Scalable Synthesis of High-Purity Chiral Succinic Acid Derivatives for Pharmaceutical Applications
Scalable Synthesis of High-Purity Chiral Succinic Acid Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously demands advanced synthetic methodologies capable of delivering complex chiral intermediates with exceptional purity and stereochemical control. Patent CN1068876C discloses a novel and robust process for the preparation of chiral succinic acid derivatives, specifically targeting compounds with valuable pharmacological properties described in prior art such as EP684240A1. This technology represents a significant leap forward in asymmetric synthesis, utilizing a sophisticated titanium-catalyzed ene reaction to establish critical stereocenters with high fidelity. By integrating unique purification protocols involving salt formation and selective deprotection, the disclosed method overcomes historical challenges related to isomer separation and optical purity. For R&D directors and procurement specialists seeking reliable sources of high-value scaffolds, this patent provides a blueprint for manufacturing processes that balance technical excellence with operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing chiral succinic acid frameworks often suffer from significant drawbacks that hinder their viability for commercial-scale API manufacturing. Conventional approaches frequently rely on resolution of racemic mixtures, a strategy that inherently caps the maximum theoretical yield at 50% and generates substantial chemical waste, thereby inflating production costs and environmental burden. Furthermore, many established asymmetric methods necessitate stringent cryogenic conditions, often requiring temperatures below -30°C to maintain acceptable levels of enantioselectivity, which imposes severe energy demands and engineering constraints on reactor design. The reliance on unstable intermediates or difficult-to-remove impurities in older pathways often leads to complex downstream processing, where multiple chromatographic steps are required to achieve the necessary purity profiles for pharmaceutical applications. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and unpredictable availability of critical intermediates for drug development programs.
The Novel Approach
The methodology outlined in CN1068876C introduces a transformative approach that addresses these systemic inefficiencies through a streamlined sequence of catalytic and stoichiometric transformations. Central to this innovation is the use of a hydantoin-based auxiliary which facilitates precise alkylation and subsequent purification via salt formation, effectively bypassing the need for laborious chromatographic separations. The process leverages a glyoxylate-ene reaction catalyzed by (R)-binaphthyloxytitanium complexes, which remarkably achieves high enantiomeric excess (approximately 98%) even at ambient temperatures, drastically reducing energy consumption and operational complexity. 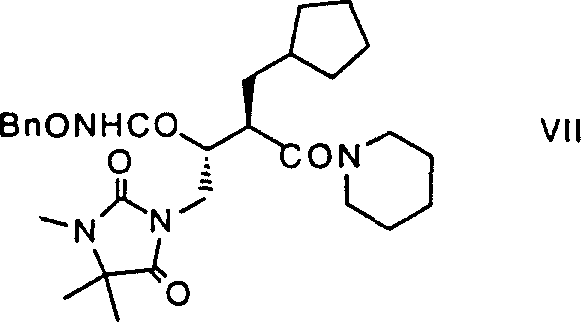 This strategic design allows for the direct conversion of esters to hydroxamic acids using activated O-benzylhydroxylamine, minimizing the number of isolation steps and maximizing overall material throughput. By combining high-yielding reactions with robust purification techniques, this novel approach offers a commercially viable pathway that significantly enhances the reliability and cost-effectiveness of producing these complex chiral building blocks.
This strategic design allows for the direct conversion of esters to hydroxamic acids using activated O-benzylhydroxylamine, minimizing the number of isolation steps and maximizing overall material throughput. By combining high-yielding reactions with robust purification techniques, this novel approach offers a commercially viable pathway that significantly enhances the reliability and cost-effectiveness of producing these complex chiral building blocks.
Mechanistic Insights into Titanium-Catalyzed Asymmetric Ene Reaction
The cornerstone of this synthetic strategy is the enantioselective glyoxylate-ene reaction, which establishes the initial chiral center with remarkable precision using a Lewis acid catalyst system. The mechanism involves the coordination of a glyoxylic acid derivative to a chiral (R)-binaphthyloxytitanium catalyst, creating a rigid chiral environment that directs the approach of the alkene substrate, such as methylenecyclopentane. Unlike traditional methods that struggle with steric hindrance from bulky substituents, this catalytic system tolerates various ester groups while maintaining high stereocontrol, yielding the hydroxyester intermediate with an enantiomeric excess of about 98%. The reaction proceeds efficiently at temperatures ranging from -20°C to 25°C, and surprisingly, room temperature conditions often favor higher selectivity, challenging the conventional wisdom that lower temperatures are always required for asymmetric induction. This mechanistic robustness ensures that the process remains scalable without the need for specialized cryogenic equipment, making it an attractive option for industrial implementation.
Following the establishment of chirality, the process employs a hydantoin-mediated alkylation strategy that further refines the stereochemical outcome and facilitates purification. The intermediate sulfonate, activated by reagents like o-nitrobenzenesulfonyl chloride, undergoes nucleophilic substitution with a malonate derivative in the presence of a strong base such as sodium hydride. 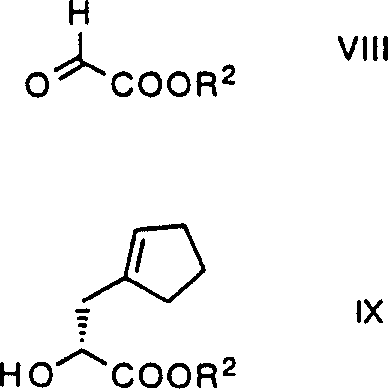 This step is critical as it sets the stage for the subsequent decarboxylation, where the choice of solvent and base, such as N-methylmorpholine in propyl acetate, influences the diastereomeric ratio, achieving a favorable 85:15 ratio of (2R,3R) to (2R,3S) isomers. The resulting acid is then purified not by chromatography, but by forming a crystalline salt with chiral amines like (+)-pseudoephedrine, which selectively precipitates the desired optical isomer. This elegant combination of kinetic control during alkylation and thermodynamic control during salt formation ensures that the final product meets the rigorous purity specifications demanded by the pharmaceutical industry.
