Advanced Catalytic Synthesis of (S)-2-chloro-1-(3,4-difluorophenyl)ethanol for Industrial Scale-up
Advanced Catalytic Synthesis of (S)-2-chloro-1-(3,4-difluorophenyl)ethanol for Industrial Scale-up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for the production of critical chiral intermediates. A significant breakthrough in this domain is detailed in patent CN107641072B, which discloses a novel method for preparing (S)-2-chloro-1-(3,4-difluorophenyl)ethanol, a key building block for the antiplatelet agent Ticagrelor. This compound, characterized by its specific stereochemistry at the benzylic position, serves as a pivotal precursor in the assembly of complex cardiovascular therapeutics. The disclosed technology leverages advanced transition metal catalysis to achieve high enantioselectivity while circumventing the severe safety and cost limitations associated with legacy synthetic routes. By employing a sophisticated chiral metal complex in conjunction with mild hydrogen donors, this process delivers optically pure products with exceptional efficiency.
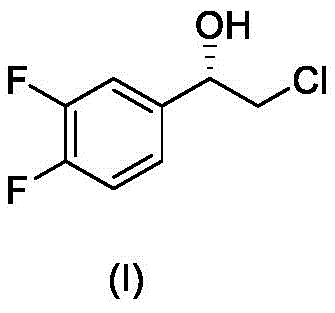
For procurement specialists and supply chain managers, the implications of this technological shift are profound. The ability to source high-purity pharmaceutical intermediates through a streamlined, low-toxicity process translates directly into enhanced supply chain reliability and reduced regulatory burden. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such process innovations allows for better risk assessment and long-term strategic planning. The method described not only ensures the consistent quality required for GMP manufacturing but also aligns with global trends towards greener chemistry, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent purity specifications demanded by top-tier drug developers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial preparation of (S)-2-chloro-1-(3,4-difluorophenyl)ethanol relied heavily on asymmetric reduction strategies utilizing borane-based reagents. Specifically, existing literature and patent applications, such as CN200780028775.8, describe the use of (S)-diphenylprolinol complexes with trimethyl borate and borane dimethyl sulfide as the reducing agent. While chemically feasible, this conventional approach presents substantial drawbacks for large-scale operations. The primary concern is the inherent toxicity and unpleasant odor of borane dimethyl sulfide, which necessitates specialized containment equipment and rigorous waste treatment protocols, thereby inflating operational expenditures. Furthermore, the chiral ligand (S)-diphenylprolinol is required in large stoichiometric quantities relative to the substrate, driving up raw material costs significantly. These factors collectively create a bottleneck for commercial scale-up of complex chiral alcohols, limiting the agility of manufacturers to respond to market demand.
The Novel Approach
In stark contrast, the method disclosed in CN107641072B introduces a paradigm shift by utilizing catalytic hydrogen transfer reduction. This innovative route employs a chiral metal catalyst, specifically complexes of Rhodium or Iridium defined by Formula (III), paired with inexpensive and safe hydrogen donors such as formic acid/triethylamine azeotropes or sodium formate. 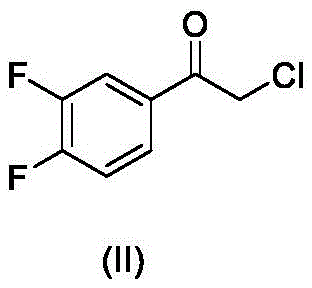 This substitution of hazardous boranes with benign carboxylic acid derivatives fundamentally alters the safety profile of the reaction, eliminating the need for pyrophoric reagent handling. The process operates under mild conditions, typically between 20°C and 75°C, and tolerates a variety of solvents including toluene and dichloromethane. By decoupling the reduction potential from dangerous hydride sources, this novel approach facilitates a smoother workflow, reduces the environmental footprint, and offers a clear pathway for reducing lead time for high-purity pharmaceutical intermediates in a commercial setting.
