Revolutionizing Chiral Amine Synthesis with High-Performance Monoamine Oxidase Mutants for Commercial Scale-up
Revolutionizing Chiral Amine Synthesis with High-Performance Monoamine Oxidase Mutants for Commercial Scale-up
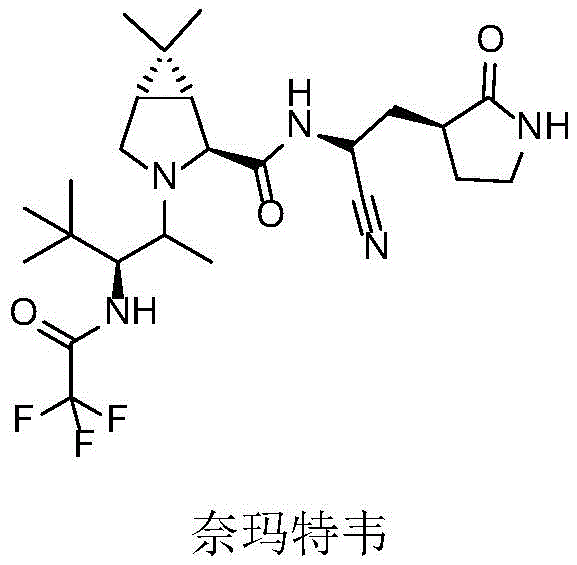
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable and highly selective biocatalytic processes, particularly for the synthesis of complex chiral amine building blocks which are ubiquitous in modern therapeutics. A groundbreaking development in this field is detailed in patent CN114736882A, which discloses novel applications of monoamine oxidase (MAO) and its engineered mutants for the asymmetric oxidation of latent chiral nitrogen heterocyclic compounds. This technology addresses critical bottlenecks in the production of key antiviral agents, including the active pharmaceutical ingredient Nirmatrelvir, a crucial component of Paxlovid used globally for COVID-19 treatment. The structural complexity of such molecules demands precision that traditional chemical synthesis often struggles to achieve without significant environmental cost.
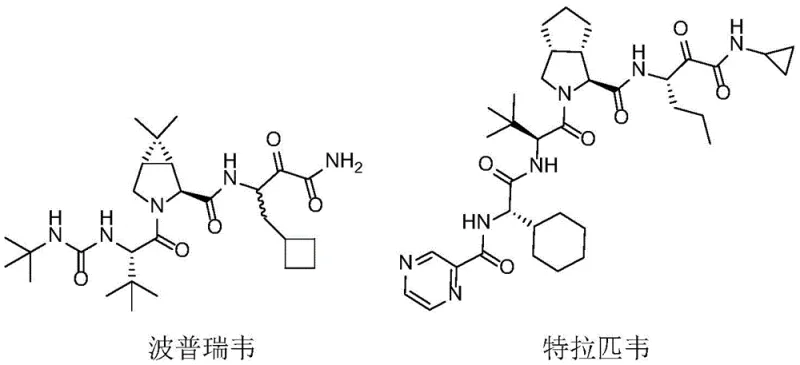
Furthermore, the relevance of this technology extends to other major antiviral therapies such as Boceprevir and Telaprevir, which are FDA-approved treatments for Hepatitis C Virus (HCV). The chemical architectures of these life-saving medications rely heavily on specific chiral amine motifs that serve as the foundational scaffolds for their biological activity. By leveraging the enzymatic specificity described in this patent, manufacturers can access these critical intermediates with unprecedented purity and efficiency. This represents a significant leap forward for any organization seeking a reliable pharmaceutical intermediate supplier capable of meeting the rigorous quality standards demanded by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral amines has relied heavily on chemical asymmetric resolution and synthesis, methodologies that are fraught with inherent inefficiencies and environmental hazards. Traditional chemical routes often necessitate the use of toxic and harmful transition metal catalysts, which introduce significant challenges in downstream processing, particularly regarding the removal of heavy metal residues to meet stringent pharmaceutical limits. Moreover, these chemical methods frequently operate under harsh reaction conditions involving extreme temperatures or pressures, leading to complex operational procedures and increased safety risks in a manufacturing environment. A major technical hurdle has been the low enantioselectivity and poor substrate tolerance of earlier biocatalysts; for instance, previous generations of monoamine oxidases like MAO-N D5 were limited to substrate concentrations below 25mM, rendering them economically unviable for large-scale production due to excessive solvent usage and low volumetric productivity.
