Advanced Biocatalytic Strategy for High-Purity Antiviral Intermediate Manufacturing and Scale-Up
Advanced Biocatalytic Strategy for High-Purity Antiviral Intermediate Manufacturing and Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable solutions for the synthesis of complex chiral intermediates, particularly those serving as the backbone for critical antiviral therapies. Patent CN114774382A introduces a groundbreaking advancement in this domain by disclosing a series of monoamine oxidases derived from Penicillium species that exhibit exceptional catalytic activity and strong chiral selectivity. This technology specifically targets the stereoselective preparation of 6,6-dimethyl-3-azabicyclo[3.1.0]hexene compounds, which are pivotal structural motifs found in prominent drugs such as Nirmatrelvir and Boceprevir. The core innovation lies in overcoming the historical bottleneck of low substrate tolerance in enzymatic processes, enabling reaction concentrations to reach an impressive 200g/L. This represents a paradigm shift from previous biocatalytic methods that were often restricted to laboratory-scale efficiencies due to poor productivity. For global supply chains, this development signals a new era of reliability for high-purity pharmaceutical intermediate suppliers who must meet stringent demand fluctuations.
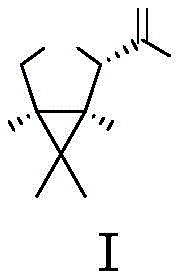
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key intermediate (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid methyl ester has been plagued by inefficiencies inherent in both purely chemical and early-generation biocatalytic routes. Traditional chemical syntheses, such as those reported in earlier literature, often involve multi-step sequences with lengthy reaction times and harsh conditions that compromise overall yield. Furthermore, achieving high enantiomeric excess (ee) frequently necessitates diastereomeric salt resolution, a process that is inherently uneconomical due to the theoretical maximum yield of 50% for the desired isomer. Even earlier enzymatic approaches, while offering better stereocontrol, suffered from critically low substrate concentrations, typically below 10g/L. This limitation drastically increases the volume of solvent and water required per kilogram of product, inflating downstream processing costs and reducing the throughput capacity of existing manufacturing infrastructure. These factors collectively create significant vulnerabilities in the supply chain for cost reduction in API manufacturing.
The Novel Approach
The methodology described in patent CN114774382A fundamentally addresses these deficiencies by leveraging a newly identified family of monoamine oxidases sourced from Penicillium species, including Penicillium sp. ART and Penicillium polionium. Unlike their Aspergillus-derived counterparts, these enzymes demonstrate superior stability and activity, allowing for a dramatic increase in substrate loading up to 200g/L without compromising conversion rates or stereoselectivity. This high-concentration capability directly translates to a more compact process footprint and reduced utility consumption. The novel approach streamlines the synthetic route by initiating with a highly selective enzymatic oxidation that establishes the critical chiral centers early in the sequence. By bypassing the need for resolution steps and minimizing the number of unit operations, this strategy offers a streamlined pathway that is inherently more suited for commercial scale-up of complex pharmaceutical intermediates. The result is a process that balances high purity with economic viability.
Mechanistic Insights into Penicillium-Derived Monoamine Oxidase Catalysis
The heart of this technological breakthrough is the asymmetric catalytic oxidation of the saturated amine substrate (Formula II) to the corresponding imine or enamine intermediate (Formula III). The monoamine oxidase (PMAON) facilitates this transformation through a flavin-dependent mechanism where molecular oxygen serves as the terminal electron acceptor. In the presence of the enzyme, the pro-chiral amine undergoes oxidative deamination to form an iminium ion, which subsequently eliminates a proton to yield the chiral alkene product with exquisite stereocontrol. The patent data indicates that this specific enzymatic step yields the (1R,5S)-isomer with 100% ee, effectively setting the stereochemical trajectory for the entire synthesis. This level of precision is difficult to achieve with transition metal catalysts, which often require expensive ligands and rigorous exclusion of air and moisture. The use of a whole-cell or crude enzyme system further simplifies the operational complexity, as the cofactor regeneration is handled internally by the biological system or supplemented catalase.
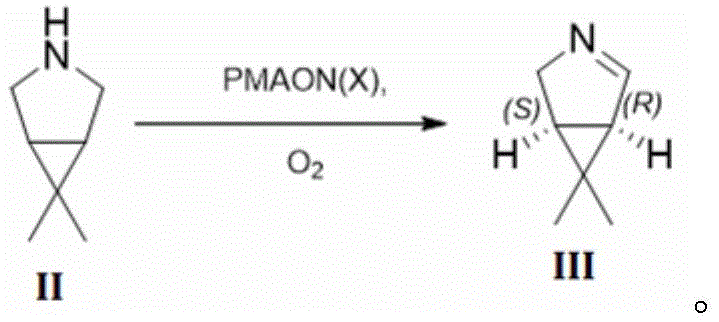
Following the enzymatic oxidation, the resulting chiral building block (Formula III) is seamlessly converted into the final ester product through a cascade of chemical transformations involving bisulfite addition, cyanation, and methanolysis. The robustness of the enzymatic step ensures that the downstream chemical processing begins with a material of exceptionally high optical purity, thereby preventing the propagation of impurities that could complicate final purification. The control of impurities is further enhanced by the specificity of the enzyme, which does not interact with other functional groups that might be present in less refined feedstocks. This orthogonal reactivity profile minimizes the formation of side products such as over-oxidized species or racemic mixtures. Consequently, the impurity profile of the final active pharmaceutical ingredient is significantly cleaner, reducing the burden on quality control laboratories and ensuring consistent batch-to-batch reproducibility. This mechanistic elegance is what makes the process attractive for regulatory filings and long-term commercial production.