This step is critical as it sets the stage for the subsequent decarboxylation, where the choice of solvent and base, such as N-methylmorpholine in propyl acetate, influences the diastereomeric ratio, achieving a favorable 85:15 ratio of (2R,3R) to (2R,3S) isomers. The resulting acid is then purified not by chromatography, but by forming a crystalline salt with chiral amines like (+)-pseudoephedrine, which selectively precipitates the desired optical isomer. This elegant combination of kinetic control during alkylation and thermodynamic control during salt formation ensures that the final product meets the rigorous purity specifications demanded by the pharmaceutical industry.
How to Synthesize Chiral Succinic Acid Derivatives Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction conditions and reagent quality to ensure consistent outcomes across different batches. The process begins with the preparation of the chiral hydroxyester via the titanium-catalyzed ene reaction, followed by activation and alkylation with the hydantoin moiety. Subsequent steps involve hydrogenolysis to remove protecting groups, decarboxylation to simplify the skeleton, and finally, coupling with piperidine and hydroxylamine derivatives to install the pharmacophore. Each transition between steps is designed to minimize isolation losses, often utilizing telescoped procedures where intermediates are carried forward as solutions or crude solids. Detailed standard operating procedures regarding temperature control, stoichiometry, and workup protocols are essential for replicating the high yields and purity reported in the patent examples.
- Perform glyoxylate-ene reaction using (R)-binaphthyloxytitanium catalyst to establish chirality.
- Alkylate the resulting intermediate with 3-bromomethyl-1,5,5-trimethylhydantoin in the presence of base.
- Execute hydrogenolysis and decarboxylation, followed by amidation and hydroxamate formation to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling advantages that directly impact the bottom line and operational resilience. The elimination of cryogenic requirements for the key chiral induction step translates into substantial energy savings and reduces the dependency on specialized low-temperature reactor infrastructure, thereby lowering capital expenditure and utility costs. Furthermore, the ability to purify intermediates through crystallization of salts rather than chromatography significantly reduces solvent consumption and waste generation, aligning with modern green chemistry principles and simplifying regulatory compliance regarding environmental discharge. The robustness of the hydantoin chemistry ensures high reproducibility, minimizing the risk of batch failures and ensuring a steady supply of materials for downstream drug formulation.
- Cost Reduction in Manufacturing: The process achieves cost optimization by replacing expensive resolution steps with a highly selective catalytic asymmetric synthesis that maximizes atom economy. By avoiding the 50% yield loss inherent in racemic resolution and utilizing readily available starting materials like methylenecyclopentane and glyoxylic esters, the overall cost of goods sold is drastically reduced. Additionally, the use of recoverable solvents and the minimization of purification steps contribute to a leaner manufacturing profile that enhances profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available reagents and the avoidance of sensitive organometallic species that require strict inert atmosphere handling enhance the reliability of the supply chain. The process tolerance to ambient temperatures reduces the risk of thermal runaways or equipment failures associated with extreme cooling, ensuring consistent production schedules. This stability allows suppliers to maintain higher inventory levels of key intermediates, reducing lead times for customers and mitigating the risks associated with global logistics disruptions.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated to work efficiently with standard laboratory glassware and adaptable to large-scale industrial reactors without significant modification. The reduction in hazardous waste through efficient salt-based purification and the use of less toxic solvents supports corporate sustainability goals. This environmental compatibility simplifies the permitting process for new manufacturing facilities and ensures long-term operational continuity in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral succinic acid derivatives. Understanding the nuances of the catalytic system and purification strategies is vital for partners looking to integrate these intermediates into their own development pipelines. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does the new process improve enantiomeric purity compared to conventional methods?
A: The process utilizes a (R)-binaphthyloxytitanium catalyst that achieves approximately 98% enantiomeric excess at room temperature, significantly outperforming prior art which often requires cryogenic conditions.
Q: What purification strategies are employed to ensure high chemical purity?
A: The method incorporates salt formation with bases like (+)-pseudoephedrine or ethanolamine, allowing for crystallization of chemically and optically pure intermediates before final coupling.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route avoids extreme cryogenic temperatures for the key chiral step and uses robust reagents like hydantoin derivatives, making it highly amenable to kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Succinic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1068876C can be translated into reliable supply for your projects. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of every batch. We understand that the success of your drug development program depends on the consistency and quality of your raw materials, which is why we adhere to the highest standards of GMP-compliant manufacturing practices. Our technical team is ready to collaborate with you to optimize this route for your specific volume requirements, ensuring a seamless transition from pilot scale to full commercialization.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates how our optimized manufacturing processes can reduce your overall development costs. Let us support your journey from discovery to market with high-quality intermediates that meet the demanding standards of the global pharmaceutical industry.
