Advanced Biocatalytic Route for (S)-N-Boc-3-hydroxypiperidine: Enhancing Purity and Commercial Scalability
Advanced Biocatalytic Route for (S)-N-Boc-3-hydroxypiperidine: Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust and sustainable methodologies for synthesizing chiral intermediates, particularly for complex therapeutic agents. Patent CN109182410B introduces a groundbreaking enzymatic preparation method for (S)-N-Boc-3-hydroxypiperidine, a critical building block widely utilized in the synthesis of analgesics, antipsychotics, and potent antitumor drugs such as Ibrutinib. This technology addresses the longstanding limitations of traditional chemical synthesis by leveraging highly specific carbonyl reductases to achieve exceptional stereoselectivity and yield. By shifting from resolution-based chemical routes to direct asymmetric biocatalysis, manufacturers can secure a more reliable pharmaceutical intermediate supplier status while drastically improving process economics. The innovation lies not just in the enzyme selection but in the holistic integration of cofactor regeneration systems that make the process viable for large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active (S)-N-Boc-3-hydroxypiperidine has relied heavily on chemical synthesis followed by chiral resolution. This conventional approach suffers from inherent inefficiencies, primarily because chemical reduction of the ketone precursor typically yields a racemic mixture. Consequently, manufacturers are forced to employ resolution techniques to isolate the desired (S)-enantiomer, a process that theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is employed, which adds further complexity. Furthermore, the separation and extraction steps associated with resolving racemates are often labor-intensive, require large volumes of organic solvents, and generate significant waste streams. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks for cost reduction in API manufacturing. The reliance on harsh chemical reagents also raises environmental compliance concerns, complicating the regulatory approval process for the final drug substance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a biocatalytic strategy that bypasses the formation of racemic mixtures entirely. By employing specific carbonyl reductases with amino acid sequences corresponding to SEQ ID NO.1 or SEQ ID NO.2, the process achieves direct asymmetric reduction of N-Boc-3-piperidone. This biological catalyst exhibits exquisite chiral selectivity, driving the reaction towards the exclusive formation of the (S)-enantiomer with enantiomeric excess values exceeding 99%. The theoretical yield approaches 100%, effectively doubling the material efficiency compared to traditional resolution methods. Moreover, the reaction conditions are remarkably mild, operating at temperatures between 30°C and 40°C and within a neutral pH range of 6.0 to 9.0. This gentleness not only preserves the integrity of sensitive functional groups but also simplifies the engineering requirements for commercial scale-up of complex pharmaceutical intermediates, ensuring a safer and more controllable production environment.
Mechanistic Insights into Carbonyl Reductase-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the precise molecular recognition and catalytic efficiency of the selected carbonyl reductases. These enzymes, derived from sources such as Novosphingobium and Lactobacillus kefir, possess active sites perfectly configured to bind the N-Boc-3-piperidone substrate in a specific orientation. Upon binding, the enzyme facilitates the transfer of a hydride ion from the reduced cofactor (NADH or NADPH) to the re-face of the carbonyl group. This stereospecific hydride transfer is the determinant step that establishes the (S)-configuration of the resulting hydroxyl group. The high fidelity of this interaction ensures that the competing formation of the (R)-enantiomer is virtually suppressed, resulting in the observed high optical purity. Understanding this mechanism is crucial for R&D directors aiming to optimize reaction parameters, as slight deviations in pH or temperature could alter the enzyme's conformation and potentially impact the stereochemical outcome.
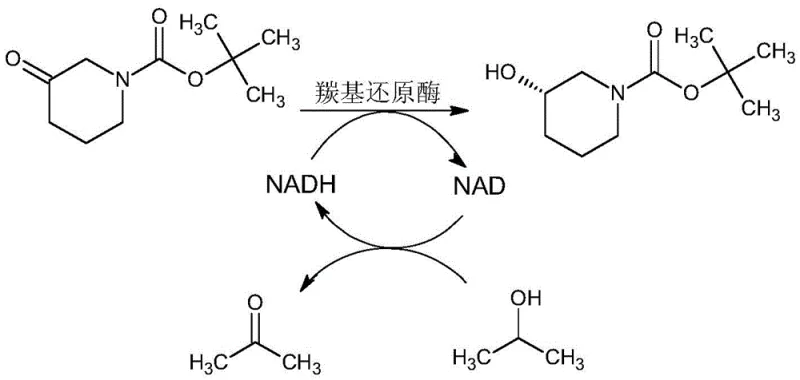
Crucial to the economic viability of this biocatalytic process is the implementation of an efficient cofactor regeneration system, as depicted in the reaction scheme above. Since stoichiometric amounts of NADH or NADPH would be prohibitively expensive for industrial applications, the patent describes a coupled enzyme system where isopropanol serves as the sacrificial hydrogen donor. In this cycle, the carbonyl reductase oxidizes isopropanol to acetone, simultaneously regenerating the reduced cofactor from its oxidized NAD+ or NADP+ form. This closed-loop system allows for the use of catalytic quantities of the cofactor, typically ranging from 0.3wt% to 0.6wt% relative to the substrate. The continuous regeneration maintains a high concentration of the active reducing agent throughout the reaction duration, driving the equilibrium towards product formation. This mechanism not only reduces raw material costs significantly but also simplifies the downstream purification process, as the byproduct acetone is volatile and easily removed, thereby enhancing the overall impurity profile of the high-purity pharmaceutical intermediate.
