Advanced Synthesis of Nalbuphine Related Compounds for Commercial Scale Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust methodologies to ensure the safety and efficacy of potent analgesics like Nalbuphine Hydrochloride. Patent CN114075209A introduces a groundbreaking nitrogen-containing compound and its preparation method, specifically designed to enhance the quality control of Nalbuphine Hydrochloride. This innovation addresses the critical lack of research on related substances, which are vital for stability and medication safety assessments. By utilizing Thebaine as a raw material, the patent outlines a streamlined three-step reaction sequence that yields the target nitrogen-containing compound shown as Formula I. This technical advancement not only provides a reliable reference standard for impurity profiling but also offers a scalable pathway for producing complex pharmaceutical intermediates. For R&D directors and supply chain leaders, understanding this novel synthesis route is essential for maintaining stringent purity specifications and ensuring continuous supply of high-quality analgesic medications in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
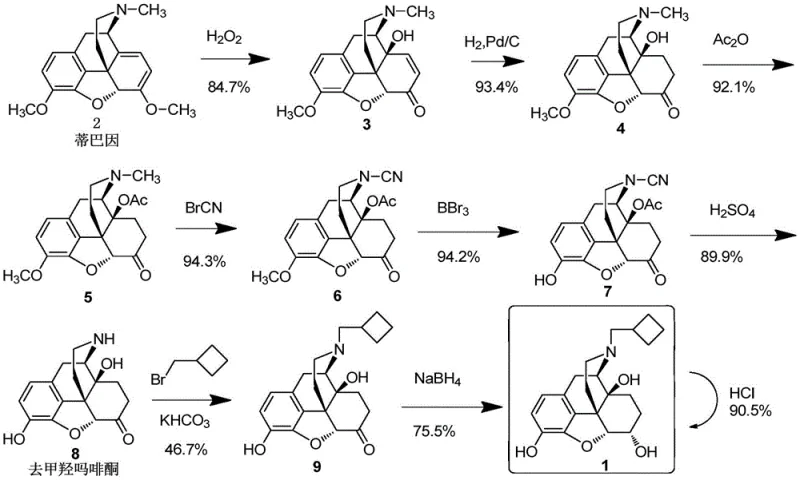
Historically, the synthesis of Nalbuphine and its related substances has relied on complex, multi-step pathways that pose significant challenges for industrial scalability and cost efficiency. As illustrated in the literature, the conventional route involves oxidizing Thebaine to 14-hydroxycodeinone, followed by catalytic hydrogenation, acetylation, and reaction with cyanogen bromide. Subsequent steps require the use of boron tribromide for demethylation and sulfuric acid for hydrolysis, creating a hazardous operational environment. These traditional methods often suffer from low overall yields due to the accumulation of impurities at each stage, necessitating extensive purification processes that drive up manufacturing costs. Furthermore, the use of aggressive reagents and multiple isolation steps increases the risk of safety incidents and environmental compliance issues. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, making the conventional approach less viable for competitive commercial production in the modern pharmaceutical landscape.
The Novel Approach
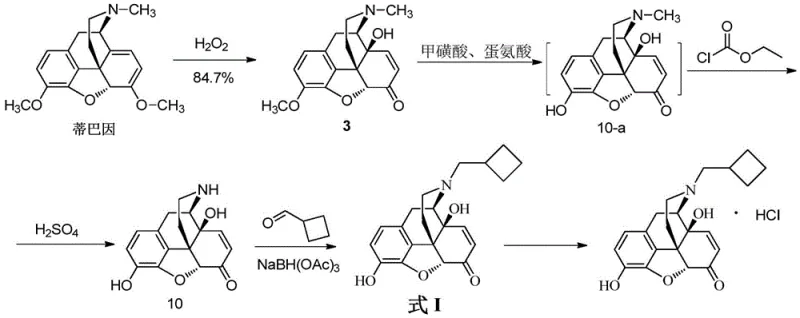
In contrast, the novel approach disclosed in patent CN114075209A offers a significantly optimized pathway that mitigates the drawbacks of traditional synthesis. This method leverages a more direct transformation starting from Thebaine, utilizing methanesulfonic acid and methionine for demethylation, which is safer and more controllable than boron tribromide. The process culminates in a condensation reaction with cyclobutylformaldehyde followed by reduction, achieving the target Formula I with high purity and yield. By reducing the number of intermediate isolation steps and employing milder reaction conditions, this new route enhances operational safety and simplifies waste management. For supply chain heads, this translates to a more reliable production schedule with reduced risk of batch failures. The streamlined nature of this synthesis allows for better resource allocation and potentially lower operational expenditures, positioning it as a superior choice for manufacturers aiming to optimize their production of high-purity pharmaceutical intermediates while maintaining strict regulatory compliance.
