Advanced Biocatalytic Synthesis of Sitagliptin Intermediates Using Novel Recombinant (R)-ω-Transaminase Mutants
The pharmaceutical industry continuously seeks robust and efficient pathways for the production of critical antidiabetic agents, particularly Sitagliptin, a potent DPP-4 inhibitor. Patent CN111411095B introduces a groundbreaking advancement in this domain by disclosing a novel recombinant (R)-ω-transaminase and its specific mutants designed for the asymmetric synthesis of Sitagliptin intermediates. This technology represents a significant leap forward in biocatalytic engineering, addressing the longstanding challenges of low enzymatic activity and insufficient stereoselectivity that have plagued earlier generations of transaminases. The patent details a sophisticated approach involving gene recombination and site-directed mutagenesis to create an enzyme variant that not only possesses high enzymatic activity reaching 1245.2 U/g but also maintains an exceptional enantiomeric excess (e.e.) value of greater than 99.9% for the R-configuration. For a reliable pharmaceutical intermediate supplier, adopting such high-performance biocatalysts is crucial for meeting the stringent purity requirements of global regulatory bodies while optimizing production throughput. The ability to efficiently catalyze the conversion of precursor ketone analogues into the desired chiral amine intermediate with a conversion rate as high as 97.4% underscores the commercial viability and technical superiority of this invention over conventional chemical synthesis methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amines like the Sitagliptin intermediate has relied heavily on either traditional chemical resolution or early-generation biocatalysts that required extensive engineering to become viable. For instance, previous efforts by industry leaders involved modifying wild-type transaminases through multiple rounds of directed evolution, sometimes requiring up to eleven iterations of molecular modification to achieve acceptable activity levels. These conventional methods often suffered from poor substrate tolerance, necessitating low substrate concentrations that resulted in large reactor volumes and high solvent consumption, thereby driving up manufacturing costs. Furthermore, early transaminases frequently exhibited inadequate stereoselectivity, leading to the formation of racemic mixtures that required costly and time-consuming separation steps to isolate the therapeutically active R-enantiomer. The reliance on such inefficient catalysts created bottlenecks in the supply chain, making it difficult to achieve cost reduction in API manufacturing at a commercial scale. Additionally, the instability of some wild-type enzymes under industrial conditions often led to inconsistent batch-to-batch performance, posing significant risks to supply continuity and quality control.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111411095B utilizes a strategic combination of gene recombination and precise site-directed mutagenesis to construct a superior biocatalyst. By recombining specific N-terminal and C-terminal regions from different transaminase sources (such as AtTA6, CeTA5, and ShTA), the inventors created a chimeric enzyme scaffold that inherently possesses better structural stability and substrate affinity. This novel approach was further refined through single-point and double-point mutations at critical amino acid positions, specifically the 72nd and 248th sites, which dramatically expanded the substrate binding pocket. The result is a mutant enzyme capable of operating efficiently at high substrate concentrations up to 1200 mM, significantly reducing the reaction volume and solvent load. 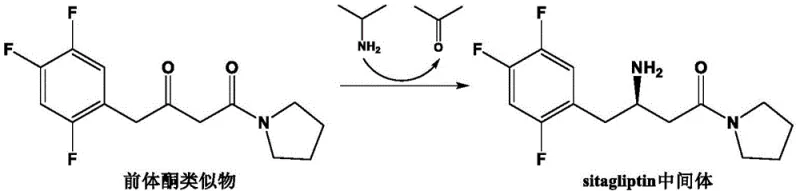 As illustrated in the reaction scheme, the process utilizes isopropylamine as an amino donor to convert the precursor ketone analogue directly into the Sitagliptin intermediate with high fidelity. This streamlined biocatalytic route eliminates the need for harsh chemical reagents and complex protection-deprotection steps, offering a greener and more economically attractive pathway for the commercial scale-up of complex chiral amines.
As illustrated in the reaction scheme, the process utilizes isopropylamine as an amino donor to convert the precursor ketone analogue directly into the Sitagliptin intermediate with high fidelity. This streamlined biocatalytic route eliminates the need for harsh chemical reagents and complex protection-deprotection steps, offering a greener and more economically attractive pathway for the commercial scale-up of complex chiral amines.
