Advanced Biocatalytic Synthesis of Diltiazem Intermediates Using Engineered Carbonyl Reductase Mutants
The pharmaceutical industry is constantly seeking more efficient and sustainable routes for the production of critical cardiovascular drug intermediates, particularly for blockbuster medications like diltiazem. A significant breakthrough in this domain is documented in patent CN113174377A, which discloses novel carbonyl reductases derived from Candida parapsilosis and their engineered mutants. This technology addresses the longstanding challenges associated with the synthesis of the chiral intermediate (2R,3S)-p-methoxyphenyl glycidic acid methyl ester [(2R,3S)-MPGM]. By leveraging advanced protein engineering techniques, the inventors have developed biocatalysts that exhibit exceptional stability and stereoselectivity, enabling a shift from traditional resolution methods to highly efficient asymmetric synthesis. For global procurement teams and R&D directors, this represents a pivotal opportunity to optimize supply chains for high-purity pharmaceutical intermediates while adhering to green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (2R,3S)-MPGM has relied heavily on enzymatic resolution using lipases or chemical asymmetric reduction. The lipase resolution method, while effective in achieving high optical purity, suffers from an inherent theoretical yield limitation of only 50%. This means that half of the valuable starting material is inevitably wasted or requires complex recycling processes, driving up raw material costs and environmental burden. Furthermore, chemical asymmetric reduction routes often necessitate the use of expensive chiral auxiliaries, harsh reaction conditions involving high pressure or extreme temperatures, and precious metal catalysts that require rigorous removal steps to meet pharmaceutical purity standards. These factors collectively contribute to a fragmented and cost-intensive manufacturing landscape that struggles to meet the growing global demand for diltiazem.
The Novel Approach
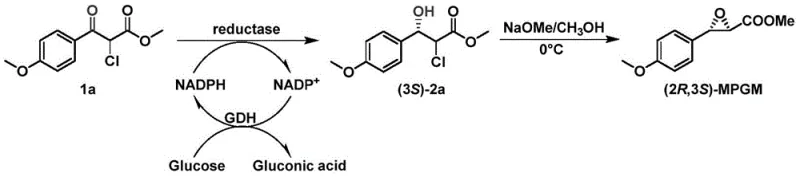
The innovative approach detailed in the patent utilizes recombinant carbonyl reductase mutants to catalyze the asymmetric reduction of the prochiral ketone substrate 1a directly to the chiral alcohol (3S)-2a with near-perfect selectivity. Unlike resolution methods, this asymmetric synthesis pathway boasts a theoretical yield of 100%, effectively doubling the output from the same amount of starting material. The process operates under mild physiological conditions, typically around 30°C and neutral pH, eliminating the need for energy-intensive heating or cooling systems. Moreover, the integration of a cofactor regeneration system ensures that the expensive NADPH coenzyme is recycled continuously, making the process economically viable on a large scale. This biological route not only simplifies the downstream purification process but also aligns perfectly with modern sustainability goals by reducing solvent usage and hazardous waste generation.
Mechanistic Insights into Carbonyl Reductase-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the precise molecular engineering of the carbonyl reductase enzyme. The wild-type enzyme, designated as CpKR, was subjected to site-directed mutagenesis to enhance its thermal stability and catalytic efficiency. Specific amino acid substitutions, such as replacing valine at position 24 with leucine and alanine at position 114 with valine, were identified as critical for stabilizing the protein structure against thermal denaturation. The mutant M15, in particular, combines multiple beneficial mutations (V24L, A114V, V120A, L169F, T181I) to achieve a half-life at 40°C that is orders of magnitude longer than the parent enzyme. This structural robustness allows the biocatalyst to maintain high activity over extended reaction periods, which is essential for achieving high substrate loading concentrations in industrial reactors without rapid loss of productivity.
The catalytic mechanism involves the stereoselective transfer of a hydride ion from the cofactor NADPH to the si-face of the carbonyl group in substrate 1a, resulting in the formation of the (S)-configured hydroxyl group. This stereochemical outcome is crucial because the subsequent intramolecular epoxidation step relies on the specific spatial arrangement of the hydroxyl and chloro groups to form the desired (2R,3S) epoxide ring. The enzyme's active site is finely tuned to exclude the formation of the unwanted (R)-enantiomer, ensuring that the crude product already possesses high optical purity before the final cyclization step. This high level of stereocontrol minimizes the formation of diastereomeric impurities, thereby simplifying the crystallization and purification stages required to meet stringent regulatory specifications for API intermediates.
