Revolutionizing Drospirenone Production: A Heavy-Metal-Free Oxidation Strategy for Commercial Scale
Introduction to Patent CN110407905B
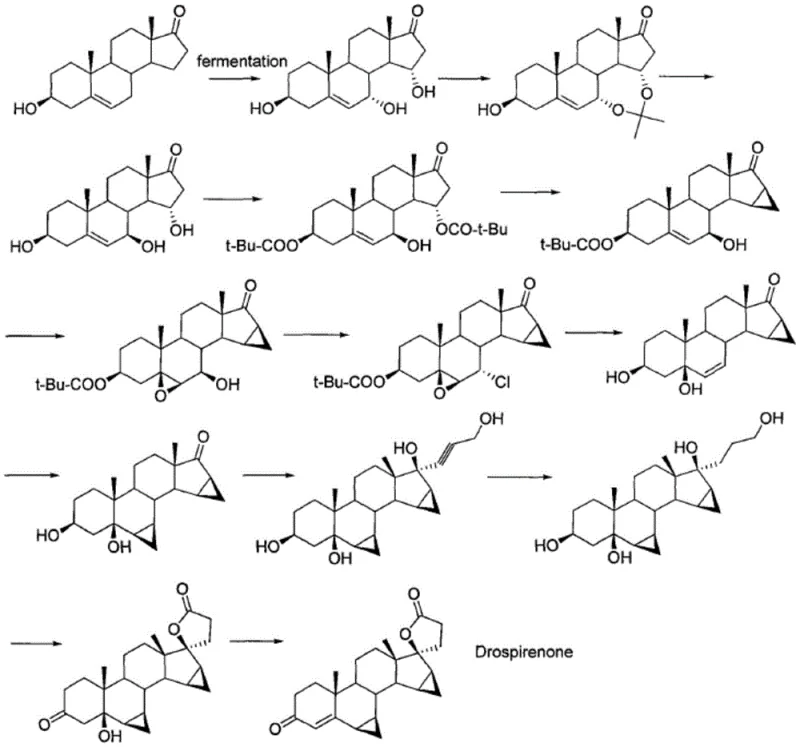
The pharmaceutical industry is constantly seeking more sustainable and cost-effective pathways for synthesizing complex hormonal active pharmaceutical ingredients (APIs). Patent CN110407905B, published in September 2021, introduces a groundbreaking preparation method for Drospirenone and its key intermediates. This technology addresses critical bottlenecks in existing manufacturing processes by replacing hazardous heavy metal oxidants with a benign hydrogen peroxide system. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for one of the world's most successful hormonal contraceptives. The patent details a robust four-step sequence that begins with a commercially available bishydroxy cyclopropyl compound and culminates in the formation of the distinctive spiro-lactone ring characteristic of Drospirenone.
The core innovation lies in the oxidative cyclization step, which traditionally relied on toxic chromium or expensive palladium catalysts. By shifting to a green chemistry approach using hydrogen peroxide, the inventors have created a pathway that is not only environmentally superior but also economically advantageous. This report analyzes the technical feasibility, mechanistic advantages, and commercial implications of adopting this novel synthesis route. As a reliable drospirenone supplier, understanding these underlying technological shifts is crucial for maintaining competitive advantage in the global market for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Drospirenone has been plagued by reliance on hazardous reagents and complex purification requirements. Early methods, such as those disclosed in European patent EP 0075189, necessitated the use of carbon tetrachloride, a solvent now recognized for its severe toxicity and environmental persistence. Furthermore, the oxidation steps in these legacy routes frequently employed chromium trioxide or ruthenium salts. Chromium (VI) compounds are known carcinogens, posing severe occupational health risks and generating toxic waste streams that require expensive remediation. The presence of heavy metal residues in the final API is a major regulatory concern, often necessitating additional, yield-reducing purification steps to meet ICH Q3D guidelines for elemental impurities.
