Advanced Enzymatic Synthesis of Vitamin D Precursors for Commercial Scale-Up and High Purity
Advanced Enzymatic Synthesis of Vitamin D Precursors for Commercial Scale-Up and High Purity
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for the synthesis of high-value active pharmaceutical ingredients (APIs) and their key intermediates. Patent CN1425019A represents a significant technological breakthrough in the field of steroid chemistry, specifically detailing a novel preparation method for Vitamin D precursors of formula (I). This patent discloses a sophisticated enzymatic asymmetrisation strategy starting from readily available 3,5-dihydroxybenzoic acid derivatives, effectively bypassing the limitations of traditional chiral pool syntheses. The core innovation lies in the construction of the dicyclo[3.1.0]hexane skeleton, which serves as the critical A-ring precursor for 19-nor-vitamin D analogues. By leveraging lipase-catalyzed kinetic resolution, this methodology achieves exceptional stereocontrol while utilizing commodity chemicals, thereby addressing critical supply chain vulnerabilities associated with scarce natural starting materials. 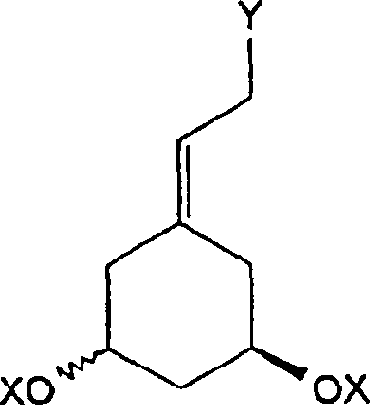
For R&D directors and process chemists, the structural versatility offered by this patent is paramount. The general formula (I) encompasses a wide range of substituents at positions A, R, R1, and R2, allowing for the tailored synthesis of diverse Vitamin D metabolites and analogues. The ability to introduce functional groups such as hydroxymethyl, formyl, or ethynyl at the C-3a position provides a flexible platform for downstream coupling reactions with CD-ring fragments. This modularity is essential for developing next-generation therapeutics for osteoporosis, psoriasis, and hyperparathyroidism, where subtle structural modifications can drastically alter biological activity and pharmacokinetic profiles. The patent further elucidates the preparation of key intermediates of formula (II), ensuring that the entire value chain from bulk chemical to advanced intermediate is covered by this intellectual property.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Vitamin D A-ring has relied heavily on the degradation of naturally occurring chiral pools, such as (-)-quinic acid or phloroglucitol. While these methods established the foundational chemistry for Vitamin D production, they suffer from severe drawbacks that hinder modern commercial manufacturing. The route originating from (-)-quinic acid, for instance, necessitates the use of deleterious tri-n-butyltin hydride for deoxygenation steps, posing significant environmental and safety hazards that are increasingly unacceptable in green chemistry frameworks. Furthermore, the pathway from phloroglucitol is excessively lengthy, often requiring up to 12 synthetic steps and two distinct enzymatic reactions, which cumulatively erode overall yield and increase production costs. Another historical approach utilizing 3-cyclopentenol is fundamentally flawed due to the commercial unavailability of the raw material, which itself must be synthesized via low-yield hydrogen-boronation reactions, creating a bottleneck at the very beginning of the supply chain.
