Scaling High-Purity Perfluoro-2,3-epoxy-2-methylpentane Production via Continuous Flow Technology
Introduction to Advanced Fluorine Intermediate Manufacturing
The global demand for environmentally benign fire suppression agents and electronic cleaning solvents has necessitated a paradigm shift in the synthesis of key fluorinated intermediates. Patent CN115385873A introduces a groundbreaking continuous synthesis method for perfluoro-2,3-epoxy-2-methylpentane, a critical precursor to perfluorohexanone. This technology represents a significant departure from legacy batch processes, leveraging continuous flow micro-reaction engineering to achieve unprecedented levels of efficiency and safety. By integrating a specific transition metal catalytic system with molecular oxygen oxidation, this method addresses the longstanding challenges of waste generation and process instability. For R&D directors and procurement strategists, understanding this technological leap is essential for securing a reliable supply chain of high-purity electronic chemical intermediates. The patent outlines a robust pathway that not only enhances product quality but also aligns with modern green chemistry principles, making it a cornerstone for future industrial applications in the specialty chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of perfluoro-2,3-epoxy-2-methylpentane has relied heavily on batch oxidation methods utilizing sodium hypochlorite as the primary oxidant, as documented in older literature such as Soviet patent SU666176A1. These traditional approaches suffer from severe drawbacks, including the generation of substantial volumes of saline wastewater which poses significant environmental disposal challenges and increases operational costs. Furthermore, sodium hypochlorite solutions are inherently unstable, often requiring on-site preparation at concentrations higher than commercially available standards to maintain efficacy, introducing variability and safety risks. Alternative gas-phase oxidation methods reported in Chinese patent CN105198719A attempted to mitigate solvent issues but struggled with relatively low conversion rates and selectivity, complicating downstream purification. The batch nature of these legacy processes also results in poor heat dissipation during the exothermic oxidation step, creating potential hotspots that can degrade the sensitive epoxide product or lead to safety incidents.
The Novel Approach
In stark contrast, the novel approach detailed in CN115385873A utilizes a continuous flow micro-reactor system that fundamentally transforms the reaction dynamics. By employing molecular oxygen as the oxidant in conjunction with a recyclable transition metal catalyst system, the process eliminates the need for hazardous halogenated oxidants and drastically reduces waste streams. The micro-reactor architecture ensures rapid mixing and superior heat transfer, allowing for precise control over reaction parameters such as temperature and residence time. This precision prevents the over-oxidation of the epoxide product, a common issue in batch reactors, thereby maintaining high selectivity profiles. The continuous nature of the operation facilitates the immediate separation of the product from the catalytic phase, enabling the direct recycling of the catalyst solution. This innovation not only streamlines the manufacturing workflow but also establishes a new benchmark for sustainability and efficiency in the production of complex fluorine fine chemicals.
Mechanistic Insights into Transition Metal-Catalyzed Epoxidation
The core of this technological advancement lies in the sophisticated catalytic system comprising a transition metal salt, specifically cobalt chloride or cobalt acetate, coordinated with a polypyridine amino compound ligand. This coordination complex acts as a highly efficient mediator for the activation of molecular oxygen, facilitating the selective transfer of an oxygen atom to the double bond of perfluoro-2-methyl-2-pentene. The polypyridine ligands, such as tris(2-pyridylmethyl)amine or N,N'-dimethyl-N,N'-bis(2-pyridylmethyl)ethylenediamine, play a crucial role in stabilizing the metal center and modulating its electronic properties to favor epoxidation over competing side reactions. The structural integrity of these ligands is paramount, as depicted in the chemical diagrams below, ensuring that the catalytic cycle remains robust under the continuous flow conditions.
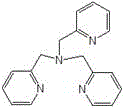
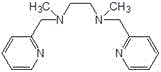
Furthermore, the integration of this catalytic chemistry within a micro-reactor environment leverages the principles of process intensification. The narrow channels of the micro-reactor create a high surface-area-to-volume ratio, which significantly enhances the mass transfer of gaseous oxygen into the liquid reaction phase. This improved gas-liquid contact is critical for maintaining a consistent concentration of the active oxidizing species throughout the reaction zone. Consequently, the system achieves conversion rates ranging from 90% to 98% and selectivity between 96% and 99%, metrics that are difficult to replicate in conventional stirred tank reactors. The precise control over residence time, typically maintained between 1 to 5 minutes, ensures that the reactive epoxide intermediate is quickly quenched or separated, minimizing degradation and preserving the purity required for downstream applications in electronic cleaning and fire suppression.
