Scaling High-Purity 4-Hydroxyisoleucine Production with Novel BsIDO Biocatalysts
Scaling High-Purity 4-Hydroxyisoleucine Production with Novel BsIDO Biocatalysts
The pharmaceutical industry's demand for potent anti-diabetic agents has intensified the search for efficient synthetic routes to key intermediates like 4-hydroxyisoleucine. A groundbreaking development in this sector is detailed in Chinese Patent CN109504645B, which discloses a highly active isoleucine dioxygenase derived from Bacillus subtilis and its engineered mutants. This technology represents a paradigm shift from low-yield natural extraction to high-efficiency biocatalysis, addressing critical bottlenecks in the supply chain for type II diabetes therapeutics. By leveraging specific genetic modifications, the disclosed method achieves unprecedented substrate tolerance and conversion rates, positioning it as a viable solution for large-scale commercial manufacturing. For R&D directors and procurement specialists, understanding the mechanistic advantages and operational parameters of this pathway is essential for securing a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of 4-hydroxyisoleucine has been plagued by significant inefficiencies inherent to traditional extraction and chemical synthesis methods. The conventional approach involves extracting the compound directly from Fenugreek seeds, a process that is not only labor-intensive but also suffers from abysmal product yields of less than 1 percent. This scarcity drives up costs and creates volatility in the supply of this critical bioactive amino acid. Alternatively, purely chemical synthetic routes, such as the eight-step synthesis from ethyl 2-methylacetoacetate, involve hazardous reagents and complex protection-deprotection strategies, resulting in overall yields as low as 39 percent. Furthermore, earlier enzymatic attempts using wild-type enzymes were limited by low substrate concentrations, typically capped at 15 mM, which necessitates massive reactor volumes to produce meaningful quantities, thereby inflating capital expenditure and operational costs significantly.
The Novel Approach
The innovative strategy outlined in the patent data overcomes these historical barriers through the application of protein engineering to enhance enzyme performance. By isolating a novel isoleucine dioxygenase (BsIDO) from Bacillus subtilis and introducing specific point mutations, the inventors have created a biocatalyst capable of operating at substrate concentrations as high as 30 g/L (approximately 228 mM). This represents a more than tenfold increase in substrate loading compared to prior art, directly translating to reduced water usage and smaller reactor footprints. The process operates under mild aqueous conditions at a neutral pH of 6.0 and moderate temperatures around 40°C, eliminating the need for extreme pressures or toxic organic solvents. This robustness allows for a streamlined workflow where high conversion rates exceeding 95 percent are achieved within 12 to 24 hours, making the process economically competitive and environmentally sustainable for modern green chemistry standards.
Mechanistic Insights into Isoleucine Dioxygenase-Catalyzed Hydroxylation
The core of this technological advancement lies in the precise molecular engineering of the BsIDO active site to optimize the hydroxylation of the gamma-carbon of L-isoleucine. The reaction mechanism relies on the enzyme's ability to activate molecular oxygen using alpha-ketoglutarate as a co-substrate and ferrous iron (Fe2+) as a cofactor. In the catalytic cycle, the enzyme facilitates the oxidative decarboxylation of alpha-ketoglutarate to succinate and carbon dioxide, generating a highly reactive ferryl-oxo species that inserts an oxygen atom into the isoleucine backbone. The patent highlights specific mutations, such as L58A and N165H, which likely alter the steric environment of the substrate binding pocket. These modifications reduce steric hindrance, allowing for tighter binding of the bulky isoleucine molecule and facilitating faster turnover rates. The double mutant BsIDO L58A/N165H, in particular, demonstrates superior kinetic properties, suggesting a synergistic effect where the structural flexibility of the active site is maximized for efficient catalysis.
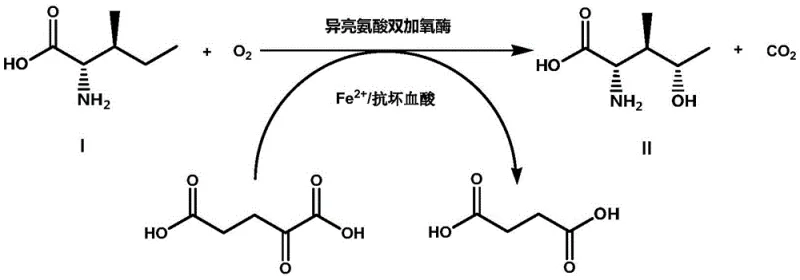
Controlling impurity profiles is another critical aspect of this mechanistic design, particularly for pharmaceutical applications where regulatory compliance is stringent. The high regioselectivity of the engineered BsIDO ensures that hydroxylation occurs specifically at the C-4 position, minimizing the formation of unwanted isomers that are difficult to separate. Traditional chemical methods often produce racemic mixtures or regio-isomers that require costly chiral resolution steps. In contrast, the biocatalytic route leverages the inherent chirality of the enzyme and the L-isoleucine starting material to produce the desired stereoisomer with high optical purity. The use of whole-cell biocatalysts further simplifies the system, as the intracellular environment helps stabilize the enzyme and regenerate necessary cofactors, reducing the risk of side reactions caused by free metal ions or unstable intermediates in the bulk solution.
