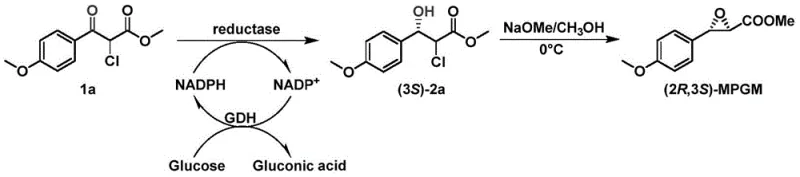
Advanced Biocatalytic Route for Diltiazem Intermediate Production and Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient pathways for the synthesis of critical cardiovascular drug intermediates, and patent CN113174377B represents a significant breakthrough in this domain. This intellectual property discloses a novel carbonyl reductase derived from Candida parapsilosis, along with specifically engineered mutants that exhibit dramatically improved stability and catalytic efficiency. The core innovation lies in the ability of these biocatalysts to perform the asymmetric reduction of methyl 2-chloro-3-(4-methoxyphenyl)-3-oxopropionate (compound 1a) with exceptional stereoselectivity. Unlike traditional chemical methods that often require harsh conditions or expensive chiral ligands, this enzymatic approach operates under mild physiological conditions, achieving conversion rates exceeding 99% and optical purity levels that meet the stringent requirements for diltiazem production. For R&D directors and process chemists, this technology offers a viable route to bypass the inherent 50% yield ceiling associated with classical kinetic resolution strategies, thereby unlocking substantial potential for process intensification and waste reduction in the manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of the key diltiazem intermediate (2R,3S)-MPGM has relied heavily on enzymatic resolution methods using lipases, a technique pioneered in the early 1990s. While effective in generating high optical purity, this approach suffers from a fundamental thermodynamic limitation: the maximum theoretical yield is capped at 50%. This is because the resolution process inherently discriminates against one enantiomer, leaving half of the starting material as an unwanted byproduct that is often difficult to recycle due to spontaneous decomposition. Furthermore, alternative chemical asymmetric reduction routes, such as those utilizing ruthenium catalysts reported by Genêt et al., while capable of higher yields, often necessitate the use of precious metals, high-pressure hydrogenation equipment, and rigorous exclusion of oxygen and moisture. These factors contribute to elevated capital expenditure and operational complexity, creating significant bottlenecks for supply chain managers who require consistent, scalable, and cost-effective production capabilities. The environmental burden of heavy metal waste and the economic inefficiency of discarding half the substrate mass render these conventional methods suboptimal for modern green chemistry standards.
The Novel Approach
The technology described in patent CN113174377B introduces a paradigm shift by employing a recombinant carbonyl reductase (CpKR) and its stability-enhanced mutants to catalyze the asymmetric reduction directly. This method transforms the prochiral ketone substrate 1a into the chiral alcohol (3S)-2a with a theoretical yield of 100%, effectively doubling the material efficiency compared to resolution techniques. The process leverages protein engineering to create mutants, such as M15, which possess multiple amino acid substitutions that confer superior thermal stability and operational robustness. This allows the reaction to proceed at higher substrate loadings, up to 100 mmol/L, without significant loss of enzyme activity. For procurement professionals, this translates to a drastic reduction in raw material consumption per kilogram of final product. The elimination of precious metal catalysts and the ability to run the reaction in aqueous buffer systems at ambient pressure significantly lower the barrier to entry for commercial scale-up, making this a highly attractive option for reducing lead time for high-purity pharmaceutical intermediates in a competitive market.
Mechanistic Insights into Carbonyl Reductase-Catalyzed Asymmetric Reduction
The core of this technological advancement is the precise stereochemical control exerted by the CpKR enzyme active site. The biocatalyst facilitates the hydride transfer from the cofactor NADPH to the si-face of the carbonyl group at the C3 position of substrate 1a, resulting in the formation of the (S)-configured hydroxyl group. This specific stereochemistry is crucial because the subsequent intramolecular SN2 cyclization reaction to form the epoxide ring in (2R,3S)-MPGM is strictly dependent on the configuration of this hydroxyl group. The enzyme's active site architecture ensures that only the desired enantiomer is produced, minimizing the formation of diastereomeric impurities that would otherwise complicate downstream purification. To maintain economic viability, the process incorporates a coupled enzyme system for cofactor regeneration. Glucose dehydrogenase (GDH) is added to the reaction mixture to oxidize glucose into gluconic acid, simultaneously reducing NADP+ back to NADPH. This cyclic regeneration means that only a catalytic quantity of the expensive cofactor is needed, sustaining the reduction cycle indefinitely as long as glucose is available.
From an impurity control perspective, the high enantioselectivity of the CpKR mutants simplifies the purification workflow significantly. In traditional chemical synthesis, removing trace amounts of the wrong enantiomer often requires costly chiral chromatography or multiple recrystallization steps. However, with the enzymatic route achieving >99% ee directly in the crude reaction mixture, the downstream processing is streamlined to simple extraction and crystallization. The stability data provided in the patent indicates that mutants like M15 retain activity for extended periods even at elevated temperatures (40°C), which suggests that the enzyme can withstand the exothermic nature of large-scale batch reactions without denaturing. This robustness is critical for maintaining consistent product quality across different production batches, ensuring that the final API intermediate meets the rigorous specifications required by global regulatory bodies. The mechanistic elegance of this system lies in its combination of high specificity, self-sustaining cofactor usage, and engineered durability.
