Scalable Chemical Synthesis of (S)-N-BOC-3-hydroxypiperidine for Industrial API Manufacturing
The pharmaceutical industry continuously seeks robust, scalable pathways for chiral intermediates, and patent CN110759853B presents a significant advancement in the synthesis of (S)-N-BOC-3-hydroxypiperidine. This vital building block is extensively utilized in the manufacture of analgesics, antipsychotics, and antitumor agents such as ibrutinib. Unlike traditional biocatalytic approaches that rely on fragile enzyme systems, this invention details a purely chemical synthetic route that leverages readily available starting materials like (S)-epichlorohydrin. The methodology addresses critical pain points in industrial chemistry by eliminating the dependency on expensive biological catalysts while maintaining high stereochemical integrity throughout the transformation. By shifting from enzymatic reduction to a sequence involving Grignard addition and controlled cyclization, the process offers a more predictable and controllable manufacturing profile. This transition is particularly valuable for supply chain managers who require consistent quality and uninterrupted raw material flow for long-term API production campaigns without the volatility associated with biotech sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active (S)-N-BOC-3-hydroxypiperidine has heavily relied on biocatalytic asymmetric reduction of ketones or resolution of racemic mixtures. While enzymatic methods using carbonyl reductases can achieve high enantiomeric excess, they suffer from inherent limitations regarding enzyme stability, cofactor regeneration costs, and the high price of specialized biocatalysts. Furthermore, the downstream processing in biocatalytic routes often involves complex separation steps to remove proteins and cellular debris, which increases waste generation and reduces overall throughput. The reliance on specific microbial strains also introduces supply chain risks, as fermentation batches can vary, leading to inconsistencies in catalytic activity. These factors collectively drive up the cost of goods sold and complicate the regulatory validation process for commercial manufacturing, making purely chemical alternatives increasingly attractive for large-scale operations seeking to de-risk their supply chains.
The Novel Approach
The novel approach disclosed in the patent circumvents these biological constraints by employing a concise four-step chemical synthesis that begins with a Grignard reaction. This strategy utilizes inexpensive, commodity-grade reagents such as 2-chloroethyl magnesium bromide and (S)-epichlorohydrin, which are produced on a massive industrial scale globally. The route is designed to minimize side reactions through precise temperature control and stoichiometric optimization, ensuring that the chiral center established in the starting material is preserved throughout the sequence. By avoiding the need for chiral resolution or enzymatic kinetic resolution, the theoretical yield is significantly improved, and the process becomes far more amenable to continuous flow or large-batch reactor processing. This chemical robustness translates directly into operational efficiency, allowing manufacturers to utilize standard stainless steel equipment rather than specialized bioreactors, thereby simplifying facility requirements and reducing capital expenditure for new production lines.
Mechanistic Insights into Grignard Addition and Cyclization Cascade
The core of this synthetic innovation lies in the initial nucleophilic attack of the Grignard reagent on the epoxide ring of (S)-epichlorohydrin. In this critical first step, the 2-chloroethyl magnesium bromide acts as a carbon nucleophile, selectively opening the strained three-membered ring at the less hindered position to extend the carbon chain while retaining the chlorine handle for future cyclization. This reaction must be conducted under strictly anhydrous conditions at low temperatures, typically between -78°C and -10°C, to prevent competing side reactions such as Wurtz coupling or protonation of the organometallic species. The resulting intermediate, (S)-1,5-dichloro-2-pentanol, serves as a versatile precursor that contains both the necessary carbon backbone and the functional groups required for subsequent ring closures. The stereochemistry is rigorously maintained during this exothermic addition, setting the foundation for the high optical purity observed in the final piperidine product.
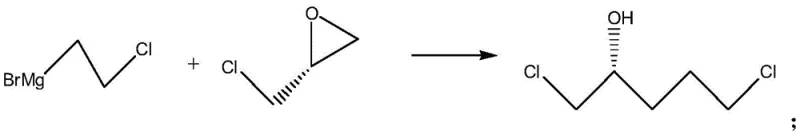
Following the chain extension, the synthesis proceeds through a sophisticated cascade of intramolecular cyclizations driven by basic conditions and ammonolysis. The second step involves treating the dichloro-alcohol with a base to form an epoxide intermediate via internal substitution, effectively activating the molecule for nitrogen insertion. Subsequently, reaction with ammonia solution under pressure facilitates the opening of this new epoxide and a second intramolecular nucleophilic substitution where the amine attacks the terminal chloride. This dual cyclization mechanism efficiently constructs the six-membered piperidine ring in a single pot operation after the ammonolysis, demonstrating high atom economy. The final protection step with di-tert-butyl dicarbonate (BOC anhydride) stabilizes the amine, yielding the target intermediate ready for downstream API synthesis. This mechanistic pathway avoids the use of heavy metal catalysts or toxic reagents, aligning with modern green chemistry principles while delivering exceptional purity profiles.