How to Synthesize 4-Hydroxyisoleucine Efficiently
Implementing this synthesis route requires a systematic approach to fermentation and biotransformation to maximize the potential of the engineered enzymes. The process begins with the cultivation of recombinant E. coli strains harboring the optimized BsIDO genes, followed by the preparation of resting cells which serve as the biocatalyst. The reaction setup involves a carefully balanced aqueous system containing the substrate, co-substrates, and essential metal cofactors. Maintaining precise control over pH and temperature is vital to sustain enzyme stability throughout the transformation period. The following guide outlines the standardized operational steps derived from the patent examples to ensure reproducible high-yield production suitable for pilot and commercial scales.
- Prepare the reaction system by suspending recombinant E. coli cells expressing BsIDO mutants in a buffered solution containing L-isoleucine, alpha-ketoglutarate, ferrous sulfate, and ascorbic acid.
- Maintain the reaction at an optimal pH of 6.0 and a temperature of 40°C with continuous aeration to ensure sufficient oxygen supply for the dioxygenase activity.
- Monitor conversion via HPLC until completion, then proceed to downstream processing involving acidification, flocculation, and cation exchange resin purification to isolate the target amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic platform offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures and improved margin structures. By eliminating the need for multi-step chemical synthesis and the associated solvent recovery systems, manufacturers can significantly lower their utility consumption and waste disposal costs. The ability to run reactions at high substrate concentrations means that existing fermentation infrastructure can produce significantly higher output without requiring proportional increases in capital investment, effectively debottlenecking production capacity. This efficiency gain is crucial for meeting the growing global demand for anti-diabetic medications without incurring prohibitive costs.
- Cost Reduction in Manufacturing: The economic model of this biocatalytic process is fundamentally superior due to the elimination of expensive protecting groups and harsh chemical reagents required in traditional synthesis. The use of whole-cell biocatalysts removes the need for costly enzyme purification steps, as the cells can be used directly after fermentation. Furthermore, the high conversion rates minimize the amount of unreacted starting material that needs to be recovered and recycled, streamlining the material flow. The reliance on readily available bulk chemicals like L-isoleucine and alpha-ketoglutarate ensures stable raw material pricing, shielding the supply chain from the volatility often associated with specialty chemical precursors.
- Enhanced Supply Chain Reliability: Dependence on agricultural sources like Fenugreek seeds introduces significant risks related to crop failures, seasonal variations, and geographical concentration. By shifting to a fermentation-based production model, supply chains become decoupled from agricultural constraints, ensuring consistent year-round availability. The robustness of the engineered strains allows for reliable scale-up from laboratory benchtops to multi-ton industrial fermenters, guaranteeing that supply commitments can be met even during periods of surging demand. This reliability is paramount for pharmaceutical companies that require uninterrupted raw material flows to maintain their own production schedules and regulatory filings.
- Scalability and Environmental Compliance: The process aligns perfectly with modern environmental, social, and governance (ESG) goals by utilizing an aqueous system that generates minimal hazardous waste. The downstream processing involves standard flocculation and ion exchange techniques that are easily scalable and do not require complex distillation or extraction units. This simplicity reduces the environmental footprint of the manufacturing site and simplifies the permitting process for new facilities. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the final active pharmaceutical ingredient, which is increasingly valued by end-users and regulators alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel biocatalytic technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the proven capabilities of the BsIDO mutants under optimized conditions.
Q: What are the advantages of this enzymatic method over traditional extraction from Fenugreek seeds?
A: Traditional extraction yields less than 1% product and involves complex separation steps. The patented enzymatic route achieves conversion rates exceeding 95% at high substrate concentrations (30 g/L), drastically improving efficiency and reducing raw material dependency.
Q: How does the BsIDO mutant improve industrial feasibility compared to wild-type enzymes?
A: The engineered mutants, such as BsIDO L58A/N165H, demonstrate significantly enhanced catalytic efficiency and substrate tolerance. They allow for shorter reaction times (12-16 hours vs 24 hours) and maintain high activity even at elevated substrate loads, which is critical for cost-effective manufacturing.
Q: Is the downstream purification process scalable for commercial production?
A: Yes, the process utilizes standard unit operations including flocculation and strong acid cation exchange resin chromatography. These methods are well-established in the industry for amino acid purification, ensuring that the transition from laboratory to multi-ton scale is seamless and robust.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxyisoleucine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the BsIDO-catalyzed synthesis route for producing high-quality 4-hydroxyisoleucine. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this critical intermediate. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical applications. We are committed to leveraging advanced biocatalytic technologies to deliver cost-effective solutions that accelerate your drug development timelines while maintaining the highest standards of quality and compliance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this enzymatic route. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with robust, scalable, and commercially viable manufacturing solutions for next-generation anti-diabetic therapeutics.
