Chlorination and DMF Scavenging
The core chemical transformation involves the electrophilic substitution of the hydroxyl group on the 3-methoxy-2-methyl-4-pyrone ring with a chlorine atom. In the presence of excess phosphorus oxychloride, the oxygen atom of the pyrone acts as a nucleophile, attacking the phosphorus center to form a reactive phosphate ester intermediate. This activation facilitates the subsequent displacement by a chloride ion, effectively aromatizing the ring to form the pyridine structure. The reaction kinetics are highly temperature-dependent, requiring a sustained thermal input between 60°C and 100°C to drive the conversion to completion without degrading the methoxy substituents. The novelty of the patented mechanism lies not in the chlorination itself, but in the fate of the unreacted POCl3. Upon cooling the mixture to 0-40°C, the added DMF acts as a Lewis base, coordinating with the remaining POCl3. This interaction prevents the POCl3 from reacting violently with water during the subsequent workup. Instead of forming corrosive acids, the POCl3-DMF complex precipitates or separates as a distinct denser phase, allowing for physical removal via decantation or separation funnels. This mechanistic diversion is crucial for maintaining the stability of the final product during isolation.
Impurity control is inherently built into this phase-separation strategy. In traditional aqueous quenches, the rapid generation of heat and gas can promote side reactions, such as the hydrolysis of the methoxy groups or the formation of polymeric tars. By removing the bulk of the reactive phosphorus species before introducing water, the new method creates a chemically quieter environment for hydrolysis. The pH adjustment to the 8-12 range using alkali is performed under mild thermal conditions (20-50°C), ensuring that any remaining acidic residues are neutralized without exposing the product to extreme pH swings that could trigger decomposition. This precise control over the chemical environment minimizes the formation of des-methoxy impurities and chlorinated byproducts, which are notoriously difficult to remove via crystallization. Consequently, the crude product obtained from this process exhibits a cleaner impurity profile, reducing the load on downstream purification steps like distillation or recrystallization and ensuring consistent quality for API synthesis.
How to Synthesize 4-Chloro-3-Methoxy-2-Methyl-4-Pyridine Efficiently
Implementing this synthesis requires strict adherence to the three-stage thermal profile defined in the patent to ensure safety and reproducibility. The process begins with the charging of 3-methoxy-2-methyl-4-pyrone into a reactor containing phosphorus oxychloride, followed by a controlled ramp to the addition temperature. Once the chlorination is verified via process control, the critical DMF treatment phase must be executed at lowered temperatures to stabilize the Vilsmeier complex. Finally, the hydrolysis stage demands careful monitoring of the exotherm to maintain product integrity. The detailed standardized operating procedures, including specific molar ratios and agitation speeds required for optimal phase separation, are essential for tech transfer teams to replicate the high yields reported in the literature.
- Addition Stage: React 3-methoxy-2-methyl-4-pyrone with excess phosphorus oxychloride at 60-100°C for 4-10 hours to form the chlorinated intermediate.
- Scavenging Stage: Cool the mixture to 0-40°C and add DMF to convert excess phosphorus oxychloride into separable Vilsmeier reagent.
- Hydrolysis Stage: Separate layers, then hydrolyze the upper layer in ice water at -5 to 30°C, adjusting pH to 8-12 to isolate the final pyridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates directly into tangible operational efficiencies and risk mitigation. The primary economic driver is the drastic reduction in waste treatment liabilities. By converting a hazardous waste stream (excess POCl3) into a separable byproduct, the facility avoids the high costs associated with neutralizing and disposing of large volumes of acidic wastewater. This aligns perfectly with global trends towards cost reduction in pharmaceutical intermediates manufacturing through green chemistry principles. Furthermore, the simplified workup reduces the consumption of auxiliary materials such as ice, alkali, and extraction solvents, which are often significant line items in the bill of materials. The ability to recover the Vilsmeier reagent also opens potential revenue streams or internal cost offsets if the facility operates other formylation processes, turning a cost center into a resource recovery node.
- Cost Reduction in Manufacturing: The elimination of aggressive aqueous quenching significantly lowers the demand for corrosion-resistant equipment and reduces maintenance downtime associated with acid handling. By avoiding the formation of heavy acid sludge, the plant reduces its dependency on external waste disposal vendors, whose fees have been rising steadily due to stricter environmental enforcement. Additionally, the higher mass yield reported in the patent examples implies that less starting material is required per kilogram of finished good, directly improving the material cost basis. This efficiency gain is compounded by the reduced energy load, as the process avoids the intense cooling requirements needed to manage the violent exotherm of traditional POCl3 hydrolysis, leading to substantial utility savings over long production campaigns.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often bottlenecked by environmental shutdowns at supplier facilities. This greener process reduces the regulatory risk profile of the manufacturing site, ensuring greater continuity of supply. The starting material, 3-methoxy-2-methyl-4-pyrone, is derived from maltol, a commodity chemical with a stable global supply chain, reducing the risk of raw material scarcity. Moreover, the robustness of the DMF scavenging step makes the process more forgiving to minor variations in reagent quality, ensuring that batch-to-batch consistency is maintained even when supply chains for bulk chemicals fluctuate. This stability is critical for long-term API contracts where supply interruption can trigger costly penalties.
- Scalability and Environmental Compliance: The phase separation technique employed in this method is inherently scalable, functioning effectively from pilot reactors to multi-ton commercial vessels without the need for complex new hardware. The reduction in acidic effluent simplifies the permitting process for capacity expansion, as the facility's environmental footprint is markedly smaller. This compliance advantage future-proofs the supply chain against tightening discharge regulations, particularly in key manufacturing hubs. The process also minimizes the generation of volatile organic compounds (VOCs) associated with extensive solvent washing in traditional workups, further aligning with corporate sustainability goals and reducing the carbon intensity of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chlorination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for feasibility assessments. Understanding these nuances is vital for engineering teams evaluating the retrofitting of existing production lines or designing new facilities for PPI intermediate synthesis. The answers highlight the balance between chemical efficiency and operational practicality that defines this modern approach.
Q: How does the DMF treatment improve the environmental profile of this synthesis?
A: Traditional methods quench excess phosphorus oxychloride with water, generating large volumes of corrosive acidic wastewater. The patented DMF treatment converts this excess reagent into Vilsmeier reagent, a valuable byproduct, thereby drastically reducing acidic effluent and waste treatment costs.
Q: What are the critical temperature controls for maximizing yield in this process?
A: Precise thermal management is essential. The addition reaction requires 60-100°C, while the DMF scavenging step must occur between 0-40°C. Crucially, the final hydrolysis is optimized at low temperatures (-5 to 5°C) to prevent side reactions and ensure high purity.
Q: Is this intermediate scalable for commercial API production?
A: Yes, the process utilizes standard industrial reagents like phosphorus oxychloride and DMF. The phase separation technique simplifies downstream processing, making it highly suitable for commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-3-Methoxy-2-Methyl-4-Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory requirement but a strategic imperative for our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the POCl3-DMF scavenging protocol are seamlessly translated from the laboratory to the manufacturing floor. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of 4-chloro-3-methoxy-2-methyl-4-pyridine meets the exacting standards required for Pantoprazole API synthesis. Our commitment to process excellence ensures that we deliver not just a chemical, but a reliable supply solution that supports your downstream manufacturing efficiency.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized production capabilities, we can help you reduce total landed costs while securing a sustainable supply chain. Please contact our technical procurement team to request specific COA data and route feasibility assessments. We are ready to demonstrate how our advanced manufacturing technologies can become a competitive advantage for your pharmaceutical portfolio, ensuring continuity and quality in an evolving market landscape.
