Advanced Catalytic Oxidation Route for High-Purity Nalbuphine Production
Advanced Catalytic Oxidation Route for High-Purity Nalbuphine Production
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex opioid analgesics, and the recent disclosure in patent CN112759596B presents a significant breakthrough in the manufacturing of Nalbuphine. This novel methodology departs from traditional reduction-heavy routes by leveraging a sophisticated catalytic oxidation strategy starting directly from morphine. By introducing the chiral center at the very beginning of the synthesis, the process inherently avoids the formation of difficult-to-remove stereoisomers that have plagued previous generations of synthetic routes. This technical advancement not only streamlines the production workflow but also ensures a level of stereochemical integrity that is critical for regulatory compliance in pain management therapeutics. As a reliable nalbuphine intermediate supplier, understanding these mechanistic shifts is vital for projecting future supply stability.
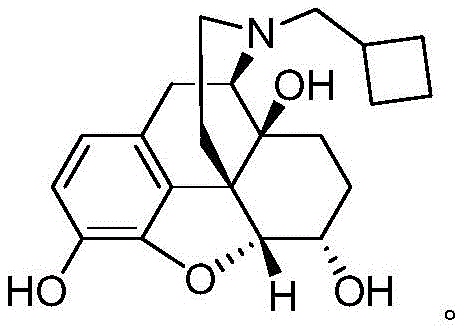
Nalbuphine serves as a crucial semi-synthetic opioid receptor mixed agonist-antagonist, widely utilized for managing moderate to severe pain in clinical settings including post-operative care and obstetrics. The structural complexity of this molecule, particularly the stereochemistry at the 6-position and the nitrogen substituent, dictates its pharmacological profile and safety margin. Traditional synthetic approaches often struggle with the thermodynamic equilibrium of the 6-hydroxyl group, leading to impurity profiles that require extensive and costly purification steps. The new route described in the patent data addresses these legacy issues head-on by preserving the natural chirality of the morphine starting material throughout the entire sequence. This strategic preservation of stereochemistry translates directly into higher overall yields and a cleaner crude product profile, which are key metrics for any procurement manager evaluating cost reduction in opioid analgesic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Nalbuphine has relied heavily on the reduction of 6-keto intermediates, such as 14-hydroxynormorphinone, using strong reducing agents like lithium aluminium hydride or sodium borohydride. While chemically feasible, these reduction steps are notoriously non-selective regarding the stereochemistry at the 6-position, invariably generating a mixture of the desired 6-alpha alcohol and the unwanted 6-beta isomer. Separating these diastereomers is a formidable challenge in process chemistry, often requiring multiple recrystallizations or chromatographic separations that drastically reduce the final mass yield. Furthermore, the use of hazardous reagents like lithium aluminium hydride introduces significant safety risks and requires specialized handling equipment, increasing the capital expenditure for manufacturing facilities. The accumulation of solvent waste from these purification steps also poses environmental compliance challenges, making the conventional route less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the innovative pathway disclosed in the patent utilizes morphine as the foundational building block, thereby locking in the correct 6-alpha stereochemistry from step one. The core of this novelty lies in a selective catalytic oxidation at the 14-position, transforming the diacetyl-morphine intermediate into a 14-benzoyloxy protected species without disturbing the sensitive 6-hydroxyl group. This approach effectively bypasses the need for a 6-keto reduction entirely, eliminating the root cause of the 6-beta isomer impurity. The subsequent steps involve standard catalytic hydrogenation and N-demethylation followed by a clean alkylation with cyclobutylmethyl bromide. This linear progression minimizes unit operations and avoids the yield losses associated with isomer separation. For supply chain heads, this represents a robust pathway for the commercial scale-up of complex morphinan derivatives, offering a predictable and consistent output of high-purity API.
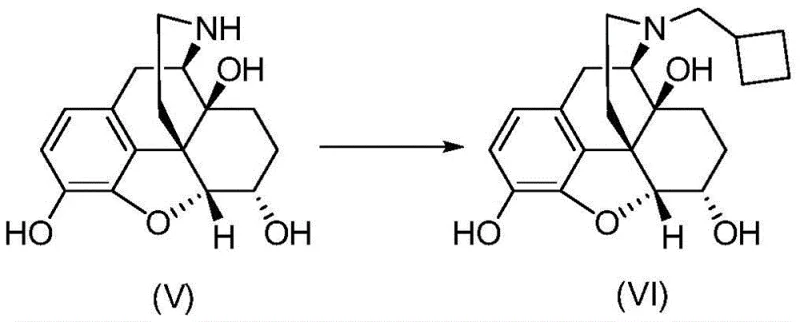
Mechanistic Insights into CuI-Catalyzed Allylic Oxidation
The centerpiece of this synthetic innovation is the transition metal-catalyzed oxidation step, which converts the acetylated morphine derivative into the 14-protected intermediate. This transformation employs a specific catalytic system comprising Cuprous Iodide (CuI) as the metal source and 1,4-diazabicyclo[2.2.2]octane (DABCO) as the nitrogen ligand, activated by tert-butyl peroxybenzoate (TBPB) as the oxidant. The mechanistic rationale involves the generation of a benzoyloxy radical from the peroxide, which selectively abstracts an allylic hydrogen at the C14 position. The presence of the DABCO ligand is critical; it complexes with the copper center to modulate its redox potential and steric environment, thereby enhancing the regioselectivity for the C14 position over other susceptible sites on the morphinan skeleton. This level of control is essential for maintaining the integrity of the molecule while installing the necessary functionality for subsequent transformations.
