Advanced Two-Step Synthesis of Aripiprazole: Enhancing Purity and Commercial Scalability
Advanced Two-Step Synthesis of Aripiprazole: Enhancing Purity and Commercial Scalability
The pharmaceutical industry constantly seeks robust manufacturing pathways for critical antipsychotic agents like Aripiprazole, a next-generation atypical antipsychotic developed by Otsuka Pharmaceutical. Patent CN101781246A introduces a transformative approach to synthesizing this complex molecule, addressing longstanding challenges in yield optimization and impurity control. The disclosed method streamlines production into two primary stages: a highly selective etherification reaction followed by a refined condensation process. By leveraging specific solvent systems and stoichiometric controls, this technology achieves a total yield exceeding 83% with final purity levels surpassing 99%, eliminating the need for energy-intensive recrystallization steps often required in conventional routes. For R&D directors and procurement specialists, this represents a significant leap forward in process chemistry, offering a reliable API intermediate supplier pathway that balances technical excellence with economic viability. The structural integrity of the final product, as depicted below, is maintained through rigorous control of reaction parameters.
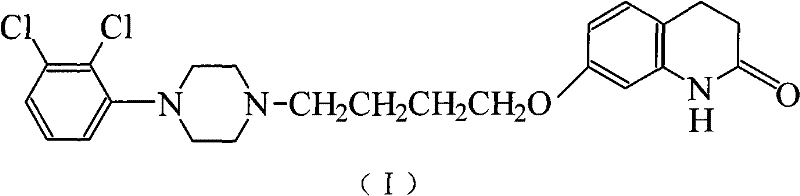
This innovation is particularly relevant for large-scale manufacturing where consistency and cost-efficiency are paramount. The patent details a mechanism where the use of non-polar solvents in the workup phase not only isolates the intermediate effectively but also facilitates the recycling of expensive alkylating agents. Such circular economy principles within the synthesis workflow are essential for modern green chemistry initiatives. Furthermore, the elimination of complex purification sequences reduces the operational footprint, making this method highly attractive for commercial scale-up of complex pharmaceutical intermediates. As we delve deeper into the technical specifics, it becomes clear that this methodology offers a competitive edge in the global supply chain for mental health therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Aripiprazole has been plagued by inefficiencies that hinder large-scale production capabilities. Traditional routes often suffer from low total yields, typically hovering between 60% and 70%, which is economically unsustainable for high-volume API manufacturing. A major technical bottleneck in these legacy processes is the propensity for di-substitution side reactions during the initial alkylation step. When the hydroxy-quinolinone precursor reacts with dihaloalkanes, controlling mono-substitution is difficult, leading to impurities that are structurally similar to the desired product and notoriously difficult to separate via standard chromatography or crystallization. Additionally, prior art methods, such as those described in earlier patents, frequently rely on water-only solvent systems for condensation. While these may offer decent single-step yields, they necessitate cumbersome post-reaction treatments including reflux, distillation, cooling, washing, and vacuum drying to achieve acceptable purity. These multi-step purification protocols increase energy consumption, extend cycle times, and introduce opportunities for product degradation, thereby compromising the overall quality and cost-effectiveness of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the improved method outlined in CN101781246A revolutionizes the synthetic landscape by introducing a strategic two-step protocol that prioritizes both selectivity and simplicity. The core innovation lies in the etherification stage, where a substantial molar excess of 1,4-dihalobutane (at least 6 equivalents) is employed to kinetically drive the reaction towards the desired mono-ether intermediate while suppressing di-substituted byproducts. Following the reaction, the introduction of a non-polar solvent, such as petroleum ether, induces the precipitation of the Aripiprazole etherate, allowing for easy filtration and simultaneous recovery of the unreacted halide. The subsequent condensation step utilizes a novel azeotropic ketone-water mixture, specifically butanone and water, which creates a homogeneous reaction environment. This solvent system enhances molecular contact between the etherate and the piperazine derivative in the presence of alkali metal iodides, driving the reaction to near-completion with yields approaching 98%. The result is a streamlined process where the final product crystallizes directly upon cooling and water addition, bypassing the need for recrystallization and delivering high-purity material suitable for immediate downstream processing.