The Novel Approach
In stark contrast, the methodology described in CN1425019A utilizes hydrogenated 3,5-dihydroxybenzoic acid derivatives as the starting point, which are inexpensive, commercially abundant, and chemically stable. This novel approach streamlines the synthesis by employing a highly efficient enzymatic asymmetrisation as the key stereo-defining step. Instead of relying on stoichiometric chiral auxiliaries or resolution via diastereomeric salt formation, the process uses lipases to kinetically resolve the meso- or prochiral diols with high enantioselectivity. This shift not only reduces the number of steps but also ensures that the intermediates obtained are predominantly crystalline solids. The ability to purify intermediates via crystallization rather than silica gel column chromatography is a game-changer for industrial scale-up, as it eliminates the solvent-intensive and laborious purification processes that typically limit batch sizes in fine chemical manufacturing. 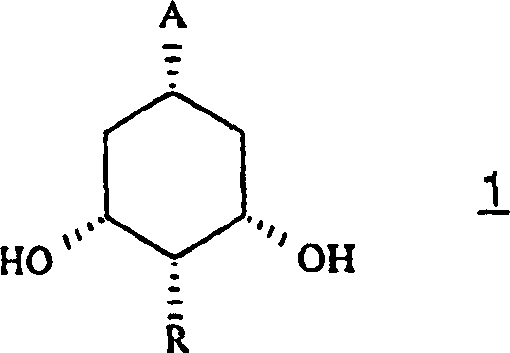
Mechanistic Insights into Lipase-Catalyzed Asymmetrisation
The heart of this synthetic strategy is the enzymatic kinetic resolution of cis,cis-3,5-dihydroxy-cyclohexanecarboxylates. The patent details the use of various lipases, such as SAM II (from Pseudomonas fluorescens), CCL (from Candida cylindracea), and PPL (from Porcine Pancreas Lipase), to achieve regioselective acylation or hydrolysis. In a typical reaction, the symmetric diol substrate is exposed to a vinyl ester (e.g., vinyl acetate) in the presence of the enzyme. The lipase selectively acylates one of the hydroxyl groups, breaking the symmetry of the molecule and establishing the desired (1S, 3S, 5R) or (1R, 3S, 5R) configuration with high optical purity. This biocatalytic step operates under mild conditions, typically between 10°C and 40°C, which preserves the integrity of sensitive functional groups and prevents thermal degradation. The mechanism relies on the precise fit of the substrate into the enzyme's active site, where steric and electronic factors dictate which hydroxyl group is accessible for nucleophilic attack on the acyl-enzyme intermediate.
Following the enzymatic resolution, the synthetic route proceeds through a series of classical organic transformations designed to construct the strained cyclopropane ring fused to the cyclohexane system. The mono-acylated intermediate undergoes selective protection of the remaining free hydroxyl group, often using silyl protecting groups like TBDMS or TBDPS to ensure orthogonality. Subsequently, the acyl group is removed or transformed, and the remaining hydroxyl is activated as a leaving group, such as a tosylate or mesylate. The crucial cyclization step involves treating this activated intermediate with a strong base, such as potassium tert-butoxide, which induces an intramolecular SN2 displacement. This reaction closes the three-membered ring to form the dicyclo[3.1.0]hexane core with inversion of configuration at the reaction center, locking in the stereochemistry required for the final Vitamin D analogue. 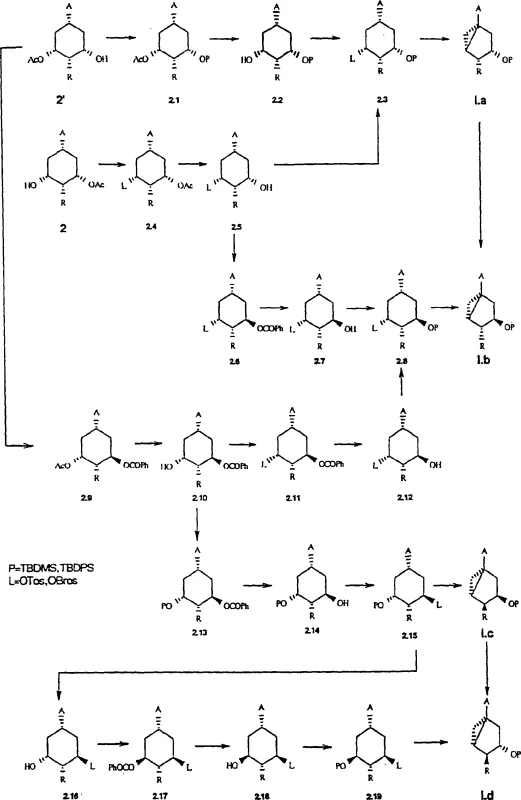
How to Synthesize Dicyclo[3.1.0]hexane Vitamin D Precursors Efficiently
The practical implementation of this synthesis requires careful control of reaction parameters to maximize yield and enantiomeric excess. The process begins with the catalytic hydrogenation of methyl 3,5-dihydroxybenzoate using rhodium on alumina under high pressure to ensure complete saturation of the aromatic ring while maintaining the cis,cis-stereochemistry of the resulting cyclohexane diol. Following isolation, the diol is subjected to the lipase-catalyzed resolution in a non-aqueous medium to drive the equilibrium towards esterification. The resulting mono-acetate is then separated from the unreacted diol or di-acetate, often simply by crystallization or flash chromatography on a pilot scale. Subsequent steps involve standard protection group chemistry, where the choice of silyl ether (TBDMS vs. TBDPS) is dictated by the stability required for downstream processing. The final cyclization is exothermic and must be managed to prevent side reactions, yielding the target bicyclic precursor in high purity.
