Advanced Synthesis of Axially Chiral Platinum Complexes for Next-Generation CPOLED Manufacturing
Advanced Synthesis of Axially Chiral Platinum Complexes for Next-Generation CPOLED Manufacturing
The rapid evolution of organic light-emitting diode (OLED) technology has created an urgent demand for advanced emissive materials capable of delivering circularly polarized luminescence (CPL) directly, eliminating the need for external polarizers that significantly reduce device efficiency. Patent CN110669078B introduces a groundbreaking class of metal platinum complexes based on an axially chiral binaphthyl backbone, specifically designed to address the critical bottlenecks in current CPOLED manufacturing. This innovation provides a robust synthetic pathway to produce materials that exhibit both high luminous efficiency and substantial luminescence asymmetry factors (g_lum), making them ideal candidates for next-generation 3D displays and optical storage applications. By leveraging the inherent chirality of commercially available binaphthyl derivatives, this technology circumvents the traditionally expensive and low-yield chiral resolution steps, offering a streamlined route to high-purity optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of CPL-active materials for organic electronics has been hindered by significant synthetic and economic challenges, particularly when relying on helicene-based architectures. Conventional approaches often necessitate complex multi-step syntheses followed by rigorous and costly high-performance liquid chromatography (HPLC) chiral resolution to isolate the desired enantiomer, a process that severely limits scalability and drives up the cost of goods sold. Furthermore, many existing small organic molecules and polymers utilized in this field are fluorescent, meaning they are fundamentally restricted by spin statistics to a maximum internal quantum efficiency of merely 25%, as they cannot utilize triplet excitons for light emission. This intrinsic limitation results in higher power consumption and lower brightness levels, which are unacceptable for high-performance commercial displays. Additionally, the structural rigidity required for strong CPL signals in traditional designs often complicates solution processing, forcing manufacturers to rely on more expensive vacuum deposition techniques.
The Novel Approach
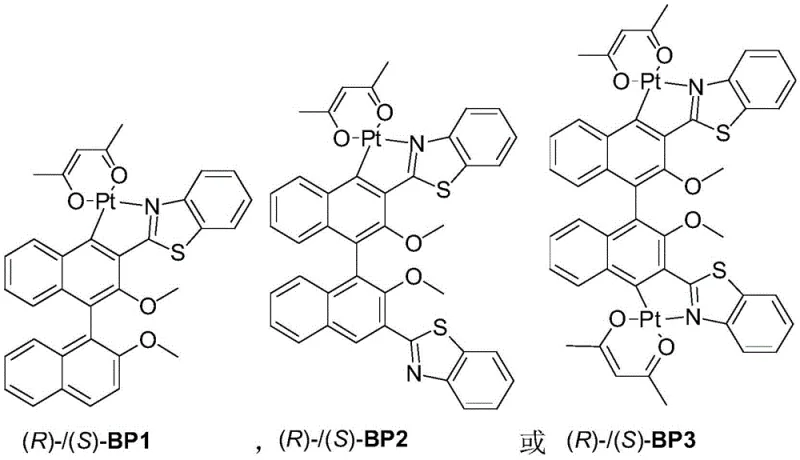
The methodology disclosed in patent CN110669078B represents a paradigm shift by utilizing axially chiral binaphthyl scaffolds that are commercially available in optically pure forms, effectively eliminating the need for post-synthetic chiral resolution. This novel approach employs a concise four-step synthetic sequence that transforms simple binaphthyl precursors into sophisticated square-planar platinum complexes capable of phosphorescent emission. By incorporating heavy platinum atoms, the complex facilitates strong spin-orbit coupling, enabling efficient intersystem crossing and harvesting of triplet excitons to theoretically achieve 100% internal quantum efficiency. The resulting materials, such as BP1, BP2, and BP3, demonstrate excellent solubility and film-forming properties, allowing for fabrication via simple spin-coating techniques which are far more cost-effective than thermal evaporation. This strategic design not only simplifies the supply chain for high-purity OLED material precursors but also ensures that the final devices can achieve high brightness levels exceeding 3500 cd/m² with significant circular polarization characteristics.