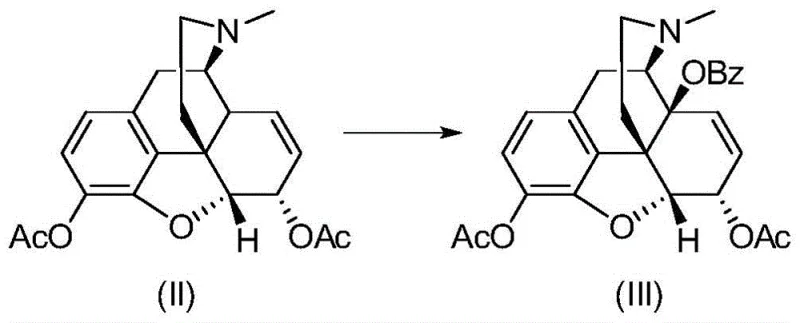
From an impurity control perspective, this catalytic system offers distinct advantages over stoichiometric oxidants. The mild reaction conditions, typically conducted between 70°C and 100°C in aprotic solvents like 1,4-dioxane, prevent thermal degradation or epimerization of the chiral centers. Experimental data within the patent indicates that the choice of ligand and copper salt significantly impacts the product content, with the CuI/DABCO combination yielding superior results compared to other copper salts or ligands like DBU. This specificity ensures that side reactions, such as over-oxidation or ring cleavage, are minimized. For R&D directors focused on purity and杂质谱 (impurity profiles), this mechanism provides a clear pathway to achieving the stringent purity specifications required for injectable analgesics, as the absence of isomeric impurities simplifies the final crystallization and quality control processes significantly.
How to Synthesize Nalbuphine Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the novel catalytic steps. The process begins with the acylation of morphine, followed by the critical copper-catalyzed oxidation, hydrogenation, deprotection, and final alkylation. Each step has been optimized to use commercially available reagents and standard laboratory or plant equipment, ensuring that the transfer from bench scale to pilot plant is seamless. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency route.
- Acylation of morphine using acetic anhydride and pyridine to form the diacetyl protected intermediate.
- Catalytic oxidation at the 14-position using tert-butyl peroxybenzoate (TBPB) with a CuI/DABCO catalyst system.
- Catalytic hydrogenation of the double bond using Pd/C followed by N-demethylation and hydrolysis.
- Final N-alkylation with cyclobutylmethyl bromide to yield high-purity Nalbuphine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route offers compelling economic and operational benefits that extend beyond simple yield improvements. By eliminating the need for chiral resolution or extensive isomer separation, the process drastically reduces the consumption of solvents and stationary phases, which are often major cost drivers in API manufacturing. The reliance on morphine, a naturally abundant alkaloid, ensures a stable and secure raw material supply base, mitigating the risks associated with synthetic starting materials that may have volatile pricing or availability. Furthermore, the use of conventional catalytic hydrogenation and standard alkylation reactions means that existing manufacturing infrastructure can be utilized without the need for significant capital investment in specialized reactors or containment systems.
- Cost Reduction in Manufacturing: The elimination of the 6-keto reduction step removes the necessity for expensive and hazardous reducing agents like lithium aluminium hydride, which require strict anhydrous conditions and careful quenching protocols. This substitution with a catalytic oxidation step not only lowers reagent costs but also reduces the energy burden associated with maintaining cryogenic temperatures or handling pyrophoric materials. Additionally, the higher selectivity of the new route means that less material is lost to waste streams during purification, effectively increasing the mass efficiency of the entire process and driving down the cost per kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the CuI/DABCO catalytic system contributes to greater batch-to-batch consistency, a critical factor for maintaining uninterrupted supply to downstream formulation partners. Because the reaction conditions are mild and tolerant of minor variations, the risk of batch failure due to excursions in temperature or mixing is significantly reduced. This reliability allows for more accurate production planning and inventory management, ensuring that lead times for high-purity nalbuphine intermediates remain short and predictable even during periods of high market demand.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are easily handled on a multi-ton scale. The avoidance of heavy metal catalysts that are difficult to remove (like some palladium variants used in other contexts) or the generation of heavy metal waste simplifies the environmental remediation process. The overall shorter route length reduces the cumulative environmental footprint of the manufacturing process, aligning with increasingly stringent global regulations on pharmaceutical emissions and waste disposal, thus future-proofing the supply chain against regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Nalbuphine synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical principles underpinning the technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this method into their existing production portfolios.
Q: How does this new route control the 6-beta isomer impurity?
A: Unlike traditional routes that reduce a 6-ketone (creating a mix of alpha and beta isomers), this method starts with morphine which already possesses the correct 6-alpha hydroxyl stereochemistry. The subsequent steps do not affect this chiral center, effectively eliminating the formation of the unwanted 6-beta isomer.
Q: What is the advantage of the CuI/DABCO catalyst system?
A: The combination of Cuprous Iodide (CuI) and DABCO ligand significantly enhances the selectivity and yield of the allylic oxidation at the 14-position. It allows for mild reaction conditions (70-100°C) compared to harsher traditional oxidants, reducing side reactions and simplifying purification.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes conventional equipment and reagents (such as Pd/C hydrogenation and standard acylation). The avoidance of cryogenic conditions or highly sensitive reagents like Lithium Aluminum Hydride makes it highly scalable and safer for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nalbuphine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic oxidation technology in reshaping the landscape of opioid analgesic production. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Nalbuphine intermediate or API meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and high-yield synthetic route for their next-generation pain management products. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive global market.
