Advanced CDI-Mediated Synthesis of Metamifop: A Technical Breakthrough for Commercial Scale-Up
The agrochemical industry is constantly seeking more efficient and environmentally benign pathways for producing high-value herbicides like Metamifop. A significant technological advancement in this domain is detailed in patent CN111574470B, which discloses a novel synthesis method utilizing carbonyldiimidazole (CDI) as a condensation reagent. This approach addresses critical pain points in traditional manufacturing, such as harsh reaction conditions, low yields, and the generation of hazardous waste. By shifting from conventional acyl chlorination or phosphine-mediated couplings to a mild, CDI-activated amide formation, manufacturers can achieve superior purity profiles while maintaining the critical optical integrity of the chiral center. This report analyzes the technical merits of this innovation and its implications for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Metamifop has relied on several distinct synthetic routes, each plagued by significant operational and environmental drawbacks. One common traditional pathway involves the acyl chlorination of the precursor carboxylic acid followed by substitution with N-methyl o-fluoroaniline. While chemically feasible, this method often requires aggressive reagents that pose safety risks and can lead to product decomposition due to the sensitivity of the benzoxazole moiety under acidic conditions. Another legacy method utilizes triphenylphosphine-based coupling systems, which are notoriously inefficient for this specific transformation.
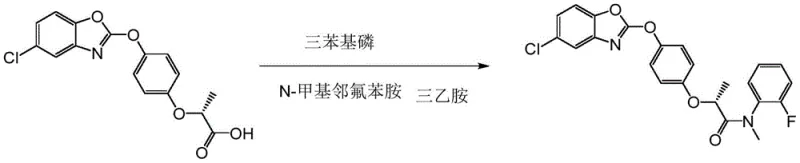
As illustrated in the reaction scheme above, the triphenylphosphine route suffers from abysmal reaction yields, reported to be as low as 23% in some contexts, alongside the generation of substantial phosphorus-containing wastewater that complicates environmental compliance. Furthermore, alternative strategies involving dehydrogenation with sodium hydride followed by nucleophilic substitution with methyl iodide introduce severe safety hazards due to the pyrophoric nature of NaH and the toxicity of alkylating agents. These conventional processes collectively result in high production costs, complex purification requirements, and inconsistent product quality, creating a pressing need for process intensification.
The Novel Approach
The methodology described in patent CN111574470B represents a paradigm shift by employing CDI (N,N'-carbonyldiimidazole) as the primary activating agent in conjunction with a specialized catalyst system. This route bypasses the formation of unstable acid chlorides and avoids the use of heavy metal or phosphine reagents entirely. The reaction proceeds under nearly neutral conditions, which is crucial for preventing the hydrolysis of the final amide product. By operating at mild temperatures ranging from 0°C to 50°C, the process ensures thermal stability and minimizes energy consumption. The use of ImTfOH (imidazole trifluoromethanesulfonate) as a catalyst further refines the reaction environment, acting as an organic salt that buffers the system against extreme acidity.
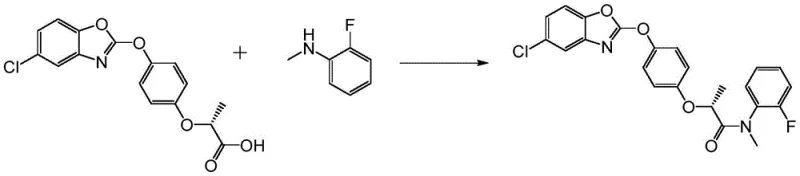
This novel approach not only simplifies the operational workflow by eliminating dangerous steps but also dramatically improves the mass balance of the synthesis. The direct coupling of (R)-2-(4-((6-chlorobenzoxazole)oxy)phenoxy)propionic acid with N-methyl o-fluoroaniline allows for precise stoichiometric control, typically utilizing a molar ratio close to 1:1. The result is a robust process capable of delivering high-purity Metamifop with excellent yields, effectively resolving the inefficiencies inherent in the older acyl chloride and phosphine-based methodologies.
Mechanistic Insights into CDI-Mediated Amide Coupling
The core of this technological breakthrough lies in the mechanism of CDI activation and the subsequent role of the ImTfOH catalyst. Initially, CDI reacts with the carboxylic acid group of the phenoxypropionic acid precursor to form a highly reactive acyl imidazole intermediate. This intermediate is significantly more electrophilic than the parent acid but remains stable enough to be handled under controlled conditions without rapid degradation. The presence of the ImTfOH catalyst facilitates the nucleophilic attack by the aniline nitrogen on the activated carbonyl carbon. Unlike strong Brønsted acids like TfOH, which can protonate the aniline nucleophile and retard the reaction or catalyze side reactions, the organic salt nature of ImTfOH maintains a balanced proton activity.
