Scalable Semi-Synthetic Production Of Astilbin For Global Pharmaceutical And Functional Food Markets
The global demand for bioactive flavonoids has surged in recent years, driven by their potent therapeutic potential in treating immune-related diseases and metabolic disorders. Among these compounds, Astilbin (CAS 29838-67-3) stands out due to its remarkable selective immunosuppressive effects and liver-protective properties, surpassing many general immunosuppressive agents currently on the market. However, traditional sourcing methods relying on direct extraction from plants like Smilax glabra have faced significant bottlenecks, primarily due to the extremely low natural content of Astilbin, typically ranging between 1 and 2 percent in raw biomass. To address these supply chain vulnerabilities and environmental concerns, the industry is shifting towards robust semi-synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN109369749B, which outlines a highly efficient preparation method transforming abundant Myricitrin into high-purity Astilbin. This technical insight report analyzes the mechanistic depth and commercial viability of this route, offering strategic value for R&D directors seeking process optimization and procurement leaders aiming for cost-effective sourcing strategies.
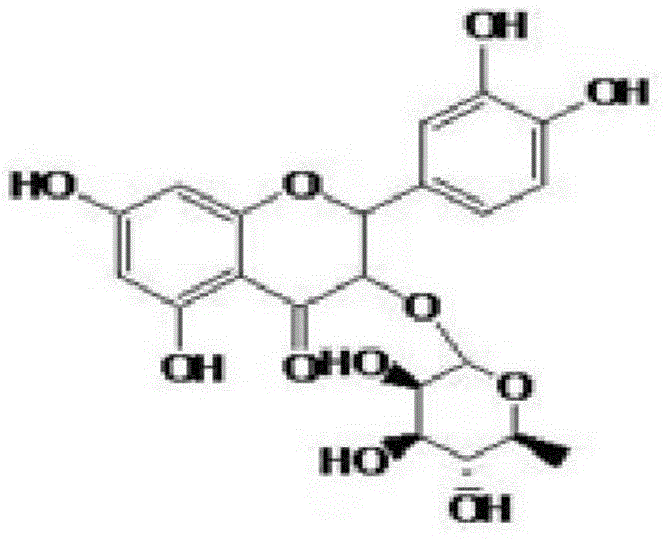
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of Astilbin has been heavily dependent on botanical extraction, a process fraught with inherent inefficiencies that hinder commercial scalability. The primary constraint lies in the scarcity of the target molecule within natural sources; even in rich sources like the rhizome of Smilax glabra, the concentration remains critically low, necessitating the processing of massive quantities of plant material to obtain negligible yields. This low efficiency not only drives up the cost of goods sold (COGS) but also imposes a heavy environmental burden through the excessive consumption of organic solvents and the generation of substantial plant waste. Furthermore, natural extraction is subject to significant variability based on harvest seasons, geographical origins, and plant age, leading to inconsistent batch-to-batch quality that complicates regulatory compliance for pharmaceutical applications. The difficulty in obtaining a product with high yield using extraction methods creates a supply-demand imbalance, limiting the compound's broader application in functional foods and novel drug development pipelines.
The Novel Approach
In stark contrast to the unpredictability of extraction, the semi-synthetic route disclosed in the patent data introduces a controlled, three-step chemical transformation that leverages Myricitrin as a stable and abundant starting material. This innovative approach bypasses the limitations of natural abundance by chemically reconstructing the Astilbin skeleton through hydrogenation, hydrolysis, and catalytic cyclization. By selecting Myricitrin, which is readily available from mature extraction industries involving bayberry and tea, the process secures a reliable feedstock supply that is decoupled from the specific seasonal constraints of Astilbin-rich plants. The methodology simplifies the operational complexity, replacing multi-stage purification of crude plant extracts with defined chemical reactions that can be precisely monitored and optimized. This shift from extraction to synthesis represents a paradigm change in manufacturing, enabling the production of Astilbin with a mass content exceeding 98 percent while drastically reducing the ecological footprint associated with solvent usage and plant resource depletion.
