Revolutionizing Atorvastatin Intermediate Production via Safe Lithiation and Dehydration Pathways
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the production of high-value statin intermediates. A significant breakthrough in this domain is detailed in patent CN108586427B, which outlines a novel preparation method for a critical atorvastatin calcium intermediate. This technology addresses the longstanding safety and regulatory challenges associated with traditional cyanide-based syntheses. By shifting towards a lithiation and dehydration strategy, the process not only enhances operator safety but also aligns with modern green chemistry principles required by top-tier regulatory bodies. For global procurement teams and R&D directors, understanding this shift is vital for securing a reliable atorvastatin intermediate supplier capable of meeting stringent compliance standards without compromising on volume or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
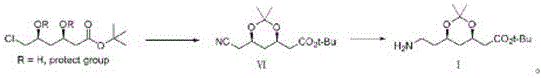
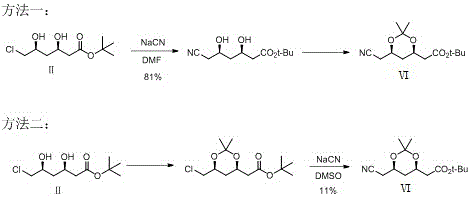
Historically, the synthesis of Compound VI, a pivotal precursor to atorvastatin calcium, has been plagued by the reliance on highly toxic reagents. As illustrated in the prior art, traditional routes predominantly utilize sodium cyanide (NaCN) for nucleophilic substitution or hydrocyanic acid for reaction media. These substances present catastrophic risks; they are acutely toxic to human physiology and pose severe threats to environmental ecosystems if not managed with extreme precision. Furthermore, the operational conditions for these legacy methods are often harsh, with some protocols requiring temperatures as high as 100°C and reaction times extending up to 30 hours. The variability in yield is another critical pain point, with some conventional pathways delivering as little as 11% efficiency, creating significant bottlenecks in cost reduction in pharmaceutical manufacturing and supply chain stability.
The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a paradigm shift by completely excising cyanide from the synthetic tree. The new route leverages a sophisticated sequence starting with the lithiation of a chloro-substituted precursor, followed by esterification, amidation, and finally, a controlled dehydration to install the nitrile group. This approach operates under much milder conditions, often utilizing temperatures ranging from -30°C to room temperature, which significantly reduces energy consumption and thermal stress on the equipment. By replacing hazardous cyanide salts with accessible reagents like lithium metal and methyl chloroformate, the process drastically simplifies the safety infrastructure required for production. This transition not only mitigates the risk of workplace exposure but also streamlines the regulatory approval process for the final API, making it a superior choice for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lithiation and Dehydration Cascade
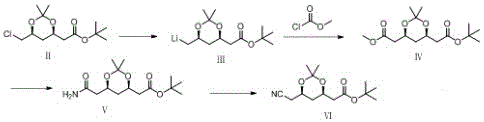
The core of this innovative synthesis lies in the precise control of organometallic transformations and functional group interconversions. The process initiates with the reaction of Compound II with a lithium metal reagent in an anhydrous ether or tetrahydrofuran solvent system at low temperatures (-5 to 10°C). This step generates a reactive organolithium species (Compound III) via halogen-lithium exchange, a transformation that requires rigorous exclusion of moisture to prevent quenching. Subsequently, this intermediate is trapped with methyl chloroformate at cryogenic conditions (-30 to 0°C) to form the ester Compound IV. The低温 control here is paramount to suppress side reactions such as over-lithiation or decomposition of the sensitive acetal protecting groups. The subsequent amidation with ammonia in methanol proceeds smoothly under alkaline conditions, converting the ester to the primary amide (Compound V) with high fidelity, setting the stage for the final nitrile installation.
The final transformation involves the dehydration of the amide Compound V to the nitrile Compound VI, a step traditionally fraught with impurities in other contexts. Here, the use of dehydrating agents like thionyl chloride or phosphorus oxychloride in the presence of a base such as pyridine ensures a clean conversion. This mechanism avoids the formation of polymeric byproducts often seen in harsher dehydration conditions. Furthermore, the final hydrogenation of Compound VI to Compound I utilizes heterogeneous catalysts like palladium on carbon or Raney nickel in acidic media (acetic acid). This reduction step is highly selective, reducing the nitrile to the primary amine without affecting the acid-sensitive dioxolane ring or the tert-butyl ester. This level of chemoselectivity is crucial for maintaining the impurity profile within the tight specifications demanded by pharmacopeial standards, ensuring the material is suitable for downstream coupling reactions.
