Advanced Synthesis of Chiral Succinic Acid Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously demands more efficient routes for complex chiral building blocks, particularly those serving as precursors for histone deacetylase (HDAC) inhibitors and other epigenetic modulators. Patent CN1068876C presents a groundbreaking methodology for the preparation of chiral succinic acid derivatives, specifically targeting compounds of general formula I which possess valuable pharmacological properties. This patent outlines a sophisticated multi-step synthesis that begins with the construction of a chiral backbone via asymmetric catalysis and proceeds through a unique hydantoin-mediated alkylation strategy. The core innovation lies in the ability to generate high optical purity intermediates without relying solely on expensive chiral pool materials, instead utilizing a catalytic asymmetric ene reaction followed by diastereoselective crystallization. This approach not only enhances the overall yield but also significantly simplifies the purification protocols required for GMP-grade production. By integrating robust chemical transformations such as thermal decarboxylation and selective hydrogenolysis, the process ensures a reliable supply of high-quality intermediates essential for modern drug discovery pipelines.
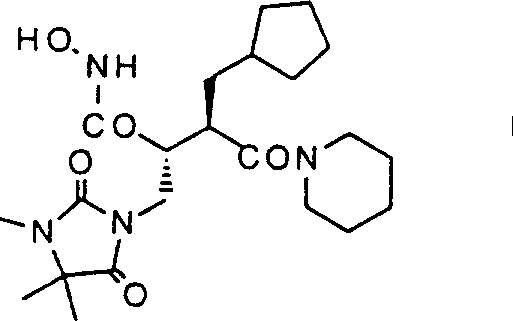
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing substituted succinic acid scaffolds often rely heavily on resolution of racemic mixtures, which inherently limits the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed. Furthermore, conventional alkylation strategies frequently suffer from poor regioselectivity and over-alkylation side reactions, leading to complex impurity profiles that are difficult to separate on a commercial scale. Many existing methods require harsh reaction conditions or exotic reagents that pose significant safety hazards and environmental burdens during waste disposal. The reliance on chromatographic purification for removing diastereomers adds substantial cost and time to the manufacturing process, creating bottlenecks in the supply chain for active pharmaceutical ingredients. Additionally, the introduction of the hydroxamic acid functionality, critical for metal-chelating activity in many drugs, is often performed late in the synthesis using unstable reagents, resulting in variable batch-to-batch consistency and lower overall process reliability.
The Novel Approach
The methodology described in CN1068876C overcomes these historical challenges by introducing a streamlined sequence that prioritizes stereochemical control at the earliest possible stage. The use of a (R)-binaphthyloxytitanium catalyst in the initial glyoxylate-ene reaction establishes the chiral center with exceptional enantiomeric excess, reportedly reaching approximately 98% ee even at room temperature, which is a significant improvement over prior art requiring cryogenic conditions. Subsequent alkylation with 3-bromomethyl-1,5,5-trimethylhydantoin provides a stable intermediate that directs the stereochemistry of further substitutions. A key feature of this novel approach is the in situ decarboxylation step coupled with salt formation using (+)-pseudoephedrine, which effectively upgrades the optical purity through crystallization rather than chromatography. This strategic combination of asymmetric catalysis and classical resolution techniques results in a process that is not only chemically elegant but also commercially viable for large-scale operations.
Mechanistic Insights into Asymmetric Glyoxylate-Ene Reaction and Hydantoin Alkylation
The foundation of this synthesis rests on the highly stereoselective reaction between a glyoxylic acid derivative and methylenecyclopentane, catalyzed by a chiral titanium complex. The mechanism involves the coordination of the Lewis acidic titanium center to the carbonyl oxygen of the glyoxylate, activating it towards nucleophilic attack by the alkene. The chiral binaphthyl ligand creates a rigid steric environment that forces the alkene to approach from a specific face, thereby dictating the absolute configuration of the newly formed carbon-carbon bond. This step is critical as it sets the stereochemistry for the entire molecule, and the patent highlights that the reaction can proceed efficiently at temperatures ranging from -70°C to 35°C, with optimal results observed near room temperature. The resulting hydroxy ester is then activated, typically via sulfonation with o-nitrobenzenesulfonyl chloride, to create a superior leaving group for the subsequent alkylation step.
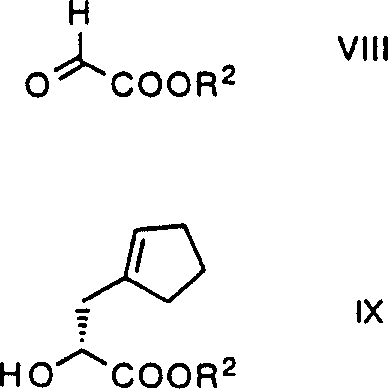
Following the establishment of chirality, the synthesis proceeds through a crucial alkylation phase where the triester intermediate reacts with 3-bromomethyl-1,5,5-trimethylhydantoin in the presence of a strong base like sodium hydride. This reaction installs the hydantoin moiety, which serves as a masked amine equivalent and a structural scaffold for the final pharmacophore. The mechanism likely involves the deprotonation of the malonate-derived triester to form a stabilized enolate, which then attacks the bromomethyl group of the hydantoin in an SN2 fashion. The patent specifies that this step can be conducted in polar aprotic solvents such as DMF or DMSO, ensuring high solubility and reaction rates. Following alkylation, the process employs a hydrogenolysis step to remove benzyl protecting groups, followed by a thermal decarboxylation. This decarboxylation is remarkably selective, favored by the use of N-methylmorpholine in propyl acetate, which helps to drive the equilibrium towards the desired mono-acid product while minimizing epimerization at the alpha-carbon.
