Revolutionizing 4-Hydroxyisoleucine Production: A Deep Dive into Multi-Enzyme Cascade Technology
The pharmaceutical industry's relentless pursuit of effective treatments for Type II diabetes has placed 4-hydroxyisoleucine (4-HIL) in the spotlight as a potent insulin secretagogue. Traditionally sourced from fenugreek seeds or synthesized via cumbersome chemical routes, the supply of high-purity 4-HIL has faced significant bottlenecks regarding cost and scalability. Patent CN113061630A introduces a groundbreaking biocatalytic solution that leverages a sophisticated multi-enzyme cascade system to overcome these historical limitations. By ingeniously coupling L-isoleucine dioxygenase with an in-situ alpha-ketoglutarate regeneration module driven by L-glutamate oxidase, this technology transforms the economic landscape of 4-HIL production. The core innovation lies in replacing expensive stoichiometric co-substrates with low-cost commodity chemicals, specifically sodium glutamate monohydrate, thereby establishing a highly efficient and economically viable pathway for large-scale manufacturing.
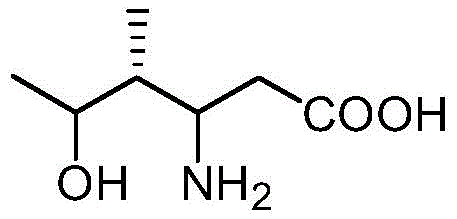
This technological leap is not merely an incremental improvement but a fundamental restructuring of the synthesis logic, addressing the critical pain points of substrate inhibition and co-substrate depletion that have plagued previous enzymatic attempts. The patent details a robust protocol where the oxidative decarboxylation of alpha-ketoglutarate is seamlessly integrated with the hydroxylation of L-isoleucine, ensuring that the reaction kinetics remain favorable throughout the process. For R&D directors and procurement specialists alike, this represents a shift towards sustainable, high-yield biomanufacturing that aligns perfectly with modern green chemistry principles while delivering the stringent purity profiles required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 4-hydroxyisoleucine was predominantly achieved through extraction from natural sources or multi-step chemical-enzymatic sequences that were inherently inefficient and costly. Chemical-enzymatic routes often involved six to eight distinct synthetic steps, starting from materials like ethyl 2-methylacetoacetate or tert-butyl bromoacetate, necessitating complex protection and deprotection strategies. These traditional pathways suffered from low overall conversion efficiencies and generated a plethora of intermediate byproducts, complicating the downstream purification process and drastically increasing the cost of goods sold. Furthermore, earlier biocatalytic attempts that relied on direct addition of alpha-ketoglutarate faced severe economic hurdles due to the high price of this co-substrate, which is consumed stoichiometrically in the reaction. The accumulation of inhibitory byproducts and the inability to maintain optimal enzyme activity over prolonged periods further restricted the achievable titers, making industrial-scale production financially unviable for many manufacturers.
The Novel Approach
The novel approach disclosed in the patent circumvents these obstacles by implementing a dynamic, two-stage biocatalytic system that regenerates the essential co-substrate alpha-ketoglutarate in real-time. Instead of purchasing expensive alpha-ketoglutarate, the process utilizes sodium glutamate monohydrate, a widely available and inexpensive bulk chemical, which is converted into alpha-ketoglutarate by L-glutamate oxidase. This clever coupling ensures a continuous supply of the co-substrate exactly where and when it is needed by the L-isoleucine dioxygenase, effectively driving the equilibrium towards the desired product. Additionally, the inclusion of catalase in the reaction mixture neutralizes hydrogen peroxide, a harmful byproduct of the oxidation step that would otherwise deactivate the sensitive dioxygenase enzyme. This synergistic multi-enzyme design not only simplifies the operational workflow but also significantly enhances the stability and longevity of the biocatalyst, enabling the system to sustain high reaction rates over extended durations.
Mechanistic Insights into Fe(II)/α-KG Dependent Dioxygenase Cascade
At the heart of this synthesis lies the Fe(II)/alpha-ketoglutarate-dependent dioxygenase superfamily, a group of enzymes renowned for their ability to activate inert C-H bonds under mild physiological conditions. In this specific application, L-isoleucine dioxygenase utilizes molecular oxygen to hydroxylate the C-4 position of L-isoleucine, a reaction that is strictly dependent on the presence of alpha-ketoglutarate as a co-substrate. The mechanistic cycle involves the coordination of Fe(II) at the active site, where it facilitates the oxidative decarboxylation of alpha-ketoglutarate to succinate and carbon dioxide, generating a highly reactive ferryl-oxo species. This high-energy intermediate is then responsible for the abstraction of a hydrogen atom from the substrate, leading to the formation of the hydroxylated product, 4-HIL. Understanding this precise mechanistic requirement is crucial for process optimization, as any imbalance in the ratio of co-substrate to substrate can lead to uncoupling of the reaction and loss of catalytic efficiency.