Mechanistic Insights into Orthogonal Metallization and Cyclization
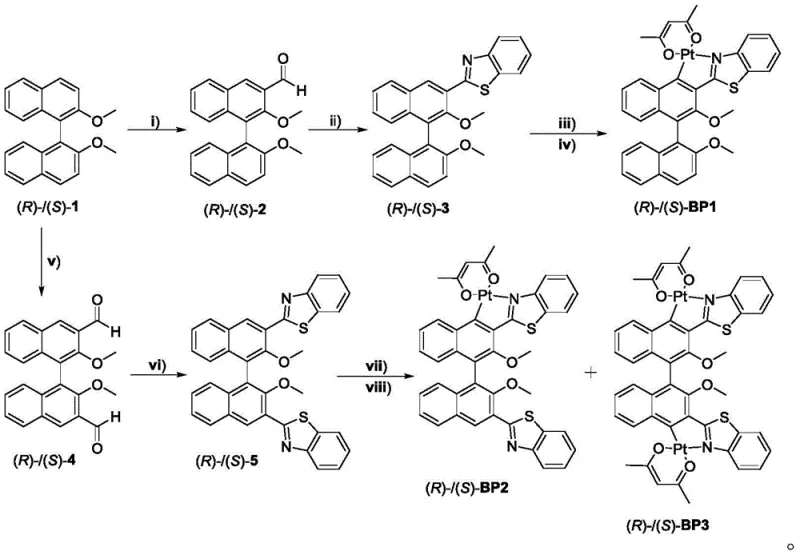
The core chemical transformation in this patent relies on a precise orthogonal metallization strategy where the platinum center is coordinated to the nitrogen of the benzothiazole ring and the carbon of the binaphthyl backbone. The synthesis initiates with the regioselective lithiation of 2,2'-dimethoxy-1,1'-binaphthyl using n-butyllithium at controlled low temperatures between -5°C and 5°C, followed by quenching with DMF to install the crucial aldehyde functionality. This aldehyde intermediate then undergoes a condensation reaction with o-aminothiophenol in the presence of hydrochloric acid and hydrogen peroxide, facilitating the oxidative cyclization to form the benzothiazole heterocycle. This step is critical as it establishes the N,C-chelating ligand framework necessary for stabilizing the platinum center. The subsequent metallation step involves reacting this ligand with potassium chloroplatinite in a mixed solvent system of ethylene glycol monoethyl ether and water at elevated temperatures of 80°C to 110°C. This conditions promote the formation of a chloro-bridged platinum dimer, which serves as a versatile precursor for the final monomeric complex.
Impurity control is meticulously managed throughout the process, particularly during the final ligand exchange where the chloro-bridge is cleaved by sodium acetylacetonate in anhydrous acetone under an inert nitrogen atmosphere. The choice of solvent systems and reaction temperatures is optimized to minimize the formation of homocoupled byproducts or unreacted starting materials, ensuring that the final product meets the stringent purity specifications required for electronic applications. The steric bulk of the methoxy groups on the binaphthyl unit plays a dual role: it prevents detrimental pi-pi stacking interactions that could lead to concentration quenching in the solid state, and it reinforces the chiral environment around the metal center to maximize the g_lum value. For R&D teams focusing on cost reduction in electronic chemical manufacturing, understanding these mechanistic nuances is vital, as slight deviations in the metallation temperature or solvent purity can significantly impact the yield and photophysical properties of the final emissive layer.
How to Synthesize Axially Chiral Platinum Complexes Efficiently
The synthesis protocol outlined in the patent offers a reproducible and scalable method for producing these high-value organometallic emitters, suitable for both laboratory research and pilot-scale production. The process is designed to be operationally simple, avoiding the need for exotic catalysts or extreme pressure conditions, which facilitates easier technology transfer to manufacturing sites. Operators must strictly adhere to the specified molar ratios, particularly the excess of sodium acetylacetonate relative to the chloro-bridge precursor, to ensure complete conversion to the monomeric species. Detailed standard operating procedures regarding the handling of air-sensitive reagents like n-butyllithium and the purification of intermediates via silica gel column chromatography are essential for maintaining batch-to-batch consistency.
- Formylation of 2,2'-dimethoxy-1,1'-binaphthyl using n-butyllithium and DMF at low temperatures (-5 to 5°C) to generate the aldehyde intermediate.
- Condensation of the aldehyde with o-aminothiophenol using hydrochloric acid and hydrogen peroxide in ethanol to form the benzothiazole ligand precursor.
- Cyclometalation reaction with potassium chloroplatinite in ethylene glycol monoethyl ether/water mixture at 80-110°C to form the chloro-bridged dimer.