Mechanistic Insights into Methanesulfonic Acid Demethylation and Reductive Amination
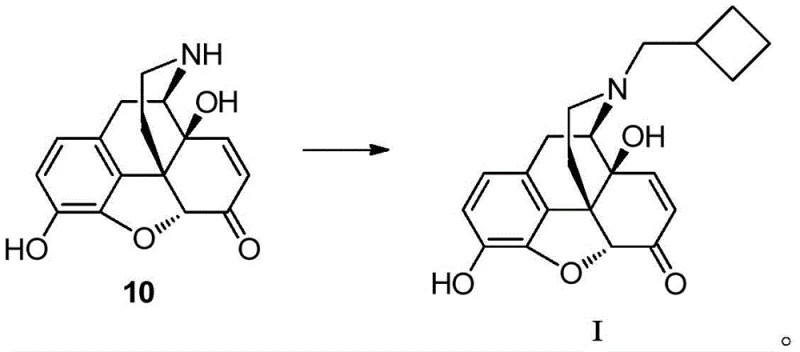
The core of this technological breakthrough lies in the precise mechanistic control of the demethylation and reductive amination steps. The demethylation of Compound 3 to Compound 10-a utilizes methanesulfonic acid in the presence of methionine, a mechanism that selectively removes the methyl group without compromising the sensitive morphinan skeleton. This selectivity is crucial for minimizing the formation of side products that could complicate downstream purification. Following this, the conversion of Compound 10 to Formula I involves a condensation reaction with cyclobutylformaldehyde in a polar solvent like DMSO. The subsequent reduction using sodium triacetoxyborohydride is highly chemoselective, ensuring that the ketone functionality is preserved while the imine intermediate is efficiently reduced to the amine. This level of mechanistic precision ensures that the final product meets stringent purity specifications, which is paramount for R&D directors focused on impurity谱 analysis. The controlled reaction environment prevents over-reduction or degradation, thereby securing the structural integrity of the nitrogen-containing compound essential for accurate quality control.
Furthermore, the impurity control mechanism inherent in this process is designed to limit the generation of unknown related substances. By optimizing the molar ratios of reagents, such as maintaining a 1:1.2 ratio of Compound 10 to cyclobutylformaldehyde, the reaction drives towards completion while minimizing excess reagent carryover. The use of specific solvents and temperature controls, ranging from 20°C to 60°C, further stabilizes the reaction intermediates. This meticulous control over reaction parameters ensures that the impurity profile remains consistent and predictable, facilitating easier validation during regulatory filings. For technical teams, understanding these mechanistic nuances allows for better troubleshooting and process optimization during scale-up. The ability to consistently produce a compound with a defined impurity profile is a significant advantage, ensuring that the final Nalbuphine Hydrochloride product meets all safety and efficacy requirements set by global health authorities.
How to Synthesize 17-(cyclobutanemethyl)-7,8-dihydro-4,5-alpha-epoxymorphinan-6-one-3,14-diol Efficiently
The synthesis of this specific nitrogen-containing compound requires strict adherence to the patented conditions to ensure high yield and purity. The process begins with the preparation of Compound 10, which serves as the key intermediate, followed by the critical reductive amination step. Operators must carefully control the temperature and stoichiometry during the condensation phase to avoid side reactions. The detailed standardized synthesis steps, including specific solvent volumes and reaction times, are critical for reproducibility and are outlined in the technical guide below. Following these protocols ensures that the final product is suitable for use as a reference standard in quality control laboratories. Adherence to these guidelines is essential for maintaining the integrity of the supply chain and ensuring that the produced intermediates meet the rigorous demands of pharmaceutical manufacturing.
- Oxidize Thebaine using hydrogen peroxide to obtain 14-hydroxycodeinone (Compound 3) with high purity.