How to Synthesize Perfluoro-2,3-epoxy-2-methylpentane Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to strict protocol regarding the preparation of the catalytic solution and the operation of the continuous flow equipment. The process begins with the formation of a homogeneous catalytic liquid by mixing the transition metal salt and the polypyridine ligand in an aprotic solvent such as DMF or acetonitrile. This solution is then pumped into the micro-reactor alongside the perfluoro-alkene substrate and oxygen gas. The detailed standardized synthesis steps, including specific flow rates, molar ratios, and pressure settings required to replicate the high yields reported in the patent, are outlined in the technical guide below.
- Prepare a homogeneous catalytic solution by mixing transition metal salts (such as CoCl2 or CoAc2) with polypyridine amino compounds in an aprotic solvent like DMF.
- Pump the perfluoro-2-methyl-2-pentene substrate, the prepared catalytic solution, and oxygen gas simultaneously into a continuous flow micro-reactor.
- Maintain reaction conditions between 100-130°C and 0.5-2.0MPa pressure with a residence time of 1-5 minutes to achieve high conversion and selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous synthesis technology offers transformative benefits that extend beyond mere technical performance. The shift from batch to continuous processing inherently reduces the physical footprint of the manufacturing facility while increasing the throughput capacity, allowing for a more agile response to market demand fluctuations. By eliminating the reliance on unstable and waste-generating oxidants like sodium hypochlorite, the process significantly lowers the costs associated with raw material procurement and hazardous waste disposal. The ability to recycle the catalytic solution multiple times without significant loss of activity further contributes to substantial cost savings in manufacturing, reducing the overall consumption of expensive transition metal salts and ligands. This economic efficiency makes the production of high-purity fluorine intermediates more viable on a commercial scale.
- Cost Reduction in Manufacturing: The implementation of this oxygen-based oxidation system removes the need for purchasing and handling large quantities of chemical oxidants, which traditionally account for a significant portion of variable costs. Additionally, the recycling of the catalytic phase means that the effective cost of the catalyst per kilogram of product is drastically reduced over time. The continuous nature of the process also minimizes solvent usage and energy consumption per unit of output compared to batch heating and cooling cycles. These factors combine to create a leaner cost structure that enhances competitiveness in the global market for specialty fluorine chemicals without compromising on quality standards.
- Enhanced Supply Chain Reliability: Continuous flow manufacturing provides a consistent and steady output of product, mitigating the risks of batch-to-batch variability that can disrupt downstream formulation processes. The intrinsic safety of the micro-reactor system, characterized by low hold-up volumes and excellent thermal control, reduces the likelihood of unplanned shutdowns due to safety incidents. Furthermore, the use of molecular oxygen, a readily available industrial gas, simplifies the raw material supply chain compared to sourcing specialized and potentially regulated chemical oxidants. This reliability ensures a stable supply of critical intermediates for clients in the electronics and fire safety industries, fostering long-term partnerships based on trust and consistency.
- Scalability and Environmental Compliance: One of the most significant advantages of micro-reactor technology is the ease of scale-up through numbering-up rather than scaling-up vessel size, which preserves the reaction kinetics and safety profile. This allows for rapid expansion of production capacity to meet growing demand without the need for massive capital investment in new infrastructure. From an environmental perspective, the process generates minimal waste, primarily consisting of recyclable solvents and catalyst residues, aligning with increasingly stringent global environmental regulations. The reduction in saline wastewater and hazardous byproducts simplifies compliance reporting and reduces the environmental liability associated with chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of perfluoro-2,3-epoxy-2-methylpentane. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of this method. Understanding these details is crucial for stakeholders evaluating the integration of this technology into their existing supply chains or R&D pipelines. The responses highlight the robustness of the catalytic system and the operational flexibility offered by the continuous flow approach.
Q: What are the primary advantages of using oxygen over sodium hypochlorite in this synthesis?
A: Using molecular oxygen eliminates the generation of large volumes of saline waste water associated with sodium hypochlorite oxidation, significantly reducing environmental disposal costs and improving the atom economy of the process.
Q: Can the catalytic system be recycled in the continuous flow process?
A: Yes, the patent data indicates that the upper layer catalytic solution can be separated and recycled for at least three cycles with no significant loss in catalytic activity, enhancing process economics.
Q: How does the micro-reactor improve safety compared to traditional batch reactors?
A: The micro-reactor technology minimizes the hold-up volume of reactive materials at any given time and offers superior heat transfer capabilities, effectively mitigating the risks of thermal runaway associated with exothermic oxidation reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoro-2,3-epoxy-2-methylpentane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of next-generation fire suppression agents and electronic cleaning fluids. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced continuous flow technologies similar to those described in recent patents, we can offer our partners a supply of perfluoro-2,3-epoxy-2-methylpentane that is both cost-effective and environmentally responsible.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our product against your internal standards, we are ready to assist. Please reach out to us for detailed route feasibility assessments and to explore how we can collaborate to drive efficiency and innovation in your fluorine chemical projects. Together, we can build a more sustainable and resilient supply chain for the future.