- Final ligand exchange with sodium acetylacetonate in anhydrous acetone under nitrogen protection to yield the target monomeric platinum complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages by fundamentally altering the cost structure of CPL material production. The most significant benefit arises from the elimination of chiral resolution steps, which traditionally account for a substantial portion of the manufacturing budget due to the low recovery rates and high solvent consumption associated with preparative HPLC. By starting with commercially available optically pure binaphthyls, the process inherently guarantees stereochemical purity without additional purification costs, leading to substantial cost savings in raw material acquisition and processing. Furthermore, the synthetic pathway utilizes commodity chemicals such as ethanol, acetone, and acetic acid, which are readily available in the global market, thereby reducing the risk of supply chain disruptions caused by reliance on specialized or proprietary reagents. This accessibility ensures a more stable and predictable supply of reliable OLED material supplier inputs, allowing for better long-term planning and inventory management.
- Cost Reduction in Manufacturing: The streamlined four-step synthesis significantly reduces the overall operational expenditure by minimizing unit operations and solvent usage compared to traditional multi-step helicene syntheses. The avoidance of expensive chiral columns and the ability to use standard filtration and crystallization techniques for purification further lowers the capital and operational costs. Additionally, the high atom economy of the cyclization reactions ensures that a greater proportion of the starting mass is converted into the final valuable product, reducing waste disposal costs and improving the overall green chemistry profile of the manufacturing process.
- Enhanced Supply Chain Reliability: Utilizing a backbone derived from bulk chemical feedstocks ensures that the production of these complexes is not bottlenecked by the availability of niche chiral auxiliaries. The robustness of the reaction conditions, which tolerate moderate variations in temperature and do not require ultra-high vacuum or cryogenic setups beyond the initial lithiation, makes the process highly transferable across different manufacturing facilities globally. This flexibility allows for the diversification of production sites, mitigating geopolitical risks and ensuring continuous supply for critical commercial scale-up of complex organometallic compounds needed in the display industry.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard reflux and stirring techniques that can be easily adapted from liter-scale reactors to multi-ton vessels. The solvents employed are largely recyclable, and the absence of heavy metal catalysts in the organic transformation steps (the platinum is the product, not a trace catalyst) simplifies the waste stream management. This aligns with increasingly strict environmental regulations in the chemical sector, reducing the compliance burden and facilitating faster regulatory approvals for new material introductions in sensitive markets like consumer electronics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and process feasibility. Understanding these details is crucial for stakeholders evaluating the integration of these materials into their existing product lines or R&D pipelines.
Q: How does this patent overcome the high cost of chiral resolution in CPL material synthesis?
A: The patent utilizes commercially available optically pure 2,2'-dimethoxy-1,1'-binaphthyl as the starting chiral source. This strategy completely bypasses the need for expensive and yield-limiting HPLC chiral separation processes typically required for helicene-based complexes, thereby drastically reducing raw material costs and processing time.
Q: What are the efficiency advantages of these phosphorescent complexes over traditional fluorescent materials?
A: Unlike traditional fluorescent materials which are theoretically limited to a 25% internal quantum efficiency due to singlet exciton utilization, these platinum complexes are phosphorescent. They harvest both singlet and triplet excitons, theoretically allowing for up to 100% internal quantum efficiency, which is critical for developing high-brightness and energy-efficient CPOLED devices.
Q: Is the synthesis process scalable for industrial production of OLED intermediates?
A: Yes, the synthesis involves only four steps with mild reaction conditions ranging from room temperature to 110°C. The use of common solvents like ethanol, acetone, and ethylene glycol monoethyl ether, along with standard reagents like n-butyllithium and potassium chloroplatinite, ensures that the process is highly amenable to commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Platinum Complex Supplier
As the demand for high-performance circularly polarized OLED materials continues to surge, partnering with an experienced chemical manufacturer is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity and trace metal analysis, which are critical for the performance of organic optoelectronic devices. We understand the complexities involved in handling air-sensitive organometallic intermediates and have established robust protocols to maintain product integrity from synthesis to delivery.
We invite you to collaborate with our technical team to explore how this patented synthesis can be optimized for your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to this binaphthyl-based route can improve your margins compared to traditional helicene sources. We encourage potential partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that the transition to these advanced CPL materials is seamless and economically advantageous for your organization.