To address the stoichiometric demand for alpha-ketoglutarate, the patent engineers a parallel catalytic loop using L-glutamate oxidase, which oxidizes L-glutamate to alpha-ketoglutarate while releasing ammonia and hydrogen peroxide. The concurrent generation of hydrogen perpose poses a significant risk of oxidative damage to the iron center of the dioxygenase, potentially leading to irreversible enzyme inactivation. To mitigate this, the system incorporates bovine liver catalase, which rapidly decomposes hydrogen peroxide into water and oxygen, thereby maintaining a reducing environment conducive to enzyme stability. This tri-enzyme system—comprising the dioxygenase, the oxidase, and the catalase—operates in a finely tuned balance where the rate of co-substrate generation matches the consumption rate of the primary hydroxylation reaction. Such mechanistic harmony is essential for achieving the reported high conversion rates and minimizing the formation of unwanted side products like succinate.
How to Synthesize 4-Hydroxyisoleucine Efficiently
The practical implementation of this multi-enzyme cascade requires careful attention to reaction staging and substrate feeding strategies to maximize yield and minimize inhibition. The patent outlines a specific methodology where the initial phase focuses on the accumulation of alpha-ketoglutarate from glutamate, followed by the introduction of L-isoleucine and the necessary cofactors like Fe(II) and ascorbic acid. This sequential addition prevents the premature consumption of resources and allows for better control over the reaction kinetics. Detailed standardized synthesis steps, including specific buffer conditions, pH controls, and temperature parameters, are critical for replicating the high efficiency demonstrated in the experimental examples. For a comprehensive guide on executing this synthesis with precision, please refer to the structured protocol below.
- Prepare the biocatalytic system by expressing and purifying L-isoleucine dioxygenase and L-glutamate oxidase using affinity chromatography.
- Initiate the first stage reaction using sodium glutamate monohydrate, L-glutamate oxidase, and catalase to generate alpha-ketoglutarate.
- Execute the second stage by adding L-isoleucine, Fe(II), and ascorbic acid, utilizing a fed-batch strategy to maintain substrate levels and maximize conversion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this biocatalytic platform offers transformative advantages that directly address the core concerns of procurement managers and supply chain directors regarding cost stability and material availability. The substitution of expensive specialty chemicals with commodity-grade sodium glutamate fundamentally alters the cost structure of 4-HIL manufacturing, removing a significant variable expense from the bill of materials. This shift not only lowers the direct production costs but also insulates the supply chain from the volatility associated with niche chemical markets. Furthermore, the simplified downstream processing, resulting from fewer byproducts and a cleaner reaction profile, reduces the burden on purification infrastructure and shortens the overall production cycle time. These factors combine to create a more resilient and cost-effective supply model that can support the growing global demand for diabetes therapeutics without compromising on quality or delivery reliability.
- Cost Reduction in Manufacturing: The elimination of exogenous alpha-ketoglutarate purchase represents a substantial cost saving, as the process generates this critical reagent in situ from low-cost glutamate. By removing the need for expensive stoichiometric additives and reducing the complexity of waste treatment associated with chemical synthesis, the overall manufacturing expenditure is drastically optimized. The high conversion efficiency further ensures that raw material utilization is maximized, minimizing waste and enhancing the economic viability of the process on an industrial scale.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals like sodium glutamate monohydrate ensures a stable and secure supply of raw materials, mitigating the risks of shortages that often plague specialized reagent markets. The robustness of the enzymatic system, supported by the protective action of catalase, leads to consistent batch-to-batch performance, which is critical for maintaining uninterrupted production schedules. This reliability allows supply chain planners to forecast inventory needs with greater confidence and reduces the safety stock requirements typically associated with less predictable synthetic routes.
- Scalability and Environmental Compliance: The fed-batch strategy described in the patent effectively manages substrate inhibition, a common barrier to scaling biocatalytic processes, allowing for high substrate loading and increased volumetric productivity. The aqueous nature of the reaction and the use of biodegradable enzymes align with stringent environmental regulations, reducing the ecological footprint of the manufacturing process. This compliance simplifies the permitting process for new facilities and supports corporate sustainability goals, making the technology attractive for long-term investment and expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this multi-enzyme cascade technology for 4-HIL production. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the system's capabilities and requirements. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: How does the multi-enzyme system reduce the cost of 4-HIL synthesis?
A: The system utilizes inexpensive sodium glutamate monohydrate as a precursor to regenerate the costly co-substrate alpha-ketoglutarate in situ, eliminating the need for external addition of expensive reagents and simplifying downstream purification.
Q: What is the role of catalase in this biocatalytic process?
A: Catalase is essential for decomposing hydrogen peroxide (H2O2), a toxic byproduct generated by L-glutamate oxidase, thereby protecting the L-isoleucine dioxygenase from oxidative deactivation and ensuring high catalytic efficiency.
Q: Can this process be scaled for industrial pharmaceutical manufacturing?
A: Yes, the patent demonstrates a fed-batch strategy that effectively mitigates substrate inhibition, allowing for high substrate concentrations (up to 500 mM) and achieving conversion rates exceeding 93%, which indicates strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxyisoleucine Supplier
As the global demand for high-purity pharmaceutical intermediates continues to surge, partnering with a technically proficient manufacturer is paramount for ensuring product quality and supply continuity. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver exceptional results for our clients. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced biocatalytic capabilities, ensuring that every batch of 4-hydroxyisoleucine meets stringent purity specifications and regulatory standards. We understand the critical nature of your supply chain and are committed to providing a seamless, reliable sourcing experience that supports your drug development and commercialization timelines.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can specifically benefit your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of our biocatalytic routes compared to traditional methods. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations.