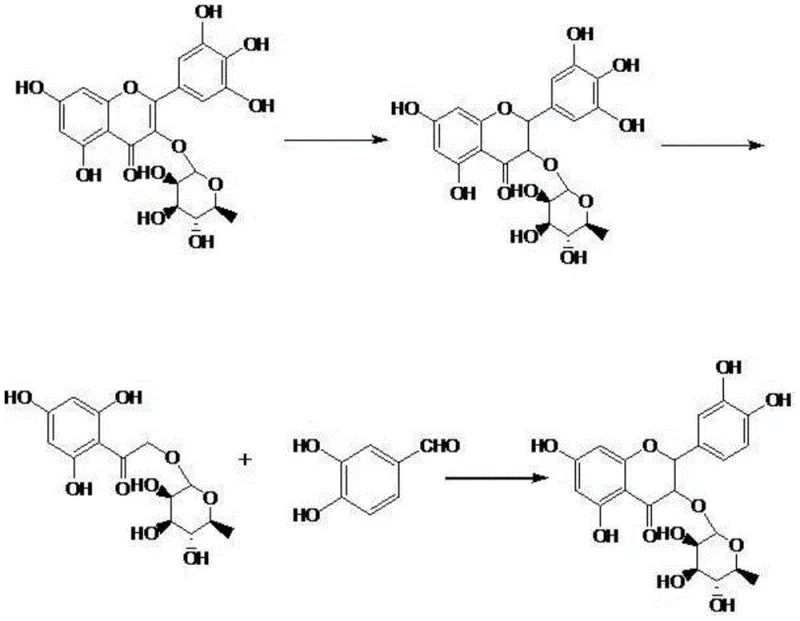
Mechanistic Insights into Proline-Catalyzed Cyclization And Hydrogenation
The core of this synthetic strategy lies in its elegant manipulation of the flavonoid structure, beginning with the selective hydrogenation of the C2-C3 double bond in Myricitrin. Under alkaline conditions using a palladium-carbon catalyst at moderate pressures of 1.5 to 2.5 MPa and temperatures between 25 and 50°C, Myricitrin is converted into Dihydromyricitrin with high fidelity. This step is crucial as it saturates the flavonoid backbone, preparing the molecule for the subsequent ring-opening hydrolysis. The reaction progress is meticulously tracked using High-Performance Liquid Chromatography (HPLC), ensuring that the reaction stops precisely when the product concentration plateaus, thereby preventing over-reduction or degradation. The use of Pd/C is strategic, offering a balance between catalytic activity and ease of removal via filtration, which is essential for maintaining the purity profile required for downstream pharmaceutical applications.
Following hydrogenation, the process employs a base-mediated hydrolysis to cleave the heterocyclic C-ring, generating a key hydroxyacetophenone intermediate. This intermediate serves as the nucleophilic partner in the final, most critical step: the organocatalytic cyclization. In the presence of L-proline, a green and non-toxic organocatalyst, the hydroxyacetophenone intermediate condenses with 3,4-dihydroxybenzaldehyde to reconstruct the flavanonol skeleton of Astilbin. The use of proline is particularly advantageous for commercial manufacturing as it eliminates the need for expensive and potentially toxic transition metal catalysts in the final bond-forming step. The reaction proceeds in polar aprotic solvents like DMF or methanol at temperatures ranging from 60 to 100°C, facilitating the stereo-selective formation of the (2R,3R) configuration. This mechanistic pathway not only ensures high chemical purity but also simplifies the impurity profile, making the subsequent recrystallization steps highly effective in achieving the target specification of greater than 98 percent purity.
How to Synthesize Astilbin Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The process begins with the preparation of dihydromyricitrin, followed by the isolation of the hydroxyacetophenone intermediate, and concludes with the proline-catalyzed cyclization and purification. Each stage demands specific attention to pH adjustment, temperature gradients, and solvent recovery to ensure economic and technical feasibility. For detailed operational protocols, including specific reagent ratios, stirring speeds, and crystallization conditions, please refer to the standardized technical guide provided below.
- Hydrogenation of Myricitrin using Pd/C catalyst under alkaline conditions to form Dihydromyricitrin.
- Alkaline hydrolysis of Dihydromyricitrin followed by acidification to isolate the hydroxyacetophenone intermediate.
