Advanced Aripiprazole Manufacturing: Overcoming Dimerization via Phosphonium Salt Mediation
Introduction to Next-Generation Aripiprazole Synthesis
The pharmaceutical landscape for antipsychotic medications continues to evolve, driven by the demand for higher purity intermediates and more sustainable manufacturing processes. Patent CN109251172B introduces a significant technological breakthrough in the synthesis of Aripiprazole, a third-generation antipsychotic known as a dopamine system stabilizer. This innovative method addresses long-standing challenges in the alkylation of quinolinone derivatives by replacing traditional halogenated alkylating agents with a phosphonium salt-mediated coupling strategy. By utilizing 7-(4-hydroxybutoxy)-3,4-dihydro-2(1H)-quinolinone as a key starting material, the process achieves exceptional reaction selectivity and minimizes the formation of problematic dimer byproducts. For R&D directors and procurement specialists, this represents a pivotal shift towards a more robust and economically viable supply chain for high-value psychiatric medications. The protocol not only streamlines the synthetic route into a manageable two-step sequence but also ensures that the final active pharmaceutical ingredient meets stringent purity specifications required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Aripiprazole has relied heavily on routes described in earlier patents such as US5006528, which utilize 1,4-dibromobutane or similar di-halogenated alkanes to link the quinolinone and piperazine moieties. 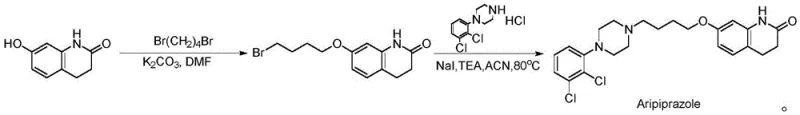 These traditional alkylation strategies suffer from inherent chemoselectivity issues, where the di-halogenated linker can react twice with the nucleophilic quinolinone oxygen, leading to the formation of symmetrical ether dimers. Literature and patent data indicate that these dimer byproducts can constitute 10-20% of the reaction mixture, creating a severe bottleneck in downstream processing. The removal of these structurally similar impurities often requires complex chromatographic separations or multiple recrystallization cycles, which drastically reduces overall yield and increases solvent consumption. Furthermore, the use of excess di-halogenated reagents poses environmental and safety concerns due to their toxicity and potential carcinogenicity. For supply chain managers, the variability in yield caused by these side reactions translates into unpredictable production schedules and inflated raw material costs, making the conventional route suboptimal for large-scale commercial manufacturing.
These traditional alkylation strategies suffer from inherent chemoselectivity issues, where the di-halogenated linker can react twice with the nucleophilic quinolinone oxygen, leading to the formation of symmetrical ether dimers. Literature and patent data indicate that these dimer byproducts can constitute 10-20% of the reaction mixture, creating a severe bottleneck in downstream processing. The removal of these structurally similar impurities often requires complex chromatographic separations or multiple recrystallization cycles, which drastically reduces overall yield and increases solvent consumption. Furthermore, the use of excess di-halogenated reagents poses environmental and safety concerns due to their toxicity and potential carcinogenicity. For supply chain managers, the variability in yield caused by these side reactions translates into unpredictable production schedules and inflated raw material costs, making the conventional route suboptimal for large-scale commercial manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN109251172B circumvents these pitfalls by employing a mono-functionalized hydroxy-intermediate, 7-(4-hydroxybutoxy)-3,4-dihydro-2(1H)-quinolinone, which is subsequently activated in situ. 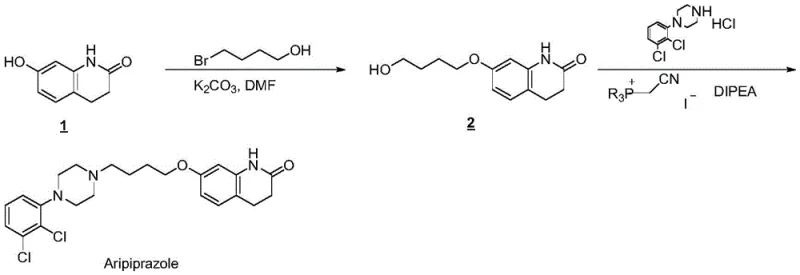 This strategic modification ensures that the alkyl chain is already attached to the quinolinone core before the coupling with the piperazine ring occurs, thereby physically preventing the formation of the symmetric dimer impurity. The activation is achieved using (cyanomethyl)trialkylphosphonium iodide in the presence of a base like diisopropylethylamine, facilitating a clean nucleophilic substitution without the need for harsh halogenating agents in the final coupling step. This approach not only simplifies the reaction profile but also enhances the safety profile of the manufacturing process by eliminating volatile and toxic halogenated solvents typically associated with older methods. The result is a streamlined workflow where the intermediate can be easily purified via recrystallization, ensuring that the final coupling reaction proceeds with high efficiency and minimal contamination from structural analogs.
