Advanced Continuous Flow Synthesis of Perfluoro-2,3-Epoxy-2-Methylpentane for Commercial Scale-Up
The global demand for high-performance fluorinated materials, particularly in the electronics and fire suppression sectors, has necessitated a paradigm shift in how key intermediates are manufactured. Patent CN115385873B introduces a groundbreaking continuous synthesis method for perfluoro-2,3-epoxy-2-methylpentane, a critical precursor to the environmentally friendly fire extinguishing agent perfluorohexanone. This technology represents a significant leap forward from traditional batch processing, leveraging the precision of continuous flow micro-reactors to achieve unprecedented levels of safety and efficiency. By replacing hazardous chemical oxidants with molecular oxygen and utilizing a recyclable transition metal catalyst system, this process addresses the long-standing challenges of waste generation and reaction control inherent in fluorine chemistry. For industry leaders seeking a reliable fluorine fine chemical supplier, understanding the mechanistic advantages of this patent is crucial for securing a sustainable supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluoro-2,3-epoxy-2-methylpentane has relied heavily on batch oxidation methods using sodium hypochlorite as the primary oxidant. As detailed in the background art of the patent, these conventional approaches suffer from severe drawbacks, including the instability of self-prepared sodium hypochlorite solutions which often exceed safe concentration limits. Furthermore, the batch nature of these reactions leads to poor heat and mass transfer, resulting in inconsistent product quality and significant safety risks due to the exothermic nature of the oxidation. The generation of large volumes of saline wastewater containing residual oxidants and organic solvents creates a substantial environmental burden, complicating compliance with increasingly stringent global regulations. Additionally, the inability to effectively recycle the oxidant or catalyst in these batch systems drives up raw material costs and operational expenditures, making the traditional route economically unsustainable for large-scale commercial production.
The Novel Approach
In stark contrast, the novel approach described in patent CN115385873B utilizes a continuous flow micro-reactor system that fundamentally alters the reaction dynamics. By employing molecular oxygen as the oxidant in the presence of a specialized transition metal catalyst, the process eliminates the need for hazardous hypochlorite reagents entirely. The micro-reactor environment ensures rapid mixing and precise temperature control, which suppresses side reactions and prevents the over-oxidation of the sensitive epoxide product. This method not only enhances the intrinsic safety of the operation by minimizing the inventory of reactive materials at any given time but also enables the continuous recycling of the catalytic solution. The result is a streamlined manufacturing process that delivers high-purity products with minimal waste, offering a compelling value proposition for cost reduction in fluorine chemical manufacturing through improved atom economy and reduced disposal costs.
Mechanistic Insights into Co-Catalyzed Oxygen Epoxidation
The core of this technological breakthrough lies in the sophisticated design of the catalytic system, which facilitates the activation of molecular oxygen under mild conditions. The catalyst is formed by coordinating transition metal salts, such as cobalt chloride (CoCl2) or cobalt acetate (CoAc2), with specific polypyridine amino ligands. These ligands, including tris(2-pyridylmethyl)amine and N,N'-dimethyl-N,N'-bis(2-pyridylmethyl)ethylenediamine, create a stable coordination environment around the metal center that promotes the selective transfer of oxygen to the perfluoro-olefin substrate. The structural integrity of these ligands is paramount for maintaining catalytic activity over multiple cycles, as they prevent the aggregation of metal species that typically leads to deactivation.
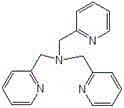
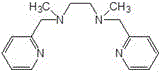
The synergy between the catalyst and the micro-reactor geometry is what drives the high performance of this process. In the micro-channels, the gas-liquid mass transfer coefficient is significantly enhanced compared to stirred tank reactors, ensuring that dissolved oxygen is readily available at the catalytic active sites. This efficient mass transfer, combined with the precise residence time control (typically 1 to 5 minutes), prevents the degradation of the epoxide product which is prone to further oxidation or hydrolysis in prolonged reaction times. Consequently, the process achieves conversion rates of 90% to 98% and selectivity of 96% to 99%, demonstrating superior control over the reaction pathway. For R&D directors, this level of mechanistic control translates directly into a cleaner impurity profile, simplifying downstream purification and ensuring the final product meets the rigorous specifications required for electronic grade applications.
How to Synthesize Perfluoro-2,3-Epoxy-2-Methylpentane Efficiently
Implementing this continuous synthesis route requires careful attention to the preparation of the catalytic feed and the optimization of flow parameters within the micro-reactor system. The process begins with the formation of a homogeneous catalytic solution by mixing the transition metal salt and the polypyridine ligand in an aprotic solvent such as DMF or acetonitrile. This solution is then pumped concurrently with the perfluoro-2-methyl-2-pentene substrate and oxygen gas into the heated micro-reactor. The detailed standardized synthesis steps, including specific molar ratios and flow rates validated by experimental data, are outlined below to ensure reproducibility and safety during scale-up.