- Proline-catalyzed condensation of the intermediate with 3,4-dihydroxybenzaldehyde to cyclize and form crude Astilbin, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction to this semi-synthetic route offers profound strategic advantages that extend beyond mere technical feasibility. The primary benefit is the stabilization of the supply chain through the utilization of Myricitrin, a commodity chemical with a mature and robust global supply network. Unlike wild-harvested Astilbin sources which are vulnerable to ecological changes and regulatory restrictions on plant collection, Myricitrin is derived from cultivated sources like bayberry trees, ensuring long-term availability and price stability. This shift effectively de-risks the procurement process, allowing for more accurate long-term forecasting and contract negotiation. Furthermore, the simplified process flow reduces the number of unit operations required, directly translating to lower capital expenditure (CAPEX) for equipment and reduced operational expenditure (OPEX) related to energy and labor.
- Cost Reduction in Manufacturing: The economic model of this synthesis is fundamentally superior to extraction due to the elimination of massive solvent volumes and the reduction in raw material throughput required per kilogram of product. By avoiding the processing of tons of plant biomass to extract trace amounts of active ingredient, manufacturers can achieve substantial cost savings in waste treatment and raw material acquisition. Additionally, the use of organocatalysts like proline in the final step removes the necessity for expensive heavy metal scavenging processes, further streamlining the cost structure. These efficiencies collectively contribute to a significantly reduced cost of goods, making high-purity Astilbin commercially viable for broader market applications including nutraceuticals and generic pharmaceuticals.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for multinational corporations, and this synthetic route offers a distinct advantage by decoupling production from agricultural seasonality. Since the starting materials are stable chemicals that can be stockpiled without degradation, production schedules can be maintained year-round regardless of harvest cycles. This reliability minimizes the risk of stockouts and allows for just-in-time manufacturing strategies that reduce inventory holding costs. The robustness of the chemical process also means that scale-up from pilot to commercial production is predictable, ensuring that supply commitments can be met consistently even as market demand fluctuates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles and stringent regulatory standards. The process generates significantly less solid waste compared to plant extraction, reducing the burden on waste management systems and lowering disposal costs. The ability to recover and recycle solvents like DMF and methanol further enhances the environmental profile, supporting corporate sustainability goals. Moreover, the absence of complex botanical matrices simplifies the purification process, reducing the risk of pesticide or heavy metal contamination often associated with herbal extracts, thereby facilitating smoother regulatory filings and market access.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for integration into their existing portfolios, we have compiled answers to common inquiries regarding the process specifics and quality control measures. These responses are derived directly from the experimental data and beneficial effects reported in the patent literature, ensuring accuracy and relevance for industrial application. Understanding these details is crucial for assessing the feasibility of technology transfer and the alignment with internal quality standards.
Q: What are the primary advantages of this semi-synthetic route over plant extraction?
A: The semi-synthetic method utilizing Myricitrin as a starting material offers significantly higher yields and better process control compared to direct plant extraction, which suffers from low natural content and high solvent waste. This approach ensures a more consistent supply of high-purity Astilbin suitable for industrial scale-up.
Q: How is the purity of the final Astilbin product controlled in this process?
A: The process employs rigorous monitoring via High-Performance Liquid Chromatography (HPLC) at each reaction stage to ensure endpoint accuracy. The final refining step involves recrystallization from purified water, consistently achieving a mass content of greater than 98 percent, meeting stringent pharmaceutical standards.
Q: Is the starting material Myricitrin readily available for large-scale production?
A: Yes, Myricitrin is a mature natural extract found abundantly in plants such as bayberry leaves and tea, ensuring a stable and cost-effective raw material supply chain. This availability mitigates the risks associated with seasonal fluctuations often seen in direct botanical extraction of rare flavonoids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Astilbin Supplier
The technical sophistication of this semi-synthetic route underscores the need for a manufacturing partner with deep expertise in complex organic synthesis and process scale-up. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling high-pressure hydrogenation and precise temperature-controlled cyclization, ensuring that the stringent purity specifications required for pharmaceutical intermediates are consistently met. With our rigorous QC labs and commitment to process safety, we provide a secure foundation for your supply chain, transforming patented laboratory methods into reliable industrial realities.
We invite you to collaborate with us to optimize your sourcing strategy for Astilbin and related flavonoid intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your product development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