Mechanistic Insights into Etherification and Azeotropic Condensation
The success of this synthetic route is underpinned by a sophisticated understanding of reaction kinetics and solubility dynamics. In the initial etherification phase, the use of excess 1,4-dihalobutane acts as both a reactant and a selectivity controller. By flooding the reaction milieu with the alkylating agent, the probability of the quinolinone oxygen attacking a second halide molecule is statistically minimized, ensuring that the primary product is the mono-substituted ether. The subsequent addition of a non-polar solvent exploits the differential solubility of the reaction components; the desired etherate precipitates out of the ethanolic solution, while the excess dihalobutane remains dissolved in the filtrate. This physical separation is crucial, as it allows for the distillation and recovery of the valuable alkylating agent, which can be reused in subsequent batches with a recovery rate of approximately 95%. This mechanism not only boosts atom economy but also drastically reduces the raw material cost per kilogram of the intermediate produced.
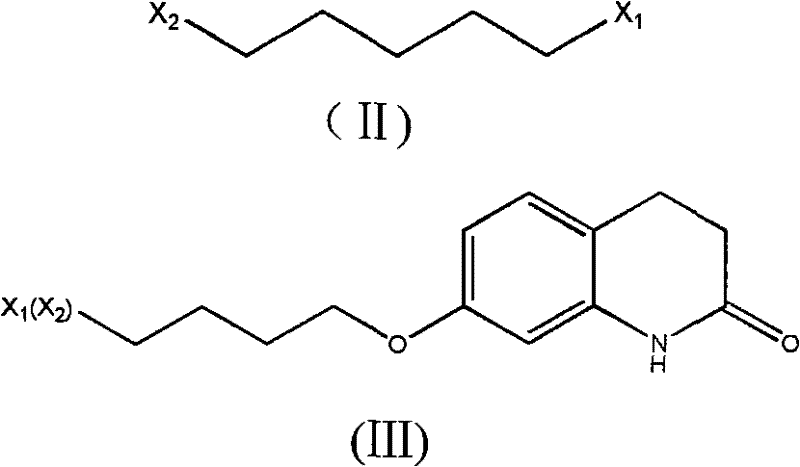
The condensation mechanism further exemplifies process intensification through solvent engineering. The use of an azeotropic mixture of butanone and water creates a unique polarity environment that solubilizes both the organic etherate and the inorganic bases required for the reaction. The presence of alkali metal iodides, such as sodium iodide or potassium iodide, serves as a potent catalyst, likely facilitating the nucleophilic substitution by enhancing the leaving group ability of the halide on the butyl chain. This homogeneous system ensures that mass transfer limitations are virtually eliminated, allowing the reaction to proceed rapidly under reflux conditions. Upon completion, the simple addition of water disrupts the solubility equilibrium, causing the highly pure Aripiprazole to crystallize out of the solution. This "crash out" crystallization technique is highly effective at excluding impurities, resulting in a final product with HPLC purity greater than 99% without the need for further purification steps, demonstrating a masterful control over the solid-state properties of the molecule.
How to Synthesize Aripiprazole Efficiently
The practical implementation of this improved synthesis method offers a clear roadmap for laboratory and pilot-scale operations aiming to optimize Aripiprazole production. The process begins with the preparation of the key ether intermediate, followed by the coupling reaction that forms the final pharmacophore. Detailed operational parameters, including specific temperature ranges, reflux times, and stoichiometric ratios, are critical to replicating the high yields reported in the patent data. Operators must pay close attention to the solvent exchange steps and the precise control of water content during the crystallization phase to ensure optimal particle size and purity profiles. The following guide outlines the standardized synthesis steps derived from the patent embodiments, providing a foundational protocol for technical teams looking to adopt this superior manufacturing route.
- Perform etherification of 7-hydroxy-3,4-dihydro-quinolone with excess 1,4-dihalobutane in ethanol, followed by precipitation in non-polar solvent to isolate the etherate intermediate.