How to Synthesize Chiral Succinic Acid Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maintain the integrity of the chiral centers and ensure high purity. The process begins with the preparation of the chiral hydroxy ester, followed by activation and alkylation with the hydantoin derivative. Subsequent steps involve careful manipulation of protecting groups and functional group interconversions to arrive at the final hydroxamic acid structure. Operators must pay close attention to temperature controls during the decarboxylation and salt formation steps, as these are critical for achieving the desired diastereomeric ratio. The detailed standardized synthesis steps below outline the specific conditions and reagent stoichiometries required to replicate the high yields and purity reported in the patent literature.
- Perform an asymmetric glyoxylate-ene reaction using a (R)-binaphthyloxytitanium catalyst to establish chirality.
- Alkylate the resulting triester with 3-bromomethyl-1,5,5-trimethylhydantoin using sodium hydride in DMF.
- Execute hydrogenolysis followed by thermal decarboxylation and chiral salt formation with (+)-pseudoephedrine for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers distinct strategic advantages over legacy manufacturing routes. The primary benefit stems from the significant reduction in processing steps required to achieve high optical purity, eliminating the need for costly and time-consuming preparative chiral chromatography. By relying on crystallization-induced diastereomeric enrichment, the process drastically reduces solvent consumption and waste generation, aligning with green chemistry principles and lowering environmental compliance costs. The use of readily available starting materials, such as methylenecyclopentane and glyoxylic acid esters, ensures a stable and resilient supply chain that is less susceptible to market fluctuations associated with exotic chiral pool reagents. Furthermore, the robustness of the reaction conditions, particularly the ability to perform key steps at ambient or moderately elevated temperatures, reduces energy consumption and simplifies the engineering requirements for reactor infrastructure.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the later stages of the synthesis, relying instead on organic bases and thermal processes, removes the need for expensive heavy metal scavenging units. This simplification directly translates to lower capital expenditure and operational costs, as the purification train becomes less complex. The high yield of the alkylation and decarboxylation steps minimizes raw material waste, ensuring that a greater proportion of input mass is converted into saleable product. Additionally, the ability to recycle solvents like propyl acetate and ethyl acetate further enhances the economic viability of the process, providing substantial cost savings compared to methods requiring specialized fluorinated solvents or cryogenic cooling.
- Enhanced Supply Chain Reliability: The synthetic route utilizes commodity chemicals that are widely produced by multiple global suppliers, reducing the risk of single-source dependency. The stability of the intermediates, particularly the hydantoin-substituted triesters, allows for flexible production scheduling and the potential for campaign manufacturing, where intermediates can be stockpiled without significant degradation. This flexibility is crucial for meeting fluctuating demand in the pharmaceutical sector without compromising on lead times. The process avoids the use of highly hazardous reagents that often trigger strict regulatory scrutiny and transportation delays, thereby smoothing the logistics flow and ensuring consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as filtration, crystallization, and standard hydrogenation that are easily transferred from pilot plant to commercial scale. The avoidance of cryogenic temperatures in the critical asymmetric step reduces the energy load on the facility, contributing to a lower carbon footprint. Waste streams are primarily composed of benign organic salts and solvents that can be treated using standard wastewater management protocols, avoiding the generation of persistent organic pollutants. This environmental profile facilitates faster regulatory approvals and supports the sustainability goals of modern pharmaceutical companies seeking to minimize their ecological impact.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this process for their specific pipeline candidates.
Q: How is stereochemical purity controlled in this synthesis?
A: Stereocontrol is achieved through an initial asymmetric catalytic ene reaction yielding ~98% ee, followed by diastereomeric enrichment via crystallization of the pseudoephedrine salt during the decarboxylation step.
Q: What are the key advantages of using the hydantoin auxiliary?
A: The 3-bromomethyl-1,5,5-trimethylhydantoin serves as a robust alkylating agent that facilitates subsequent functionalization while allowing for easy removal or transformation into the final hydroxamic acid motif.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes standard reagents like Pd/C for hydrogenation and avoids cryogenic conditions in later steps, making it highly scalable for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Succinic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the enantiomeric excess and chemical identity of every batch. Our expertise in asymmetric catalysis and complex heterocycle synthesis positions us as a strategic partner capable of delivering the complex chiral succinic acid derivatives required for your most challenging projects.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this optimized process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your drug development timelines while optimizing your manufacturing budget.