How to Synthesize Atorvastatin Calcium Intermediate Efficiently
Implementing this synthesis requires a disciplined approach to reaction engineering, particularly regarding temperature gradients and reagent addition rates. The patent provides a clear roadmap for executing the lithiation and subsequent functionalization steps with high reproducibility. Operators must ensure that the lithium metal is fully activated and that the solvent systems are strictly anhydrous to maximize the yield of the organolithium intermediate. The quenching and workup procedures are equally critical, involving careful pH adjustments and extraction protocols to isolate the intermediates in high purity. For a detailed breakdown of the specific molar ratios, solvent volumes, and isolation techniques that guarantee success, please refer to the standardized protocol below.
- React Compound II with a lithium metal reagent in ether or THF at -5 to 10°C to generate the organolithium intermediate Compound III.
- Mix Compound III with methyl chloroformate at low temperature (-30 to 0°C), then warm to room temperature to form the ester Compound IV.
- React Compound IV with an ammonia methanol solution under alkaline conditions to produce the amide Compound V.
- Dehydrate Compound V using thionyl chloride or phosphorus oxychloride to yield the nitrile Compound VI, followed by hydrogenation to obtain Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this cyanide-free methodology offers profound advantages that extend beyond mere chemical elegance. The elimination of sodium cyanide and hydrocyanic acid removes a massive logistical and compliance burden from the supply chain. Procuring and transporting Class A poisons requires specialized licensing, secure storage facilities, and rigorous tracking, all of which inflate the overhead costs of production. By switching to reagents like lithium metal and common organic solvents, manufacturers can significantly reduce the administrative friction and insurance premiums associated with raw material handling. This simplification directly translates to a more resilient supply chain, less prone to disruptions caused by regulatory crackdowns on hazardous chemical transport.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the removal of expensive waste treatment protocols associated with cyanide detoxification. Traditional processes require extensive downstream processing to neutralize toxic effluents before discharge, a costly operation that adds no value to the final product. By avoiding these toxins entirely, the new method minimizes waste disposal fees and reduces the complexity of the wastewater treatment infrastructure. Additionally, the higher yields observed in key steps, such as the 91% conversion in the final hydrogenation, mean that less raw material is wasted per kilogram of output, effectively lowering the cost of goods sold (COGS) without the need for aggressive price negotiation.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than restricted toxic substances enhances the reliability of the supply chain. Lithium metal, methyl chloroformate, and standard dehydrating agents are widely available from multiple global vendors, reducing the risk of single-source dependency. In contrast, the supply of high-purity sodium cyanide can be volatile due to fluctuating environmental regulations and limited manufacturer capacity. This diversification of the raw material base ensures consistent production schedules and shorter lead times for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their inventory with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: Scaling this process to multi-ton production is inherently safer and more straightforward. The reaction conditions, primarily operating near ambient temperatures after the initial cryogenic steps, are easier to manage in large-scale reactors compared to the high-temperature (100°C) requirements of older methods. This thermal efficiency reduces the energy load on the plant. Moreover, the environmental footprint is drastically smaller, aligning with the increasingly strict ESG (Environmental, Social, and Governance) mandates of multinational pharmaceutical corporations. Facilities adopting this technology can market their production capabilities as 'green' or 'sustainable,' a significant competitive advantage when bidding for contracts with eco-conscious global partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. They are designed to clarify the operational feasibility and the strategic benefits of transitioning to this safer manufacturing platform for key stakeholders in the pharmaceutical value chain.
Q: Why is the new synthesis method for Atorvastatin intermediates considered safer than conventional routes?
A: Conventional methods rely heavily on sodium cyanide (NaCN) or hydrocyanic acid, which are extremely toxic and pose severe environmental and safety risks. The novel process described in patent CN108586427B completely eliminates the need for these hazardous cyanide sources, utilizing safer reagents like lithium metal and dehydrating agents instead.
Q: What are the typical yields achieved in the key steps of this new preparation method?
A: The patent data indicates robust yields across the synthetic sequence. For instance, the esterification step yields approximately 70% of Compound IV, the amidation step achieves around 85% for Compound V, and the final dehydration to Compound VI reaches 80%. The subsequent hydrogenation to Compound I demonstrates a high yield of roughly 91%.
Q: Which catalysts are suitable for the final hydrogenation step to produce Compound I?
A: The process allows for flexibility in catalyst selection to optimize cost and availability. Suitable catalysts include palladium on carbon (Pd/C), platinum on carbon (Pt/C), or Raney nickel. These can be used individually or in mixtures, typically in solvents like acetic acid or alcohols at mild temperatures between 20-30°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Calcium Intermediate Supplier
The technological advancements detailed in patent CN108586427B represent a significant leap forward in the manufacturing of statin intermediates, offering a pathway that is both chemically robust and commercially viable. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into full-scale commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of temperature and reagent stoichiometry required for this lithiation-dehydration sequence is maintained with precision. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for GMP-grade API synthesis.
We invite potential partners to engage with us to explore how this optimized route can enhance your supply chain security and cost structure. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to secure a sustainable and efficient supply of high-quality atorvastatin intermediates for your global operations.