- Conduct condensation reaction between the isolated etherate and 1-(2,3-dichlorophenyl)piperazine using butanone-water azeotrope with sodium iodide catalyst.
- Complete the process by adding water to the reaction mixture, cooling for crystallization, and filtering to obtain high-purity Aripiprazole without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this improved synthesis method translates into tangible strategic benefits that extend beyond mere technical metrics. The primary advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the need for recrystallization and complex distillation sequences, the manufacturing cycle time is significantly reduced, allowing for faster throughput and increased facility utilization rates. This efficiency gain directly impacts the cost of goods sold (COGS), making the final API more competitive in the global marketplace. Furthermore, the ability to recover and recycle high-value reagents like 1,4-dihalobutane creates a closed-loop system that insulates the production process from volatile raw material price fluctuations. This stability is crucial for long-term supply contracts and ensures consistent availability of the drug substance for downstream formulation.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the high recovery rates of key starting materials. The patent explicitly demonstrates that unreacted 1,4-dihalobutane can be recovered from the filtrate with an efficiency of approximately 95% and directly reused in subsequent batches. This circular usage pattern means that the effective consumption of this expensive alkylating agent is minimal, leading to substantial cost savings over the lifecycle of the production campaign. Additionally, the removal of the recrystallization step saves significant amounts of energy and solvent, further driving down operational expenditures. The high yield of the condensation step, reaching nearly 98%, ensures that very little material is lost to waste streams, maximizing the return on investment for every kilogram of precursor purchased.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the reaction conditions. The use of common, commercially available solvents like ethanol, petroleum ether, and butanone reduces dependency on specialized or hazardous reagents that might face supply disruptions. The process tolerance is high, as evidenced by the consistent results across multiple embodiments, which minimizes the risk of batch failures that could delay shipments. Moreover, the shortened processing time means that inventory turnover is faster, allowing manufacturers to respond more agilely to market demand spikes. This reliability makes the manufacturer a preferred partner for pharmaceutical companies seeking a dependable source of high-quality intermediates without the risk of production bottlenecks.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with modern green chemistry principles. The reduction in solvent usage and the elimination of energy-intensive purification steps lower the overall carbon footprint of the manufacturing process. The ability to recycle solvents and reagents minimizes the volume of chemical waste requiring treatment, simplifying compliance with increasingly stringent environmental regulations. The process is inherently scalable, as the unit operations involved—reflux, filtration, and crystallization—are standard in industrial chemical engineering and do not require exotic equipment. This ease of scale-up ensures that production can be ramped from pilot quantities to multi-ton annual capacities smoothly, securing the supply chain for future commercial needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved Aripiprazole synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and quality outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the improved method prevent di-substitution side reactions?
A: The method utilizes a significant molar excess (at least 6 equivalents) of 1,4-dihalobutane during the etherification step. This stoichiometric imbalance kinetically favors the mono-substitution required for the intermediate, drastically reducing the formation of di-substituted byproducts that are difficult to separate.
Q: What are the advantages of using an azeotropic ketone-water mixture?
A: Using a butanone-water azeotropic mixture creates a homogeneous reaction system during the condensation phase. This enhances the contact between reactants, improves overall yield, and simplifies post-treatment, allowing for direct crystallization upon cooling without the need for complex distillation or recrystallization steps.
Q: Can the raw materials be recovered to reduce costs?
A: Yes, the process is designed for high efficiency. The filtrate from the etherification step allows for the recovery of approximately 95% of the unreacted 1,4-dihalobutane and the non-polar solvent, which can be directly recycled into subsequent batches, significantly lowering raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceuticals like Aripiprazole. Our team of expert chemists has thoroughly analyzed the technological advancements presented in CN101781246A and is fully equipped to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and compliant with cGMP standards. Our state-of-the-art facilities are designed to handle the specific solvent systems and reaction conditions required for this process, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch released. We are committed to delivering a product that meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By leveraging our manufacturing expertise, you can achieve a Customized Cost-Saving Analysis tailored to your volume needs, ensuring maximum economic efficiency. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your API sourcing strategy. Together, we can drive innovation and reliability in the production of essential mental health medications.