Mechanistic Insights into Recombinant (R)-ω-Transaminase Catalysis
The core of this technological breakthrough lies in the intricate mechanistic function of the (R)-ω-transaminase, a pyridoxal 5'-phosphate (PLP) dependent enzyme that facilitates the transfer of amino groups with high precision. During the catalytic cycle, the enzyme alternates between its PLP-bound form and its pyridoxamine 5'-phosphate (PMP) form, enabling the reversible transfer of an amino group from the donor (isopropylamine) to the carbonyl acceptor (the precursor ketone). The novel recombinant enzyme described in the patent has been structurally optimized to stabilize the transition state of this reaction, thereby lowering the activation energy and accelerating the reaction rate. The specific mutations introduced at positions 72 and 248 play a pivotal role in reshaping the active site geometry, allowing bulky substrate molecules like the trifluorophenyl-containing ketone to bind more effectively without steric hindrance.  This enhanced binding affinity is critical for maintaining high catalytic turnover numbers even when the substrate concentration is increased to industrially relevant levels. Furthermore, the enzyme's rigid active site architecture ensures that the protonation and deprotonation steps occur in a highly stereo-specific manner, locking the product into the desired R-configuration and preventing the formation of the S-enantiomer. This level of mechanistic control is essential for producing high-purity pharmaceutical intermediates that meet the rigorous specifications required for final drug substance manufacturing.
This enhanced binding affinity is critical for maintaining high catalytic turnover numbers even when the substrate concentration is increased to industrially relevant levels. Furthermore, the enzyme's rigid active site architecture ensures that the protonation and deprotonation steps occur in a highly stereo-specific manner, locking the product into the desired R-configuration and preventing the formation of the S-enantiomer. This level of mechanistic control is essential for producing high-purity pharmaceutical intermediates that meet the rigorous specifications required for final drug substance manufacturing.
From an impurity control perspective, the high stereoselectivity of this mutant enzyme serves as a powerful tool for minimizing downstream processing burdens. In traditional chemical synthesis, the generation of diastereomers and enantiomeric impurities often requires multiple crystallization steps or chiral chromatography, which are both yield-loss intensive and expensive. By achieving an e.e. value of >99.9% directly in the biotransformation step, the novel transaminase effectively acts as a 'biological filter' that excludes incorrect stereoisomers at the source. This intrinsic purity advantage significantly simplifies the workup procedure, as the reaction mixture contains predominantly the desired product along with benign byproducts like acetone (from the isopropylamine donor). The ability to operate at a pH of 8-9 in a triethanolamine-HCl buffer further contributes to product stability, preventing racemization or degradation of the sensitive beta-amino ketone structure during the reaction. Consequently, the overall impurity profile of the crude product is markedly cleaner, facilitating easier isolation and higher final yields.
How to Synthesize Sitagliptin Intermediate Efficiently
The implementation of this biocatalytic process involves a series of well-defined steps that leverage the high activity of the recombinant E. coli strains. The synthesis begins with the preparation of the biocatalyst, where the genetically engineered bacteria expressing the mutant transaminase are cultured and induced to produce the enzyme intracellularly. Following cell disruption, the whole-cell catalyst or the crude enzyme extract is utilized in a buffered aqueous system containing the precursor ketone analogue and the amino donor. The reaction conditions are carefully optimized to balance enzyme stability with reaction kinetics, typically maintaining a temperature of 50 ℃ and a pH of 9.0 to maximize conversion efficiency. Detailed standardized synthetic steps see the guide below.
- Construct the recombinant vector by cloning the novel (R)-ω-transaminase gene or its mutant (specifically D72G and A248S variants) into an expression plasmid such as pET28b.
- Transform the recombinant vector into E. coli BL21(DE3) competent cells and induce expression using IPTG at 28 ℃ to produce the active biocatalyst.
