Revolutionizing Cefonicid Intermediate Production via Single-Stage Catalytic Condensation
Introduction to Advanced Cefonicid Intermediate Manufacturing
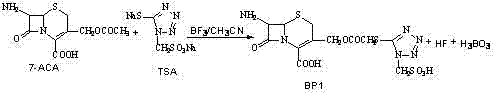
The production of second-generation cephalosporin antibiotics relies heavily on the efficiency and purity of key intermediates, specifically the synthesis of 7-amino-3-[methylsulfonic acid base-1-H-tetrazole-5-sulphur methyl]-3-cephem-4-carboxylic acid, commonly referred to in the industry as BP1. A pivotal advancement in this domain is detailed in Chinese Patent CN102432628A, which discloses a novel single-stage method that fundamentally restructures the synthetic pathway. Unlike traditional multi-step protocols that suffer from low yields and complex purification burdens, this innovation leverages a direct condensation strategy using a boron trifluoride-acetonitrile complex. For R&D directors and process engineers, this patent represents a significant leap forward in process intensification, offering a route that bypasses the notorious bottlenecks associated with boric acid removal. By integrating the reaction and purification into a streamlined sequence, the technology not only enhances the chemical yield to near-theoretical limits but also mitigates the severe equipment corrosion risks inherent in handling hydrofluoric acid byproducts. This report analyzes the technical merits of this approach, positioning it as a cornerstone for reliable pharmaceutical intermediates supplier strategies aiming for cost-effective and scalable antibiotic production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of BP1 has been plagued by the inefficiencies of the two-step method described in prior art such as U.S. Patent 5625058. In this conventional workflow, the condensation of 7-ACA and TSA generates a crude product that is inextricably mixed with substantial quantities of boric acid and hydrofluoric acid resulting from the hydrolysis of the boron catalyst. The subsequent purification requires a dedicated filtration step to remove the boric acid sludge, a process that is technically arduous and operationally hazardous. The presence of hydrofluoric acid creates a highly corrosive environment that aggressively attacks standard stainless steel filtration equipment, leading to frequent maintenance downtime and potential safety incidents. Furthermore, the physical properties of the reaction mixture often lead to premature crystallization of the product within the filter cake, causing significant product entrapment and yield loss. This necessitates a secondary dissolution and recrystallization step, effectively doubling the processing time to approximately 48 hours and introducing multiple opportunities for impurity introduction and material loss.
The Novel Approach
In stark contrast, the methodology outlined in CN102432628A introduces a transformative single-stage protocol that elegantly circumvents these historical obstacles. By optimizing the stoichiometry and reaction conditions, the new process allows for the direct crystallization of high-purity BP1 from the reaction mixture without the need for an intermediate isolation of the crude solid. The key innovation lies in the controlled addition of water post-reaction to fully dissolve the soluble tetrazole byproducts and boric acid species, followed by a precise pH adjustment to induce selective crystallization of the target molecule. This approach effectively keeps the boric acid in the mother liquor, rendering the problematic filtration step entirely obsolete. Consequently, the total processing time is slashed by half, dropping from 48 hours to roughly 24 hours, while the molar yield sees a dramatic escalation from a typical 90% to an impressive 99%. This simplification not only stabilizes the manufacturing process but also significantly lowers the barrier for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into BF3-Catalyzed Condensation
The core of this synthetic breakthrough relies on the precise activation of the cephalosporin nucleus using a Lewis acid catalyst system. The reaction initiates with the formation of a complex between boron trifluoride and acetonitrile (BF3/CH3CN), which serves as a potent electrophilic activator for the 7-ACA substrate. When 7-aminocephalosporanic acid (7-ACA) and 1-methanesulfonic acid-5-mercapto-1,2,3,4-tetrazole (TSA) are introduced into the acetonitrile solvent at 0°C, the boron catalyst facilitates the nucleophilic attack of the tetrazole thiol group onto the C-3 position of the cephem ring. Maintaining the temperature at 0°C during the addition of 3 to 8 moles of the catalyst is critical to suppress potential degradation of the beta-lactam ring, which is sensitive to harsh acidic conditions. As the reaction progresses and warms to 10°C over 4 hours, the thermodynamic drive favors the formation of the stable C-S bond, displacing the acetoxymethyl leaving group. This mechanistic pathway ensures high regioselectivity, minimizing the formation of isomeric impurities that often complicate downstream purification in less controlled environments.
Following the condensation, the workup procedure is designed to exploit the solubility differences between the product and the byproducts. The addition of water post-reaction serves a dual purpose: it hydrolyzes the boron complexes and dissolves the excess TSA and boric acid salts, which remain highly soluble in the aqueous-organic mixture. The subsequent addition of ammonia water to adjust the pH to the narrow range of 2.5 to 3.0 triggers the precipitation of BP1. At this specific acidity, the zwitterionic nature of the cephem carboxylic acid reduces its solubility, causing it to crash out as white crystals, while the impurities remain in solution. This precise pH control is the linchpin of the purification strategy, acting as a chemical switch that separates the product from the reaction matrix without mechanical filtration of solids. This mechanism effectively resolves the issue of product loss due to co-crystallization with boric acid, ensuring that the final HPLC purity exceeds 99.1% directly from the reactor.