This mechanistic nuance is vital for impurity control, particularly regarding the preservation of chirality. The (R)-configuration at the alpha-carbon of the propionic acid chain is essential for the herbicidal activity of Metamifop. Harsh acidic or basic conditions often promote racemization via enolization of the alpha-proton. However, the nearly neutral environment provided by the CDI/ImTfOH system suppresses this pathway. Analytical data from the patent indicates that the enantiomeric excess (ee) of the starting material is quantitatively transferred to the product, with values remaining consistently around 99.6%. This stereochemical fidelity ensures that the final agrochemical product meets the stringent biological efficacy standards required for rice field applications without the need for costly chiral resolution steps post-synthesis.
How to Synthesize Metamifop Efficiently
Implementing this synthesis route requires careful attention to reagent addition rates and temperature control to maximize the formation of the acyl imidazole intermediate before introducing the amine. The process is designed to be scalable, utilizing common organic solvents such as dichloromethane or acetonitrile which facilitate easy workup procedures. Operators must ensure that the CDI is added in portions to manage the exotherm associated with the activation step, followed by the controlled addition of the aniline component to drive the coupling to completion. For a comprehensive understanding of the specific operational parameters and safety protocols, please refer to the standardized synthesis guide below.
- Dissolve (R)-2-(4-((6-chlorobenzoxazole)oxy)phenoxy)propionic acid in dichloromethane and add CDI and ImTfOH catalyst in portions at 20°C.
- Prepare a solution of N-methyl o-fluoroaniline in solvent and slowly drip it into the activated acid mixture while maintaining temperature control.
- Stir the reaction to completion, extract the organic phase with water, remove the solvent, and isolate the high-purity Metamifop product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this CDI-mediated synthesis offers tangible strategic benefits beyond mere chemical elegance. The elimination of hazardous reagents like thionyl chloride or triphenylphosphine translates directly into reduced regulatory burdens and lower waste disposal costs. Traditional methods often require extensive downstream processing to remove phosphorus residues or neutralize acidic byproducts, which adds time and expense to the manufacturing cycle. By contrast, the new method generates imidazole as a byproduct, which is easier to separate and less environmentally toxic, thereby streamlining the purification train and reducing the overall cost of goods sold.
- Cost Reduction in Manufacturing: The shift to a high-yield coupling reaction significantly improves atom economy and reduces the consumption of expensive starting materials. By avoiding the low-yield triphenylphosphine route, manufacturers can drastically cut down on raw material waste. Furthermore, the mild reaction conditions reduce energy demands for heating or cooling, contributing to substantial operational expenditure savings. The simplified workup procedure also lowers labor costs and solvent usage, making the overall production economics far more favorable compared to legacy processes.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically the chiral acid precursor and N-methyl o-fluoroaniline, are commercially mature and widely available from established suppliers. This reduces the risk of supply bottlenecks that often plague processes relying on specialized or hazardous reagents. The robustness of the reaction means that production schedules are less likely to be disrupted by batch failures or quality deviations, ensuring a steady flow of high-purity agrochemical intermediates to meet seasonal demand in the farming sector.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns with modern green chemistry principles by minimizing the generation of hazardous waste streams. The absence of heavy metals and phosphorus compounds simplifies wastewater treatment, allowing facilities to meet increasingly strict environmental regulations with greater ease. The process has been demonstrated to be scalable from laboratory to industrial quantities without loss of efficiency, providing a reliable pathway for commercial scale-up of complex herbicide intermediates while maintaining a smaller environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, offering clarity on process capabilities and product specifications. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: Why is ImTfOH preferred over TfOH as a catalyst in this synthesis?
A: ImTfOH (imidazole trifluoromethanesulfonate) is an organic salt that maintains a nearly neutral reaction system. Unlike strong acid TfOH, it prevents the partial hydrolysis of the sensitive Metamifop product, ensuring higher stability and yield.
Q: Does this CDI-mediated process affect the optical purity of the product?
A: No, the process is designed to be mild and non-racemizing. The patent data confirms that the ee value remains consistent (e.g., 99.6%) throughout the reaction, preserving the critical stereochemistry required for herbicidal activity.
Q: What are the environmental benefits compared to traditional acyl chloride methods?
A: This method eliminates the need for hazardous acyl chlorination reagents and avoids the generation of phosphorus-containing wastewater associated with triphenylphosphine routes, significantly reducing three-waste treatment burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metamifop Supplier
The synthesis method disclosed in CN111574470B represents a significant opportunity for optimizing the production of high-value herbicide intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Metamifop or related intermediate meets the exacting standards required by global agrochemical formulators.
We invite potential partners to engage with our technical team to explore how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient methodology. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your volume requirements.