The Novel Approach
In stark contrast, the novel approach outlined in patent CN114736882A utilizes specifically screened and engineered monoamine oxidases, such as PsMAO, ZsMAO, and AsMAO, along with their hyper-active mutants, to catalyze the desymmetry oxidation of azacyclic compounds. This biocatalytic strategy operates under mild reaction conditions, typically between 10-30°C and near-neutral pH, drastically reducing energy consumption and safety risks associated with high-pressure or high-temperature operations. The most transformative aspect of this technology is its ability to tolerate extremely high substrate concentrations, reaching up to 120g/L, which is a massive improvement over prior art that struggled beyond 65g/L. This high loading capacity directly translates to reduced reactor volumes and solvent waste, offering a clear pathway for cost reduction in API manufacturing while maintaining exceptional stereocontrol.
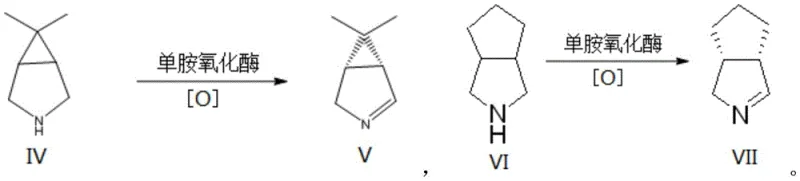
Mechanistic Insights into Monoamine Oxidase Catalyzed Asymmetric Oxidation
The core of this technological advancement lies in the precise mechanistic action of the monoamine oxidase enzyme on prochiral nitrogen heterocyclic substrates. The enzyme facilitates a desymmetrization reaction where a symmetric or meso-compound is selectively oxidized to generate a single enantiomer of a chiral imine with high fidelity. The patent details specific substrate scopes, including 6,6-dimethyl-3-azabicyclo[3.1.0]hexane and octahydrocyclopenta[c]pyrrole, which are converted into their corresponding chiral imine intermediates. The catalytic cycle involves the abstraction of a hydride from the substrate, coupled with the reduction of molecular oxygen to hydrogen peroxide, which is subsequently decomposed by co-added catalase to prevent enzyme inactivation. This elegant mechanism ensures that the reaction proceeds with minimal byproduct formation, preserving the integrity of the sensitive chiral centers required for downstream drug synthesis.
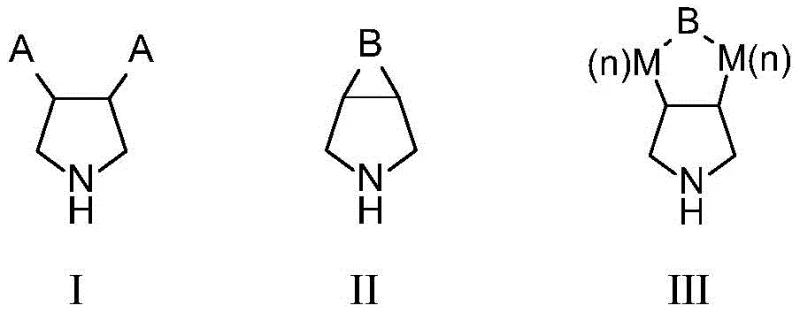
Impurity control is another critical dimension where this enzymatic method excels, primarily due to the innate specificity of the protein catalyst. Unlike chemical oxidants which may attack various sites on a molecule leading to complex impurity profiles, the monoamine oxidase mutants described herein exhibit strict regioselectivity and stereoselectivity. The engineered mutants, such as PsMAO-M18, have been optimized through directed evolution to minimize non-specific oxidation events, resulting in product optical purity exceeding 99% ee. This high level of purity simplifies the purification workflow, often eliminating the need for costly and yield-reducing recrystallization or chromatographic separation steps. For R&D directors, this means a more predictable and robust process that consistently delivers high-quality intermediates, reducing the risk of batch failures and ensuring compliance with tight impurity specifications.
How to Synthesize Chiral Imine Efficiently
The implementation of this biocatalytic route is designed for seamless integration into existing manufacturing infrastructure, requiring standard fermentation and reaction equipment. The process begins with the preparation of a buffered aqueous system containing the specific azacyclic substrate and the recombinant enzyme, either as a free crude extract or immobilized form. To maintain optimal reaction kinetics and prevent oxidative damage from accumulated hydrogen peroxide, catalase is added to the system, and oxygen or air is continuously sparged to serve as the terminal electron acceptor. The reaction progress is monitored via gas chromatography or HPLC, and upon reaching near-complete conversion, the product is extracted using organic solvents like methyl tert-butyl ether. For a detailed breakdown of the specific operational parameters and step-by-step execution, please refer to the standardized guide below.