This strategic modification ensures that the alkyl chain is already attached to the quinolinone core before the coupling with the piperazine ring occurs, thereby physically preventing the formation of the symmetric dimer impurity. The activation is achieved using (cyanomethyl)trialkylphosphonium iodide in the presence of a base like diisopropylethylamine, facilitating a clean nucleophilic substitution without the need for harsh halogenating agents in the final coupling step. This approach not only simplifies the reaction profile but also enhances the safety profile of the manufacturing process by eliminating volatile and toxic halogenated solvents typically associated with older methods. The result is a streamlined workflow where the intermediate can be easily purified via recrystallization, ensuring that the final coupling reaction proceeds with high efficiency and minimal contamination from structural analogs.
Mechanistic Insights into Phosphonium Salt-Mediated Coupling
The core innovation of this synthesis lies in the utilization of (cyanomethyl)trialkylphosphonium iodide as a coupling promoter, which functions through a mechanism distinct from traditional Mitsunobu or halogenation protocols. In this system, the phosphonium salt acts as an activating agent for the hydroxyl group on the butoxy side chain of the quinolinone intermediate, converting the poor leaving group (-OH) into a highly reactive species capable of undergoing nucleophilic attack by the piperazine nitrogen. The presence of the electron-withdrawing cyano group on the methyl position of the phosphonium salt enhances the electrophilicity of the phosphorus center, facilitating the formation of an oxyphosphonium intermediate. This intermediate is then displaced by the nucleophilic nitrogen of 1-(2,3-dichlorophenyl)piperazine, driving the reaction forward under mild thermal conditions ranging from 30°C to 40°C. The use of diisopropylethylamine serves a dual purpose: it neutralizes the hydrochloride salt of the piperazine to generate the free base and scavenges any acidic byproducts generated during the activation cycle, maintaining the reaction environment conducive to high conversion rates.
From an impurity control perspective, this mechanistic pathway offers superior selectivity compared to acid-catalyzed dehydrations or halogen-based activations. Because the activation is specific to the primary alcohol and the subsequent substitution is intermolecular rather than intramolecular or polymeric, the generation of oligomeric side products is thermodynamically disfavored. The patent data highlights that single impurities in the intermediate stages can be controlled to meet bulk drug standards simply through recrystallization from ethanol or ethanol-water mixtures. This level of control is critical for R&D teams aiming to minimize genotoxic impurities and ensure that the final API complies with ICH Q3 guidelines. The ability to achieve a total yield of 60-70% for the qualified raw medicine, with final step yields reaching as high as 83% in optimized examples, underscores the efficiency of this catalytic system in preserving molecular integrity throughout the synthesis.
How to Synthesize Aripiprazole Efficiently
The practical implementation of this synthesis route involves a straightforward sequence that begins with the preparation of the hydroxy-intermediate, followed by the critical phosphonium-mediated coupling step. Operators charge the reactor with 7-(4-hydroxybutoxy)-3,4-dihydro-2(1H)-quinolinone and the piperazine hydrochloride salt in a solvent such as dichloromethane or toluene under an inert nitrogen atmosphere. The addition of the phosphonium salt and base is carefully controlled to manage exotherms, after which the mixture is stirred at moderate temperatures to ensure complete conversion. Following the reaction, the workup involves a simple pH adjustment to alkaline conditions to separate the organic product from aqueous salts, followed by standard drying and concentration procedures.