How to Synthesize (2R,3S)-MPGM Efficiently
The synthesis of this critical cardiovascular intermediate involves a streamlined two-step sequence that begins with the biocatalytic reduction followed by a chemical cyclization. The process is designed to be operationally simple, requiring standard fermentation and chemical processing equipment that is readily available in most multipurpose pharmaceutical manufacturing facilities. The initial step focuses on the expression of the recombinant enzyme in a microbial host, followed by its application in a buffered aqueous system containing the substrate and a cheap sugar source for energy. This biological step sets the stereochemistry for the entire molecule, ensuring that the subsequent chemical transformation proceeds with high fidelity. For detailed laboratory protocols and specific parameter optimization, please refer to the standardized synthesis guide below.
- Clone and express the carbonyl reductase gene (CpKR) or its stability-enhanced mutants (e.g., M15) in E. coli BL21(DE3) using the pET28a vector.
- Prepare the biocatalytic reaction mixture containing substrate 1a (up to 100 mmol/L), glucose (1.0-1.5 molar ratio), NADP+ (0.2-0.5 mmol/L), and glucose dehydrogenase for cofactor recycling in phosphate buffer (pH 6.0).
- Incubate the reaction at 30°C until conversion exceeds 99%, extract the product (3S)-2a, and perform base-catalyzed cyclization with sodium methoxide to obtain (2R,3S)-MPGM.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this enzymatic technology offers compelling strategic advantages that go beyond simple technical metrics. The primary value driver is the fundamental improvement in atom economy; by shifting from a 50% yield resolution process to a near-quantitative asymmetric reduction, the effective demand for starting materials is halved for the same output volume. This structural change in the manufacturing process leads to substantial cost savings in raw material procurement, insulating the supply chain from volatility in the pricing of specialized organic precursors. Furthermore, the removal of heavy metal catalysts eliminates the need for expensive scavenging resins and complex waste treatment protocols associated with metal contamination, thereby reducing both operational expenditures and environmental compliance costs. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The transition to a 100% theoretical yield process fundamentally alters the cost structure of production. By eliminating the waste of half the substrate mass, which is inherent in resolution methods, manufacturers can achieve a drastic reduction in the cost of goods sold. Additionally, the use of a recombinant enzyme produced in E. coli avoids the licensing fees and high costs associated with precious metal catalysts like ruthenium. The simplified downstream processing, driven by high crude purity, further reduces solvent usage and labor hours required for purification, resulting in a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the engineered CpKR mutants ensures consistent production performance, minimizing the risk of batch failures due to enzyme instability. The reliance on widely available commodities such as glucose for cofactor regeneration removes dependency on specialized and potentially scarce chemical reagents. This simplifies the supplier base and reduces the risk of supply disruptions. Moreover, the scalability of fermentation-based enzyme production means that biocatalyst supply can be rapidly ramped up to meet surges in demand, providing a flexible and resilient supply chain capable of adapting to market dynamics without long lead times for catalyst synthesis.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic step significantly reduces the volume of organic solvents required compared to traditional chemical synthesis, easing the burden on solvent recovery systems and waste incineration capacity. The absence of toxic heavy metals simplifies regulatory filings and reduces the environmental liability associated with manufacturing. This green chemistry profile facilitates easier approval in regions with strict environmental regulations and enhances the marketability of the final drug product as being produced via sustainable methods. The process is inherently safer, operating at atmospheric pressure and moderate temperatures, which lowers insurance premiums and safety infrastructure costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic route. They are derived from the specific experimental data and comparative analysis presented in the patent documentation, aimed at clarifying the practical implications for industrial adoption. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: How does the CpKR mutant improve upon traditional lipase resolution methods?
A: Traditional lipase resolution is limited to a maximum theoretical yield of 50% because it discards the unwanted enantiomer. The CpKR mutant enables asymmetric reduction with a theoretical yield of 100%, effectively doubling the output from the same amount of starting material while operating under milder conditions.
Q: What are the stability characteristics of the M15 mutant compared to the wild type?
A: The M15 mutant features five specific amino acid substitutions that significantly enhance thermal stability. Data indicates that at 40°C, the half-life of the M15 mutant is drastically extended compared to the parental CpKR, allowing for robust performance in industrial batch processes without rapid enzyme deactivation.
Q: Is the cofactor NADPH cost-prohibitive for large-scale production?
A: No, the process utilizes an in situ cofactor regeneration system. By adding glucose and glucose dehydrogenase (GDH) to the reaction mixture, the oxidized NADP+ is continuously recycled back to NADPH, meaning only a catalytic amount of the expensive cofactor is required for the entire production run.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2R,3S)-MPGM Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic route described in patent CN113174377B for the production of diltiazem intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of (2R,3S)-MPGM meets the highest international standards for pharmaceutical grade materials. We are committed to leveraging advanced biocatalysis to deliver superior value to our partners in the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the specific economic benefits tailored to your production volume. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about adopting this high-efficiency manufacturing technology for your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