- Perform the biocatalytic reaction using the whole cell catalyst with the precursor ketone analogue, isopropylamine as the amino donor, and PLP as a coenzyme in a triethanolamine-HCl buffer at 50 ℃.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel recombinant transaminase technology translates into tangible strategic benefits that extend beyond mere technical performance. The primary advantage lies in the substantial cost savings achieved through process intensification; because the mutant enzyme can tolerate substrate concentrations as high as 1000 mM to 1200 mM, manufacturers can significantly reduce the volume of solvents and water required per kilogram of product. This reduction in volumetric throughput allows existing fermentation and reaction infrastructure to produce more product in less time, effectively increasing asset utilization without the need for capital-intensive facility expansions. Moreover, the elimination of transition metal catalysts and harsh chemical reagents associated with traditional synthetic routes simplifies waste management and reduces the environmental footprint, aligning with increasingly strict global sustainability mandates.
- Cost Reduction in Manufacturing: The high enzymatic activity of 1245.2 U/g means that significantly less biocatalyst is required to drive the reaction to completion, directly lowering the cost of goods sold (COGS). By avoiding the use of expensive chiral ligands or precious metal catalysts often needed in asymmetric hydrogenation, the process achieves a leaner cost structure. The high conversion rate of 97.4% minimizes the loss of valuable starting materials, ensuring that raw material costs are optimized and waste disposal costs are kept to a minimum. This efficiency creates a buffer against fluctuations in raw material pricing, providing greater financial predictability for long-term supply contracts.
- Enhanced Supply Chain Reliability: The robustness of the E. coli expression system ensures a consistent and scalable supply of the biocatalyst, mitigating the risk of production delays caused by enzyme instability. The ability to achieve high yields in a single step reduces the number of unit operations required, thereby shortening the overall manufacturing lead time and accelerating time-to-market for new drug filings. This streamlined process enhances the agility of the supply chain, allowing manufacturers to respond more rapidly to changes in market demand for Sitagliptin and related DPP-4 inhibitors. Furthermore, the use of a biological system reduces dependence on petrochemical-derived reagents, diversifying the supply base and reducing exposure to volatility in the fossil fuel sector.
- Scalability and Environmental Compliance: The process operates under mild aqueous conditions, which inherently reduces the generation of hazardous organic waste streams and lowers the energy consumption associated with heating and cooling large reactor volumes. The high substrate tolerance allows for concentrated reactions, which drastically reduces the size of downstream separation equipment such as evaporators and dryers, leading to lower utility costs. This green chemistry profile facilitates easier regulatory approval and compliance with environmental regulations in key markets like Europe and North America. The scalability of the fermentation process ensures that production can be seamlessly ramped up from pilot scale to multi-ton commercial production without encountering the mass transfer limitations often seen in heterogeneous chemical catalysis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recombinant transaminase technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and performance metrics. Understanding these details is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing workflows. The data confirms that the mutant strains offer a distinct advantage over prior art in terms of both activity and selectivity.
Q: What is the specific activity improvement of the novel mutant compared to wild-type enzymes?
A: The novel recombinant (R)-ω-transaminase mutant described in patent CN111411095B exhibits a significantly enhanced enzyme activity of up to 1245.2 U/g, which is substantially higher than the initial screened enzymes and previously reported variants, allowing for much faster reaction kinetics.
Q: How does this technology address the issue of stereoselectivity in sitagliptin synthesis?
A: The engineered mutant demonstrates exceptional stereoselectivity with an e.e. value exceeding 99.9% for the R-configuration, effectively eliminating the formation of unwanted S-enantiomer impurities and simplifying downstream purification processes.
Q: Can this biocatalytic process handle high substrate concentrations for industrial scaling?
A: Yes, the mutant strain is capable of tolerating substrate concentrations up to 1200 mM, achieving a conversion rate of 97.4% even at 1000 mM, which indicates strong potential for high-density manufacturing and reduced solvent usage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the novel recombinant (R)-ω-transaminase technology disclosed in CN111411095B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-activity enzyme are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical e.e. value of >99.9% required for the Sitagliptin intermediate. We are committed to leveraging our expertise in protein engineering and bioprocess optimization to deliver a consistent, high-quality supply of this key building block to our global partners.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to explore the full economic and technical advantages of this biocatalytic route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential reductions in solvent usage and raw material costs specific to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this advanced enzymatic technology into your supply chain for reduced lead time for high-purity pharmaceutical intermediates.