How to Synthesize Cefonicid Intermediate BP1 Efficiently
Implementing this single-stage synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process begins by charging equimolar amounts of 7-ACA and a slight excess of TSA into dry acetonitrile, followed by cooling the reactor to 0°C to manage the exotherm upon catalyst addition. The detailed standardized operating procedures, including specific agitation rates and addition profiles for the BF3/CH3CN complex, are critical for maintaining the integrity of the beta-lactam ring. Once the reaction reaches completion, the controlled quenching with water and pH adjustment becomes the defining step for product quality. For process chemists looking to adopt this technology, the following guide outlines the essential operational framework derived from the patent examples, ensuring a seamless transition from laboratory optimization to pilot plant execution.
- Charge 7-ACA and TSA into acetonitrile and cool the mixture to 0°C before adding the BF3/CH3CN complex catalyst.
- Warm the reaction mixture to 10°C and maintain stirring for 4 hours to ensure complete condensation.
- Add water to dissolve reactants, adjust pH to 2.5-3.0 with ammonia water for crystallization, then filter and dry the white crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this single-stage BP1 synthesis offers profound economic and logistical benefits that extend beyond simple yield improvements. The elimination of the intermediate filtration and drying steps translates directly into a reduction of unit operations, which decreases energy consumption and labor requirements per kilogram of output. By removing the need to handle and dispose of large volumes of boric acid filter cakes contaminated with hydrofluoric acid, manufacturers can significantly reduce waste treatment costs and mitigate environmental compliance risks. Furthermore, the drastic reduction in cycle time from 48 hours to 24 hours effectively doubles the throughput capacity of existing reactor assets without requiring capital expenditure on new equipment. This enhanced asset utilization allows for more responsive production scheduling, enabling suppliers to meet fluctuating market demands for cephalosporin antibiotics with greater agility and reliability.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of the boric acid filtration stage. In traditional methods, this step is not only labor-intensive but also necessitates the use of specialized corrosion-resistant materials for filtration equipment due to the presence of hydrofluoric acid. By eliminating this unit operation, the new method removes the associated capital depreciation and maintenance costs of such specialized hardware. Additionally, the increase in molar yield from 90% to 99% implies a substantial reduction in the raw material cost per unit of finished product, as less 7-ACA and TSA are wasted in mother liquors or trapped in filter cakes. This material efficiency, combined with lower utility usage from shortened reaction times, results in a significantly leaner cost structure for API manufacturing.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by equipment failures and extended batch cycles. The corrosive nature of the traditional process frequently leads to unplanned downtime for repairing or replacing damaged filtration units. The new non-corrosive workflow minimizes these mechanical failure points, ensuring more consistent batch-to-batch turnaround times. Moreover, the simplified process flow reduces the dependency on complex auxiliary systems, making the supply chain more robust against operational disruptions. This reliability is crucial for securing long-term contracts with global pharmaceutical companies that require guaranteed delivery schedules for critical antibiotic intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste management challenges, particularly when dealing with hazardous byproducts like hydrofluoric acid and solid boric acid waste. The single-stage method inherently generates less solid waste, simplifying the effluent treatment process and reducing the environmental footprint of the facility. The absence of difficult-to-filter sludge makes the process highly amenable to large-scale continuous or semi-continuous manufacturing setups. This scalability ensures that as demand for cefonicid grows, production can be ramped up efficiently without encountering the bottlenecks associated with solid-liquid separation, thereby supporting sustainable growth in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced BP1 synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing innovation.
Q: How does the single-stage method improve upon traditional BP1 synthesis?
A: The traditional two-step method requires isolating crude BP1 and filtering out large amounts of boric acid byproduct, which causes severe equipment corrosion due to hydrofluoric acid and clogs filters. The new single-stage method eliminates the intermediate filtration step entirely, dissolving the byproducts directly and crystallizing the pure product, thereby increasing yield from 90% to 99%.
Q: What are the critical reaction conditions for this condensation?
A: The reaction utilizes a BF3/CH3CN complex as a Lewis acid catalyst. Critical control points include maintaining the initial temperature at 0°C during catalyst addition to prevent side reactions, followed by a controlled warm-up to 10°C for a 4-hour duration. Final crystallization is achieved by adjusting the pH strictly between 2.5 and 3.0 using ammonia water.
Q: Why is the removal of the boric acid filtration step significant for manufacturing?
A: In conventional processes, the hydrolysis of the boron catalyst generates substantial boric acid and hydrofluoric acid. Filtering this mixture under strong acidic conditions is hazardous, corrosive to stainless steel equipment, and prone to premature crystallization of the product within the filter cake. Eliminating this step drastically reduces maintenance costs and operational hazards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefonicid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the single-stage BP1 process requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102432628A are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to monitor critical parameters such as residual boron levels and isomeric purity, guaranteeing that every batch meets the exacting standards required for downstream antibiotic synthesis. Our commitment to process excellence allows us to deliver high-purity pharmaceutical intermediates that support the global supply of life-saving medications.
We invite pharmaceutical partners to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your production of cefonicid and related cephalosporins is built on a foundation of efficiency, quality, and reliability.