- React 7-(4-hydroxybutoxy)-3,4-dihydro-2(1H)-quinolinone with 1-(2,3-dichlorophenyl)piperazine hydrochloride in the presence of (cyanomethyl)trialkylphosphonium iodide and diisopropylethylamine.
- Adjust the reaction mixture pH to alkaline conditions (pH 11-12) using saturated sodium carbonate solution to facilitate phase separation.
- Extract the organic phase, dry over anhydrous sodium sulfate, concentrate under reduced pressure, and recrystallize from ethanol to obtain high-purity Aripiprazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this phosphonium-mediated synthesis offers tangible benefits that extend beyond mere chemical elegance, directly impacting the bottom line through operational efficiency and risk mitigation. The elimination of dimer byproducts, which historically plagued older synthesis routes, removes the need for expensive and time-consuming purification steps such as preparative HPLC or extensive column chromatography. This simplification of the downstream processing significantly reduces solvent usage and waste disposal costs, aligning with modern green chemistry initiatives while lowering the overall cost of goods sold. Furthermore, the reliance on commercially available and low-cost reagents, such as simple alkyl phosphonium salts and common organic solvents, ensures a stable supply chain that is less susceptible to the volatility associated with specialized or hazardous halogenated reagents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in reaction selectivity, which inherently boosts the effective yield of the desired product. By avoiding the formation of 10-20% dimer impurities seen in conventional methods, the process maximizes the utilization of raw materials, meaning less starting material is wasted on side products that must be discarded. Additionally, the ability to purify intermediates via simple recrystallization rather than complex chromatographic techniques translates to substantial savings in both consumable materials and labor hours. The use of inexpensive solvents like toluene or dichloromethane, which can be readily recovered and recycled, further contributes to a leaner manufacturing cost structure without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-proprietary reagents that are widely available from multiple global chemical suppliers. Unlike processes that depend on custom-synthesized catalysts or unstable intermediates, the reagents used here—such as (cyanomethyl)trialkylphosphonium iodide—are stable and easy to store, reducing the risk of production delays due to material degradation or shortage. The simplified two-step nature of the final coupling sequence also shortens the overall production cycle time, allowing manufacturers to respond more agilely to fluctuations in market demand for antipsychotic medications. This reliability is crucial for maintaining continuous supply to downstream formulators and ensuring that inventory levels remain optimal throughout the distribution network.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with reaction conditions that are easily transferable from laboratory benchtop to multi-ton industrial reactors. The moderate temperature requirements (30-40°C) and atmospheric pressure operations minimize energy consumption and reduce the engineering complexity of the production equipment. From an environmental standpoint, the avoidance of heavy metal catalysts and the reduction of halogenated waste streams simplify wastewater treatment and effluent management, helping facilities maintain compliance with increasingly stringent environmental regulations. The high purity of the final product (99.85-99.98%) also reduces the burden on quality control laboratories, enabling faster batch release times and quicker time-to-market for the finished pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: How does this new method improve upon traditional alkylation routes?
A: Traditional methods using 1,4-dibromobutane often suffer from poor selectivity, generating 10-20% dimer byproducts that are difficult to remove. This novel approach utilizes a hydroxy-intermediate activated by phosphonium salts, effectively suppressing dimerization and simplifying purification.
Q: What is the expected purity profile of the final API intermediate?
A: The process allows for recrystallization of intermediates at each step, ensuring single impurity levels meet bulk drug standards. The final product consistently achieves HPLC purity between 99.85% and 99.98%.
Q: Are the reagents used in this phosphonium-mediated coupling commercially viable?
A: Yes, the method relies on low-cost, commercially available reagents such as (cyanomethyl)trialkylphosphonium iodide and standard solvents like dichloromethane or toluene, making it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively analyzed the phosphonium-mediated coupling route described in CN109251172B and validated its potential for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Aripiprazole intermediate we produce meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners and contract manufacturers to collaborate with us to leverage this superior synthesis technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your operational efficiency and reduce your overall procurement costs. Let us be your trusted partner in delivering high-quality Aripiprazole intermediates that drive the success of your final drug products.