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]hexane Efficiently
The implementation of this biocatalytic route requires a coordinated approach involving fermentation technology and downstream chemical processing. The process begins with the cultivation of recombinant E. coli strains harboring the specific PMAON genes, followed by the preparation of a crude enzyme solution that retains high specific activity. The oxidation reaction is conducted in a buffered aqueous medium under controlled pH and temperature conditions to maximize enzyme longevity. Once the bioconversion is complete, the product is extracted and subjected to the subsequent chemical steps to finalize the molecular architecture. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Prepare recombinant E. coli expressing Penicillium monoamine oxidase (PMAON) and cultivate to obtain crude enzyme solution.
- Perform asymmetric oxidation of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane (Formula II) using PMAON and catalase in buffer at pH 5.0-8.0 to yield Formula III.
- Convert Formula III to the final methyl ester hydrochloride (Formula I) via bisulfite addition, cyanation, and methanolysis followed by salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this high-concentration biocatalytic process offers tangible strategic benefits that extend beyond simple technical metrics. The ability to run reactions at 200g/L substrate concentration fundamentally alters the economics of production by drastically reducing the volume of water and solvents that must be heated, cooled, and treated. This reduction in volumetric throughput allows existing manufacturing assets to produce significantly more material in the same amount of time, effectively debottlenecking production capacity without the need for capital-intensive expansion. Furthermore, the elimination of resolution steps removes a major source of yield loss and waste generation, aligning the process with modern sustainability goals and reducing the environmental footprint associated with waste disposal. These factors combine to create a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substantial increase in space-time yield achieved through high substrate loading. By processing 200g/L instead of the conventional 10g/L, the fixed costs associated with reactor occupancy, labor, and energy are amortized over a much larger mass of product. Additionally, the avoidance of chiral resolution agents and the reduction in solvent usage for extraction and crystallization lead to direct savings in raw material expenditures. The simplified workflow also reduces the consumption of auxiliary chemicals and lowers the overall cost of goods sold, providing a competitive pricing advantage in the global market for antiviral intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step synthetic routes that rely on scarce reagents or specialized equipment. This biocatalytic method utilizes recombinant enzymes that can be produced consistently in standard fermentation facilities, ensuring a stable and renewable source of the catalyst. The robustness of the Penicillium-derived enzymes against process variations means that batch failures are less likely, leading to more predictable delivery schedules. For buyers, this translates to reduced risk of stockouts and the ability to secure long-term contracts with confidence, knowing that the manufacturing partner has a technically superior and reliable production platform capable of meeting surging demand.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often challenging due to oxygen transfer limitations, but the high activity of this specific monoamine oxidase mitigates such issues, facilitating a smoother transition from pilot to commercial scale. The process generates significantly less organic waste and saline effluent compared to resolution-based chemical methods, simplifying compliance with increasingly stringent environmental regulations. The reduced E-factor (mass of waste per mass of product) not only lowers disposal costs but also enhances the corporate social responsibility profile of the supply chain. This makes the technology particularly attractive for multinational corporations aiming to green their supplier base while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel biocatalytic technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the maximum substrate concentration achievable with this new biocatalytic method?
A: Unlike prior art methods limited to less than 10g/L, this novel Penicillium-derived monoamine oxidase allows substrate concentrations up to 200g/L, significantly enhancing production efficiency.
Q: How does this method improve chiral purity compared to chemical synthesis?
A: The enzymatic oxidation yields the intermediate (1R,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hex-2-ene with 100% ee, eliminating the need for complex resolution steps required in traditional chemical routes.
Q: Is this process suitable for industrial scale-up?
A: Yes, the high catalytic activity and ability to operate at high substrate loadings make this method specifically designed for industrial mass production, addressing previous yield limitations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]hexane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this biocatalytic method are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for antiviral drug synthesis. Our commitment to quality assurance means that we can navigate the complexities of enzyme handling and downstream processing to deliver a product that is ready for immediate use in your API synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how the high-concentration biocatalysis method compares to your current sourcing strategy in terms of total landed cost and risk mitigation. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to secure a sustainable and efficient supply of this critical pharmaceutical building block.