This substitution of hazardous boranes with benign carboxylic acid derivatives fundamentally alters the safety profile of the reaction, eliminating the need for pyrophoric reagent handling. The process operates under mild conditions, typically between 20°C and 75°C, and tolerates a variety of solvents including toluene and dichloromethane. By decoupling the reduction potential from dangerous hydride sources, this novel approach facilitates a smoother workflow, reduces the environmental footprint, and offers a clear pathway for reducing lead time for high-purity pharmaceutical intermediates in a commercial setting.
Mechanistic Insights into Asymmetric Transfer Hydrogenation
The core of this technological advancement lies in the precise design of the chiral catalyst system. The catalyst, represented generally by Formula (III), features a pentamethylcyclopentadienyl (Cp*) metal center coordinated with a chiral diamine ligand derived from diphenylethylenediamine. 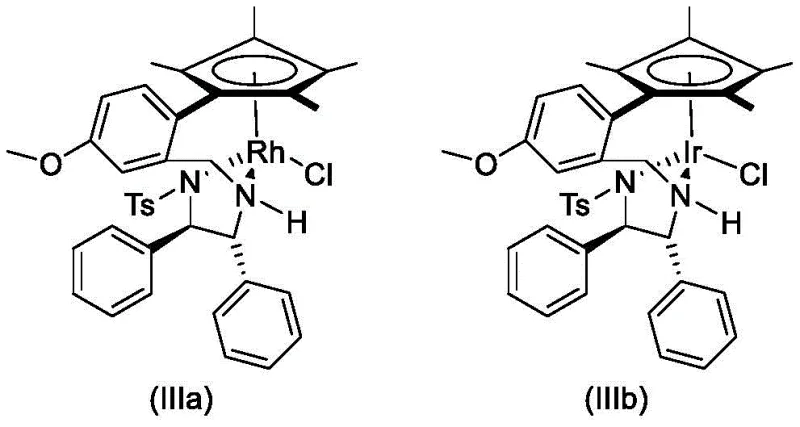 In this coordination sphere, the metal atom (M = Rh or Ir) acts as the active site for hydride transfer. The chirality of the diamine ligand creates a rigid, sterically defined environment around the metal center, which dictates the facial selectivity of the hydride attack on the prochiral ketone substrate. When the ketone coordinates to the metal-hydride species formed in situ from the formic acid donor, the bulky phenyl groups on the ligand shield one face of the carbonyl group, forcing the hydride to attack from the opposite side. This steric control is the mechanistic basis for the high enantiomeric excess (ee) observed, consistently exceeding 97% in experimental trials. The presence of the tosyl (Ts) group on the nitrogen further modulates the electronic properties of the ligand, stabilizing the catalytic cycle and preventing decomposition.
In this coordination sphere, the metal atom (M = Rh or Ir) acts as the active site for hydride transfer. The chirality of the diamine ligand creates a rigid, sterically defined environment around the metal center, which dictates the facial selectivity of the hydride attack on the prochiral ketone substrate. When the ketone coordinates to the metal-hydride species formed in situ from the formic acid donor, the bulky phenyl groups on the ligand shield one face of the carbonyl group, forcing the hydride to attack from the opposite side. This steric control is the mechanistic basis for the high enantiomeric excess (ee) observed, consistently exceeding 97% in experimental trials. The presence of the tosyl (Ts) group on the nitrogen further modulates the electronic properties of the ligand, stabilizing the catalytic cycle and preventing decomposition.
From an impurity control perspective, this mechanism offers distinct advantages over stoichiometric reductions. Because the catalyst operates in a true cyclic manner with turnover numbers (TON) exceeding 1000, the residual metal content in the final product can be managed more effectively than in processes requiring stoichiometric chiral auxiliaries. The reaction proceeds through a well-defined six-membered transition state involving the metal, the hydride, the proton from the amine ligand, and the carbonyl oxygen. This concerted mechanism minimizes side reactions such as over-reduction or dehalogenation, which are common pitfalls when using aggressive reducing agents like boranes on chlorinated substrates. Consequently, the crude product profile is cleaner, simplifying downstream purification and ensuring that the final API intermediate meets the rigorous impurity thresholds required for cardiovascular drug synthesis.