Other approaches, such as those found in patent CN101503455, attempted to mitigate toxicity by using palladium catalysts. However, palladium is a precious metal with volatile pricing and supply chain constraints. The cost of removing trace palladium from the final product to acceptable ppm levels adds significant operational expenditure (OPEX). Additionally, some prior art routes suffered from low yields in the critical spiro-ring formation step or required harsh acidic conditions that led to epimerization and ring-opening byproducts. These inefficiencies result in poor atom economy and increased production costs, making them less attractive for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology described in CN110407905B offers a decisive break from these limitations by utilizing hydrogen peroxide as the sole oxidant for the construction of the spiro-lactone ring. This reagent is inexpensive, readily available in bulk quantities, and produces water as the only byproduct, aligning perfectly with green chemistry principles. The process operates under mild conditions, typically between 30°C and 50°C, which reduces energy consumption compared to high-temperature alternatives. By eliminating heavy metals like chromium and ruthenium, the new route simplifies the downstream processing workflow. There is no need for complex metal scavenging resins or extensive washing protocols to remove toxic residues, which directly translates to higher throughput and reduced cycle times.
Furthermore, the starting materials for this route, specifically the bishydroxy cyclopropyl compound (Compound 1), are commercially accessible and relatively inexpensive. The use of conventional solvents such as tetrahydrofuran, acetone, and N,N-dimethylformamide ensures that the process can be easily implemented in existing multipurpose reactors without requiring specialized corrosion-resistant equipment needed for highly acidic or halogenated environments. This accessibility makes the novel approach highly suitable for the commercial scale-up of complex steroid intermediates, offering a clear path to industrial mass production with improved safety profiles and lower total cost of ownership.
Mechanistic Insights into Hydrogen Peroxide Oxidative Cyclization
The centerpiece of this patent is the transformation of the diol intermediate (Compound 3) into the spiro-lactone (Compound 4). Mechanistically, this involves the selective oxidation of the primary alcohol and the tertiary alcohol functionalities to form the five-membered lactone ring fused to the steroid backbone. In the presence of hydrogen peroxide and a suitable solvent system, the reaction proceeds through a nucleophilic attack of the hydroperoxide anion or a similar activated oxygen species on the carbonyl or activated alcohol centers. The patent specifies that the reaction can be effectively conducted in amide-based solvents like DMF, ketone-based solvents like acetone, or mixtures thereof. The choice of solvent plays a critical role in stabilizing the transition state and ensuring the solubility of the steroidal substrate.
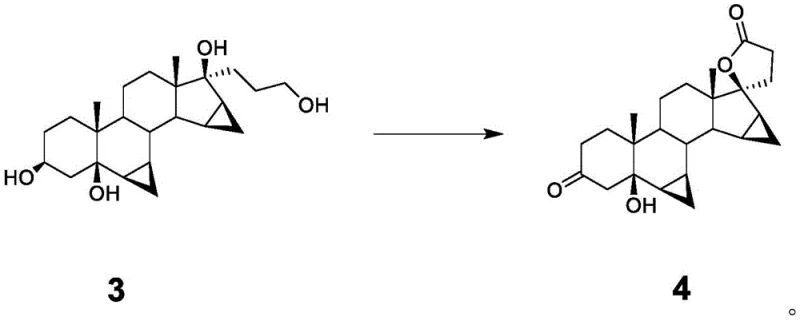
One of the most remarkable aspects of this mechanism is its chemoselectivity. Despite the presence of other sensitive functional groups within the complex steroid framework, such as the cyclopropane rings and the existing ketone at position 3, the hydrogen peroxide system selectively targets the 17-alpha side chain for lactonization. This specificity minimizes the formation of over-oxidized byproducts or degradation of the steroid nucleus. The patent data indicates that controlling the molar ratio of hydrogen peroxide to Compound 3 is vital; ratios between 34.8 and 81.2 are preferred to drive the reaction to completion without causing excessive decomposition. The reaction progress is easily monitored via TLC or HPLC, with the disappearance of Compound 3 serving as the endpoint indicator, typically achieved within 5 to 6 hours.
From an impurity control perspective, this mechanism is superior because it avoids the radical pathways often associated with metal-catalyzed oxidations, which can lead to unpredictable side reactions. The absence of transition metals also means there is no risk of metal-induced catalysis of unwanted rearrangements during the workup. The resulting Compound 4 is obtained with high chemical purity (reported up to 99.0% in examples), which significantly eases the burden on the final crystallization step. This high level of purity is essential for ensuring the quality of the final Drospirenone API, as impurities in the intermediate can propagate through the synthesis and become difficult to remove in later stages.