- Perform demethylation on Compound 3 using methanesulfonic acid and methionine to yield Compound 10-a, followed by hydrolysis to Compound 10.
- Conduct condensation with cyclobutylformaldehyde and reduce with sodium triacetoxyborohydride to finalize the nitrogen-containing compound Formula I.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthesis route offers substantial commercial benefits for organizations focused on cost efficiency and supply chain resilience. The elimination of hazardous reagents like boron tribromide reduces the need for specialized handling equipment and safety protocols, leading to significant operational cost savings. Additionally, the simplified process flow decreases the time required for production cycles, allowing for faster response to market demands. For procurement managers, this means a more stable supply of critical intermediates with reduced risk of disruption due to safety incidents or regulatory hurdles. The enhanced efficiency of the process also contributes to a lower environmental footprint, aligning with corporate sustainability goals and reducing waste disposal costs. These qualitative improvements collectively strengthen the overall value proposition of the manufacturing process.
- Cost Reduction in Manufacturing: The streamlined synthesis route eliminates the need for expensive and hazardous reagents, which directly lowers the raw material costs associated with production. By reducing the number of purification steps required, the process also minimizes solvent consumption and energy usage, leading to further cost optimizations. The higher yield stability ensures that less raw material is wasted, maximizing the output from each batch. These factors combine to create a more economically viable production model that enhances profit margins without compromising on quality. Consequently, manufacturers can offer more competitive pricing for their pharmaceutical intermediates in the global market.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents ensures that the supply chain is less vulnerable to disruptions caused by scarce or regulated materials. The robust nature of the reaction conditions allows for consistent production schedules, reducing the likelihood of batch failures that can delay deliveries. This reliability is crucial for maintaining continuous operations and meeting the strict deadlines imposed by downstream pharmaceutical clients. Furthermore, the simplified process reduces the dependency on specialized equipment, making it easier to scale production across different facilities. This flexibility enhances the overall resilience of the supply chain against external shocks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard industrial solvents and equipment that are readily available in most manufacturing plants. The reduction in hazardous waste generation simplifies compliance with environmental regulations, reducing the administrative burden and potential fines associated with waste management. This ease of compliance facilitates smoother regulatory approvals and faster time-to-market for new products. Additionally, the safer operating conditions improve worker safety, contributing to a more sustainable and responsible manufacturing environment. These attributes make the process highly attractive for large-scale commercial adoption.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this nitrogen-containing compound. These answers are derived directly from the technical specifications and beneficial effects described in the patent data. They provide clarity on the process capabilities and its alignment with industry standards for quality and safety. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this technology into their existing production frameworks. The information provided here serves as a foundational guide for further technical discussions and partnership opportunities.
Q: What is the primary advantage of the new synthesis route for Nalbuphine intermediates?
A: The new route described in patent CN114075209A significantly simplifies the process by reducing the number of steps and avoiding harsh reagents like boron tribromide, leading to better impurity control and higher overall safety.
Q: How does this method improve quality control for Nalbuphine Hydrochloride?
A: By providing a reliable method to synthesize specific related substances (Formula I), this process enables manufacturers to produce accurate reference standards, ensuring rigorous quality control and medication safety for the final API.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the use of conventional solvents like DMSO and chloroform, along with manageable reaction temperatures (20-60°C), makes the process highly scalable and compliant with standard industrial safety and environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17-(cyclobutanemethyl)-7,8-dihydro-4,5-alpha-epoxymorphinan-6-one-3,14-diol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at implementing complex synthesis routes like the one described in CN114075209A, ensuring that stringent purity specifications are met consistently. We operate rigorous QC labs equipped with advanced analytical instruments to verify the identity and purity of every batch. This commitment to quality ensures that our clients receive pharmaceutical intermediates that are ready for immediate use in drug development and manufacturing. Our infrastructure supports the high demands of the global pharmaceutical industry, providing a secure and reliable source for critical compounds.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our Customized Cost-Saving Analysis. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your project needs. By partnering with us, you gain access to a wealth of technical expertise and production capacity that can accelerate your product development timelines. Contact us today to discuss how we can support your requirements for high-purity pharmaceutical intermediates and ensure the success of your commercial projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