- Prepare a reaction system containing a buffer solution (pH 5.0-9.0), the latent chiral nitrogen heterocyclic substrate (10-120 g/L), and catalase.
- Add the recombinant monoamine oxidase mutant (e.g., PsMAO-M18) or resting cells expressing the enzyme to the reaction mixture.
- Maintain the reaction at 10-30°C with continuous oxygen or air supply until conversion exceeds 99%, then isolate the chiral imine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this high-performance monoamine oxidase technology offers profound advantages for procurement and supply chain management, fundamentally altering the cost structure of chiral amine production. The shift from chemical synthesis to biocatalysis eliminates the dependency on expensive and volatile transition metal catalysts, thereby removing a significant variable cost driver and mitigating supply risks associated with precious metal markets. Furthermore, the ability to run reactions at high substrate concentrations significantly reduces the volume of solvents required per kilogram of product, leading to substantial savings in raw material procurement, waste disposal fees, and energy costs associated with solvent recovery and distillation. These efficiencies collectively contribute to a more resilient and cost-effective supply chain, enabling manufacturers to offer competitive pricing without compromising on quality or delivery reliability.
- Cost Reduction in Manufacturing: The elimination of toxic heavy metal catalysts not only reduces direct material costs but also simplifies the downstream purification process, as there is no need for specialized metal scavenging resins or extensive washing steps to meet residual metal limits. Additionally, the high catalytic efficiency and turnover number of the engineered mutants mean that less enzyme is required per unit of product, further driving down the cost of goods sold. The mild reaction conditions also translate to lower energy consumption for heating or cooling, providing a dual benefit of economic savings and a reduced carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: By utilizing robust enzyme mutants that exhibit high thermostability and resistance to substrate inhibition, the manufacturing process becomes far less susceptible to batch-to-batch variability and unexpected shutdowns. The high substrate tolerance allows for smaller reactor footprints to produce the same amount of product, increasing overall plant capacity and flexibility to respond to surges in market demand. This operational robustness ensures a consistent and uninterrupted supply of critical intermediates, which is paramount for pharmaceutical companies managing tight production schedules for life-saving medications.
- Scalability and Environmental Compliance: The biocatalytic process is inherently scalable, as demonstrated by successful liter-scale trials achieving high isolated yields, making the transition from pilot to commercial production straightforward and low-risk. From an environmental standpoint, the use of water as the primary solvent and the generation of benign byproducts align perfectly with green chemistry principles, facilitating easier regulatory approval and reducing the burden of environmental compliance. This sustainability profile is increasingly becoming a key differentiator in supplier selection, appealing to global partners committed to reducing their environmental impact.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the performance and scope of these monoamine oxidase mutants. The following insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development decisions. These questions address the critical pain points of substrate inhibition, optical purity, and scalability that often dictate the success of a synthetic route in a commercial setting.
Q: What are the limitations of conventional monoamine oxidases in industrial applications?
A: Conventional monoamine oxidases, such as MAO-N D5, often suffer from low catalytic activity and severe substrate inhibition, limiting substrate concentrations to below 25mM or 65g/L, which hinders efficient industrial scale-up.
Q: How do the new PsMAO mutants improve process efficiency?
A: The novel mutants, particularly PsMAO-M18, exhibit significantly enhanced thermostability and catalytic activity, allowing for substrate loadings up to 120g/L with over 99% conversion and >99% ee, effectively eliminating substrate and product inhibition.
Q: Is this biocatalytic method suitable for large-scale manufacturing?
A: Yes, the method has been validated in liter-scale reactors with high isolated yields (over 85%) and maintains high stereoselectivity, making it highly suitable for the commercial production of chiral amine intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced biocatalytic technology and are fully equipped to leverage it for your custom synthesis needs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to full-scale manufacturing. Our state-of-the-art facilities are staffed by expert enzymologists and process chemists who specialize in optimizing biocatalytic reactions to meet stringent purity specifications, utilizing our rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to unlock the full commercial potential of your chiral amine projects using this cutting-edge monoamine oxidase platform. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your timeline and optimize your supply chain for long-term success.