How to Synthesize (S)-2-chloro-1-(3,4-difluorophenyl)ethanol Efficiently
The practical implementation of this synthesis is designed for scalability and ease of operation, making it highly suitable for contract development and manufacturing organizations (CDMOs). The general procedure involves charging a reactor with the ketone substrate, the pre-formed or in-situ generated chiral catalyst, and a suitable organic solvent. The hydrogen donor system is then introduced, often as a premixed solution to ensure homogeneity. The reaction is allowed to proceed under thermal conditions until conversion is deemed complete by TLC or HPLC analysis. Following the reaction, a simple aqueous workup quenches the process, allowing for phase separation and isolation of the product. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Charge the reactor with 2-chloro-1-(3,4-difluorophenyl)ethanone, a chiral Rh or Ir catalyst (Formula III), and an organic solvent such as toluene or dichloromethane.
- Add the hydrogen donor system, typically a premixed solution of formic acid and triethylamine or an aqueous sodium formate solution, to the reaction mixture under stirring.
- Heat the mixture to reflux or maintain at 30-40°C until conversion is complete, then quench with water, separate layers, and purify the organic phase to obtain the optically pure alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the transition to this catalytic transfer hydrogenation method offers compelling economic and logistical benefits. The elimination of expensive and hazardous borane reagents removes a significant cost driver and a major safety liability from the manufacturing process. Furthermore, the drastic reduction in catalyst loading—achieving effective turnover with substrate-to-catalyst ratios of 1000:1 or higher—means that the consumption of precious metals and complex chiral ligands is minimized. This efficiency directly contributes to substantial cost savings in raw material procurement, allowing for more competitive pricing models for the final intermediate without sacrificing margin.
- Cost Reduction in Manufacturing: The replacement of stoichiometric chiral inducers and expensive borane reducing agents with catalytic amounts of metal complexes and cheap formic acid derivatives results in a leaner cost structure. The process avoids the need for specialized infrastructure required to handle pyrophoric materials, thereby lowering capital expenditure and maintenance costs associated with safety systems. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal fees, further enhancing the overall economic viability of the production campaign.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like formic acid, triethylamine, and sodium formate, the process mitigates the risk of supply disruptions often associated with specialty reagents. The robustness of the catalyst system ensures consistent batch-to-batch quality, reducing the likelihood of failed runs that could delay downstream API synthesis. This reliability is crucial for maintaining uninterrupted supply chains for critical medications like Ticagrelor, ensuring that pharmaceutical partners can meet their own production schedules without interruption.
- Scalability and Environmental Compliance: The mild reaction conditions and low toxicity profile of the reagents make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The absence of noxious sulfur-containing byproducts simplifies废气 (exhaust gas) treatment and wastewater management, facilitating compliance with increasingly stringent environmental regulations. This 'green' attribute not only reduces regulatory risk but also aligns with the sustainability goals of modern pharmaceutical companies, making the supplier a more attractive partner for long-term collaborations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the process capabilities. Understanding these details helps R&D and procurement teams evaluate the feasibility of adopting this technology for their specific supply chain needs.
Q: What is the optical purity achieved with this new catalytic method?
A: The patented process consistently achieves high optical purity, with enantiomeric excess (ee) values ranging from 97.5% to 98.5%, meeting the stringent requirements for Ticagrelor synthesis.
Q: How does this method improve upon traditional borane reduction?
A: Unlike traditional methods using toxic and odorous borane dimethyl sulfide, this process utilizes benign formic acid/triethylamine or sodium formate systems, significantly lowering toxicity and operational hazards.
Q: What catalyst loading is required for effective conversion?
A: The process is highly efficient, operating effectively with substrate-to-catalyst molar ratios of at least 1000:1, drastically reducing the consumption of expensive chiral metal complexes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-chloro-1-(3,4-difluorophenyl)ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes for high-value pharmaceutical intermediates. Our technical team has thoroughly analyzed the methodology described in CN107641072B and possesses the expertise to implement this advanced catalytic transfer hydrogenation process at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical purity required for Ticagrelor synthesis.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. By leveraging our technical proficiency and capacity, you can secure a stable source of high-quality material while benefiting from the cost and safety advantages of this modern synthetic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your project timelines and regulatory filings.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