How to Synthesize (S)-N-BOC-3-hydroxypiperidine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale execution, emphasizing strict control over reaction parameters to maximize yield and safety. The process begins with the preparation of the Grignard reagent, followed by its controlled addition to the chiral epoxide, requiring careful thermal management to handle the exotherm. Subsequent steps involve base-mediated cyclization and high-pressure ammonolysis, which necessitate appropriate reactor ratings and safety interlocks. The final crystallization from ethyl acetate and petroleum ether ensures the removal of trace impurities and isomers. For detailed operational procedures, including specific solvent volumes, stirring rates, and quenching protocols, please refer to the standardized guide below which breaks down the exact experimental conditions for reproducible results.
- React (S)-epichlorohydrin with 2-chloroethyl magnesium bromide to form (S)-1,5-dichloro-2-pentanol.
- Perform intramolecular ring closure with base to generate (S)-5-chloro-1,2-epoxypentane.
- React with ammonia solution to form the piperidine ring, followed by BOC anhydride protection.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this chemical route offers substantial advantages by decoupling production from the volatile biotechnology sector. The primary raw materials, including epichlorohydrin and simple alkyl halides, are petrochemical derivatives available from multiple global suppliers, ensuring a competitive pricing environment and reducing the risk of single-source dependency. By eliminating the need for proprietary enzymes and cofactor recycling systems, the manufacturing cost structure is significantly simplified, removing the premium pricing often associated with biocatalytic processes. This shift allows procurement managers to negotiate better terms based on commodity chemical indices rather than specialized biotech contracts, leading to more predictable budgeting and reduced cost of goods sold over the lifecycle of the drug product.
- Cost Reduction in Manufacturing: The elimination of expensive biocatalysts and the associated fermentation infrastructure drastically lowers the fixed and variable costs of production. Without the need for sterile environments, complex media preparation, or enzyme purification, the operational expenditure is reduced, allowing for more competitive pricing of the final intermediate. Furthermore, the use of standard organic solvents like tetrahydrofuran and dichloromethane, which are easily recovered and recycled, minimizes waste disposal costs and enhances the overall economic efficiency of the plant.
- Enhanced Supply Chain Reliability: Relying on stable, shelf-stable chemical reagents rather than perishable biological agents ensures consistent supply continuity regardless of seasonal or biological variations. The robustness of the chemical synthesis means that production schedules can be tightly adhered to without the risk of batch failures due to enzyme deactivation or contamination. This reliability is crucial for just-in-time manufacturing models used by major pharmaceutical companies, where any delay in intermediate delivery can halt entire API production lines and impact market availability.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as distillation and crystallization that are well-understood and easily expanded from kilogram to multi-ton scales. The absence of biological waste streams simplifies effluent treatment, as the waste consists primarily of organic solvents and salts that can be managed through standard incineration or recovery systems. This alignment with environmental regulations reduces the permitting burden and facilitates faster technology transfer to commercial manufacturing sites, accelerating time-to-market for new drug candidates utilizing this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical disclosures within the patent documentation to provide clarity on process capabilities and product quality. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific supply chain requirements.
Q: Why is this chemical route preferred over enzymatic methods for (S)-N-BOC-3-hydroxypiperidine?
A: Enzymatic methods often require expensive, specialized ketoreductases and coenzyme systems with limited supply stability. This chemical route utilizes commodity reagents like epichlorohydrin and Grignard reagents, ensuring consistent availability and significantly lower raw material costs for large-scale production.
Q: What are the critical purity specifications achievable with this synthesis?
A: The patent data indicates that through optimized distillation and crystallization steps, the final product can achieve purity levels exceeding 99%, with high enantiomeric excess retained from the chiral starting material, meeting stringent requirements for API intermediates.
Q: Is this process suitable for multi-ton commercial manufacturing?
A: Yes, the process avoids complex biocatalytic fermentation steps and uses standard chemical reactors capable of handling Grignard reactions and high-pressure ammonolysis, making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual capacity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-N-BOC-3-hydroxypiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of (S)-N-BOC-3-hydroxypiperidine meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence ensures that clients receive materials with consistent chirality and minimal impurity profiles, supporting robust regulatory filings.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this chemical methodology. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a secure and cost-effective supply of this essential pharmaceutical building block for your upcoming campaigns.