How to Synthesize (S)-N-Boc-3-hydroxypiperidine Efficiently
Implementing this synthesis route requires careful attention to biocatalyst preparation and reaction control to maximize the benefits of the enzymatic system. The process begins with the fermentation of recombinant E. coli strains engineered to express the specific carbonyl reductase, followed by cell disruption to release the intracellular enzyme. The resulting crude enzyme solution or lyophilized powder is then introduced into a buffered reaction system containing the ketone substrate and the isopropanol regeneration couple. Maintaining the pH within the optimal window of 6.0 to 9.0 using potassium phosphate buffer is essential to sustain enzyme activity over the prolonged reaction period of 20 to 24 hours. Detailed standardized synthetic steps see the guide below.
- Preparation of the biocatalyst involves fermenting recombinant E. coli expressing Carbonyl Reductase (SEQ ID NO.1 or NO.2) in TB medium, followed by cell harvesting and lysis to obtain the active enzyme solution.
- Establish the reaction system by suspending the enzyme in potassium phosphate buffer (pH 6.0-9.0), adding the substrate N-Boc-3-piperidone, isopropanol as the hydrogen donor, and the necessary cofactor (NAD or NADP).
- Maintain the reaction temperature between 30°C and 40°C with stirring for approximately 20-24 hours, followed by extraction with ethyl acetate and crystallization using n-hexane to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic methodology offers profound strategic advantages beyond mere technical superiority. The elimination of chiral resolution steps fundamentally alters the cost structure of the intermediate, removing the massive material loss associated with discarding the unwanted enantiomer. This improvement in atom economy translates directly into substantial cost savings in raw material procurement, allowing for more competitive pricing models without sacrificing margin. Furthermore, the simplified workflow reduces the number of unit operations required, which decreases energy consumption and labor costs associated with multiple separation and purification stages. The ability to source this intermediate from a reliable supplier utilizing such efficient technology ensures greater price stability and protects against market volatility associated with inefficient legacy processes.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic increase in yield efficiency. By achieving near-quantitative conversion with high stereoselectivity, the requirement for starting materials is effectively halved compared to resolution-based routes. Additionally, the use of isopropanol as a dual-purpose solvent and reducing agent eliminates the need for specialized, expensive chemical reducing agents like borohydrides or aluminum hydrides, which often require stringent safety measures and generate hazardous waste. The removal of heavy metal catalysts or harsh acidic/basic conditions further reduces the cost burden related to waste treatment and environmental compliance, leading to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step chemical syntheses that are prone to bottlenecks. This biocatalytic route streamlines the production timeline by consolidating multiple steps into a single, robust transformation. The mild reaction conditions reduce the risk of equipment corrosion and safety incidents, minimizing unplanned downtime. Moreover, the enzymes used are produced via fermentation, a highly scalable and consistent biological process that is less susceptible to the supply chain disruptions common with petrochemical-derived reagents. This reliability ensures that downstream API manufacturers can maintain consistent production schedules, reducing lead time for high-purity pharmaceutical intermediates and securing their own supply chains against external shocks.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is inherently safer and more straightforward than scaling exothermic chemical reductions. The reaction operates at ambient pressure and moderate temperatures, reducing the engineering controls needed for heat dissipation and pressure containment. From an environmental perspective, the process aligns perfectly with green chemistry principles by utilizing renewable biocatalysts and generating benign byproducts like acetone. The aqueous nature of the reaction medium significantly reduces the volume of organic solvents required, lowering the facility's carbon footprint and simplifying the permitting process for capacity expansion. This environmental compatibility is increasingly becoming a prerequisite for partnerships with top-tier global pharmaceutical companies focused on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Stakeholders can use this information to assess the feasibility of integrating this route into their existing supply networks or R&D pipelines.
Q: What are the advantages of this enzymatic method over traditional chemical resolution?
A: Unlike chemical synthesis which often produces racemic mixtures requiring complex and yield-limiting resolution steps, this enzymatic method utilizes specific carbonyl reductases to achieve theoretical 100% yield and >99% ee directly, significantly simplifying downstream processing.
Q: How is the cofactor regenerated in this process to ensure cost efficiency?
A: The process employs an efficient cofactor regeneration system where isopropanol serves as both a solvent and a hydrogen donor. It is oxidized to acetone while regenerating the reduced cofactor (NADH/NADPH) from its oxidized form, eliminating the need for stoichiometric amounts of expensive cofactors.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (30°C-40°C, neutral pH) using robust recombinant enzymes derived from sources like Novosphingobium or Lactobacillus kefir, making it highly scalable, safe, and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-N-Boc-3-hydroxypiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in Patent CN109182410B for producing high-value chiral intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical enantiomeric excess values required for oncology and CNS drug applications. We are committed to delivering not just a chemical product, but a guaranteed supply solution that meets the exacting standards of the global pharmaceutical industry.
We invite forward-thinking procurement leaders and R&D directors to collaborate with us to leverage this advanced biocatalytic technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest levels of quality, efficiency, and supply chain security available in the market.