- Hydrogenation of 3,5-dihydroxybenzoic acid derivatives to obtain cis,cis-3,5-dihydroxy-cyclohexanecarboxylates.
- Enzymatic kinetic resolution using lipases (e.g., SAM II, PPL) in vinyl esters to achieve regioselective acylation and chirality.
- Protection of hydroxyl groups, activation of the remaining hydroxyl as a leaving group (tosylate/mesylate), and base-induced intramolecular cyclization to form the dicyclo[3.1.0]hexane core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic route offers tangible benefits in terms of cost stability and supply security. The reliance on 3,5-dihydroxybenzoic acid derivatives decouples the production of Vitamin D precursors from the fluctuating markets of natural extracts like quinic acid, which are subject to agricultural variability and geopolitical supply risks. By utilizing petrochemical-derived or fermentation-derived aromatic acids, manufacturers can secure long-term contracts for raw materials at predictable price points. Furthermore, the elimination of toxic tin reagents and the reduction in solvent usage for purification align with increasingly stringent environmental regulations, reducing the operational costs associated with waste disposal and regulatory compliance. This process optimization translates directly into a more resilient supply chain capable of withstanding market shocks.
- Cost Reduction in Manufacturing: The economic advantage of this method is driven primarily by the efficiency of the enzymatic step and the physical properties of the intermediates. Because the key intermediates are crystalline, they can be purified to high standards using recrystallization, a significantly cheaper unit operation than preparative HPLC or extensive column chromatography. This reduction in purification complexity lowers the consumption of silica gel and organic solvents, directly reducing the cost of goods sold (COGS). Additionally, the high stereoselectivity of the lipase minimizes the formation of unwanted diastereomers, thereby improving the overall mass balance and reducing the loss of valuable material during separation. These factors combine to create a manufacturing process that is inherently more cost-effective than legacy routes.
- Enhanced Supply Chain Reliability: Supply continuity is critical for pharmaceutical manufacturers, and this synthetic route enhances reliability by simplifying the raw material portfolio. The starting materials are commodity chemicals available from multiple global suppliers, eliminating single-source dependencies. Moreover, the robustness of the enzymatic reaction allows for flexible manufacturing; the biocatalyst can be immobilized or used in batch processes that are easily scaled from kilograms to tons without significant re-optimization. This scalability ensures that production capacity can be ramped up quickly to meet surges in demand for Vitamin D therapies, providing a strategic advantage in a competitive market where lead times are often a deciding factor for partnerships.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant improvement over traditional methods. The avoidance of tri-n-butyltin hydride removes a major hazardous waste stream, simplifying effluent treatment and reducing the risk of heavy metal contamination in the final API. The use of enzymes, which are biodegradable and operate under mild conditions, reduces the energy footprint of the synthesis. Furthermore, the ability to isolate intermediates as solids minimizes the volume of liquid waste generated during workups. These attributes facilitate easier regulatory approval and support sustainability goals, making the manufacturing site more attractive to eco-conscious stakeholders and investors who prioritize green chemistry principles in their supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is crucial for evaluating the feasibility of adopting this route for large-scale production. The answers are derived directly from the experimental data and claims within the patent specification, ensuring accuracy and relevance for technical decision-makers.
Q: How does the enzymatic route improve purity compared to traditional quinic acid methods?
A: The enzymatic route utilizes highly selective lipases to resolve racemic mixtures, avoiding the toxic tin hydride reagents and complex purification steps associated with quinic acid degradation, resulting in crystalline intermediates that are easily purified.
Q: What are the scalability advantages of using 3,5-dihydroxybenzoic acid derivatives?
A: Unlike volatile low-molecular-weight intermediates in previous methods, the intermediates derived from resorcylic acid are predominantly crystalline solids, allowing for purification via crystallization rather than difficult column chromatography, which is critical for ton-scale production.
Q: Can this process access different diastereomers required for various Vitamin D analogues?
A: Yes, by varying the sequence of protection, activation, and cyclization steps, as well as selecting specific lipases for kinetic resolution, the process can selectively produce both 3aS and 3aR configurations essential for different 19-nor and 1-alpha-hydroxy vitamin D analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin D Precursor Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our facilities are equipped with state-of-the-art reactors capable of handling sensitive enzymatic reactions and cryogenic conditions required for the synthesis of complex chiral intermediates like those described in CN1425019A. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Vitamin D precursor meets the highest international standards, providing you with the confidence needed to advance your clinical programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation Vitamin D analogues. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and cost-efficient supply chain solution that empowers your R&D efforts and accelerates your time to market.