- Prepare a homogeneous catalytic solution by mixing transition metal salts (e.g., CoCl2), polypyridine amino compounds, and an aprotic solvent like DMF.
- Pump perfluoro-2-methyl-2-pentene, the catalytic solution, and oxygen gas simultaneously into a continuous flow microreactor maintained at 60-150°C and 0.5-2.0MPa.
- Separate the reaction effluent to isolate the product layer, while recycling the upper catalytic layer for subsequent reaction cycles to maximize efficiency.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from batch hypochlorite oxidation to continuous oxygen epoxidation offers transformative benefits that extend beyond mere technical metrics. The elimination of sodium hypochlorite removes a major logistical and safety bottleneck, as this reagent is difficult to transport and store in large quantities due to its instability. By switching to molecular oxygen, which can be sourced onsite or delivered via standard industrial gas supply chains, the dependency on volatile chemical markets is reduced, leading to enhanced supply chain reliability. Furthermore, the ability to recycle the catalytic liquid multiple times without significant loss of activity means that the consumption of expensive transition metals and specialized ligands is drastically minimized, driving down the variable cost of goods sold.
- Cost Reduction in Manufacturing: The implementation of this continuous flow technology fundamentally alters the cost structure of producing perfluoro-2,3-epoxy-2-methylpentane by removing the need for stoichiometric amounts of chemical oxidants. In traditional methods, the oxidant is consumed and generates waste, whereas here, oxygen acts as a clean reagent, and the catalyst is recycled. This shift eliminates the costs associated with purchasing, handling, and disposing of large volumes of hazardous hypochlorite solutions. Additionally, the high selectivity of the reaction reduces the burden on downstream separation units, lowering energy consumption and solvent usage during purification, which collectively contributes to substantial cost savings in the overall manufacturing process.
- Enhanced Supply Chain Reliability: Adopting a continuous manufacturing model significantly mitigates the risks associated with batch-to-batch variability and production downtime. The micro-reactor system operates with a small hold-up volume, meaning that in the event of a process deviation, the amount of affected material is negligible, ensuring consistent product quality. Moreover, the modular nature of flow chemistry allows for flexible capacity expansion; additional reactor modules can be added in parallel to meet increasing demand without the need for constructing massive new facilities. This scalability ensures that suppliers can maintain steady delivery schedules even during market fluctuations, providing buyers with a secure and predictable source of high-purity fluorine intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with green chemistry principles by minimizing waste generation at the source. The absence of saline wastewater and chlorinated by-products simplifies effluent treatment and reduces the facility's environmental footprint. The continuous nature of the reaction also improves energy efficiency through better heat integration and reduced heating/cooling cycles compared to batch operations. These factors make the technology highly scalable and compliant with strict environmental regulations, future-proofing the supply chain against potential regulatory tightening regarding fluorine chemical production and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of perfluoro-2,3-epoxy-2-methylpentane. These answers are derived directly from the experimental data and technical specifications provided in patent CN115385873B, offering clarity on the feasibility and advantages of this advanced manufacturing route. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing supply networks.
Q: What are the primary safety advantages of this continuous flow method over traditional batch oxidation?
A: The continuous flow method utilizes molecular oxygen instead of unstable sodium hypochlorite, significantly reducing the risk of explosive decomposition and eliminating the handling hazards associated with large volumes of hazardous oxidants in batch reactors.
Q: How does the catalyst system contribute to cost reduction in fluorine chemical manufacturing?
A: The specific transition metal-polypyridine catalyst system allows for the recycling of the catalytic liquid multiple times without significant loss of activity, thereby drastically reducing the consumption of expensive metal salts and ligands per unit of product.
Q: What conversion rates and selectivity can be achieved with this microreactor technology?
A: According to patent data, the optimized continuous process achieves conversion rates between 90% and 98%, with selectivity ranging from 96% to 99%, ensuring high purity and minimizing downstream purification costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoro-2,3-Epoxy-2-Methylpentane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced fluorine chemicals requires more than just laboratory success; it demands robust engineering and unwavering commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless. Our state-of-the-art facilities are equipped with the necessary continuous flow infrastructure to implement the oxygen epoxidation technology described in CN115385873B, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are dedicated to delivering high-purity perfluoro-2,3-epoxy-2-methylpentane that meets the exacting standards of the global electronics and fire safety industries.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this continuous process can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply of this critical fluorine intermediate.