How to Synthesize (2R,3S)-MPGM Efficiently
The synthesis of this critical diltiazem precursor involves a streamlined two-step biocatalytic and chemical sequence that can be readily implemented in standard fermentation and processing facilities. The process begins with the preparation of the recombinant biocatalyst, followed by the reduction reaction in an aqueous buffer system supplemented with a cofactor regeneration module. The high efficiency of the mutant enzymes allows for substrate concentrations as high as 100 mmol/L, demonstrating excellent tolerance to organic solvents and high substrate loads. Detailed standardized synthetic protocols, including specific buffer compositions, induction temperatures, and work-up procedures, are essential for reproducing the high yields and purity reported in the patent literature.
- Clone the carbonyl reductase gene (CpKR) or its mutant variants into an expression vector like pET28a and transform into E. coli BL21(DE3) for recombinant protein production.
- Cultivate the recombinant transformant in LB medium, induce expression with IPTG at low temperature (16°C), and harvest cells to obtain the biocatalyst.
- Perform asymmetric reduction of substrate 1a in a phosphate buffer with glucose and glucose dehydrogenase for cofactor regeneration, followed by epoxidation to yield (2R,3S)-MPGM.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond simple unit cost metrics. The shift from a 50% yield resolution process to a 100% yield asymmetric synthesis fundamentally alters the raw material balance sheet, effectively halving the requirement for the expensive chloroketone starting material per kilogram of final product. This drastic reduction in material intensity directly lowers the variable cost of goods sold (COGS) and reduces the exposure to volatility in upstream chemical markets. Additionally, the mild reaction conditions reduce the energy footprint of the manufacturing process, contributing to lower utility costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability targets.
- Cost Reduction in Manufacturing: The elimination of chiral resolving agents and the ability to recycle the cofactor system significantly lower the consumable costs associated with production. By avoiding the use of precious metal catalysts and harsh chemical reagents, the process also reduces the complexity and cost of waste treatment and disposal. The high space-time yield of the mutant enzymes means that smaller reactor volumes can produce the same output, optimizing capital expenditure and facility utilization rates.
- Enhanced Supply Chain Reliability: The superior thermal stability of the M15 mutant ensures consistent batch-to-batch performance, reducing the risk of production failures due to enzyme degradation. This reliability allows for tighter production scheduling and shorter lead times, as the process is less sensitive to minor fluctuations in operating conditions. Furthermore, the use of a recombinant E. coli expression system leverages well-established fermentation infrastructure, ensuring that the supply of the biocatalyst itself is robust and scalable.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium and the absence of heavy metals simplify the regulatory compliance landscape, facilitating faster approval for new manufacturing sites. The process generates significantly less organic waste compared to traditional chemical synthesis, aligning with strict environmental regulations in major pharmaceutical markets. This eco-friendly profile enhances the marketability of the final API to environmentally conscious partners and end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonyl reductase technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is critical for R&D teams planning technology transfer and for procurement teams negotiating supply agreements.
Q: What is the primary advantage of using CpKR mutants over traditional lipase resolution for diltiazem intermediates?
A: Traditional lipase resolution is limited by a maximum theoretical yield of 50%, whereas the asymmetric reduction using CpKR mutants offers a theoretical yield of 100%, significantly reducing raw material consumption and waste.
Q: How does the stability of the M15 mutant compare to the wild-type enzyme?
A: The M15 mutant exhibits drastically improved thermal stability, with a half-life at 40°C that is over 100-fold greater than the parent enzyme, allowing for more robust industrial processing conditions.
Q: Is the cofactor NADPH consumed stoichiometrically in this process?
A: No, the process utilizes a coupled enzyme system with glucose dehydrogenase (GDH) and glucose to regenerate NADPH in situ, meaning only a catalytic amount of the expensive cofactor is required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2R,3S)-MPGM Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in the production of complex pharmaceutical intermediates like (2R,3S)-MPGM. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis, to guarantee that every batch meets the exacting standards required for cardiovascular drug manufacturing.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your diltiazem supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in biocatalytic process development can drive value and efficiency for your organization.