How to Synthesize Drospirenone Efficiently
The synthesis of Drospirenone via this patented route involves a logical sequence of addition, reduction, oxidation, and dehydration steps. The process begins with the nucleophilic addition of propiolic alcohol to the ketone group of the starting steroid, followed by catalytic hydrogenation to saturate the alkyne. The critical oxidation step then installs the spiro-lactone moiety, and a final acid-catalyzed dehydration introduces the requisite double bond in the A-ring. Each step has been optimized for yield and operational simplicity, utilizing standard laboratory and plant equipment. The detailed standardized synthesis steps, including specific reagent quantities, temperature profiles, and workup procedures, are outlined in the guide below.
- Perform an addition reaction between the bishydroxy cyclopropyl starting material (Compound 1) and propiolic alcohol using an organic base like potassium methoxide in tetrahydrofuran to generate the alkynyl intermediate (Compound 2).
- Execute a catalytic hydrogenation reduction of Compound 2 using a palladium-on-carbon (Pd/C) catalyst in an alcohol solvent to saturate the triple bond, yielding the diol precursor (Compound 3).
- Conduct the critical oxidation step by reacting Compound 3 with hydrogen peroxide in a mixed solvent system (e.g., DMF and acetone) at controlled temperatures to form the spiro-lactone ring (Compound 4).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN110407905B offers tangible strategic benefits beyond mere technical novelty. The primary advantage is the drastic simplification of the supply chain for raw materials. By relying on commodity chemicals like hydrogen peroxide and common organic solvents, manufacturers are no longer held hostage by the volatility of the precious metals market or the regulatory restrictions surrounding hexavalent chromium. This shift enhances supply chain reliability, ensuring that production schedules are not disrupted by the scarcity of specialized catalysts. Furthermore, the use of non-toxic reagents simplifies the logistics of hazardous material handling and storage, reducing insurance premiums and compliance overheads.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts such as palladium and ruthenium directly lowers the bill of materials (BOM) cost. More importantly, it removes the need for costly metal scavenging steps and the associated loss of product yield during purification. The high yields reported in the patent examples (e.g., 94.4% for the oxidation step) mean that less starting material is wasted, improving the overall mass balance of the process. Additionally, the reduced toxicity of the waste stream lowers the cost of environmental compliance and waste disposal, contributing to substantial cost savings in pharmaceutical intermediates manufacturing over the long term.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for specialized chiral catalysts or toxic oxidants creates a single point of failure in the supply chain. In contrast, hydrogen peroxide and the solvents used in this process are produced by multiple global vendors, ensuring a robust and competitive supply base. This diversification mitigates the risk of supply interruptions due to geopolitical issues or plant outages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply of high-purity hormonal contraceptives to the market.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on heavy metal residues in APIs, processes that inherently avoid these contaminants provide a significant compliance buffer. This green chemistry approach facilitates easier regulatory filings and approvals, reducing lead time for high-purity hormonal contraceptives to reach the market. The process is designed for industrial mass production, with reaction parameters that are easily scalable from pilot plant to multi-ton commercial reactors. The absence of corrosive or highly toxic reagents also extends the lifespan of production equipment, reducing capital expenditure on maintenance and replacement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within patent CN110407905B, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: Why is the hydrogen peroxide oxidation method superior to traditional chromium-based routes for Drospirenone?
A: Traditional routes often utilize chromium trioxide or ruthenium salts, which are highly toxic, carcinogenic, and create significant hazardous waste disposal challenges. The patented hydrogen peroxide method eliminates heavy metals entirely, drastically reducing environmental toxicity and simplifying the purification process to meet stringent pharmaceutical residual metal limits.
Q: What are the typical yields and purity levels achievable with this new synthetic route?
A: According to the experimental data in patent CN110407905B, the oxidation step converting Compound 3 to Compound 4 achieves yields exceeding 94% with chemical purity reaching 99.0%. The final dehydration step to Drospirenone maintains high efficiency, with overall process yields demonstrating strong viability for industrial mass production.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for scalability. It utilizes conventional, commercially available reagents like hydrogen peroxide and standard solvents (DMF, acetone, THF) rather than expensive or unstable specialty catalysts. The reaction conditions operate at moderate temperatures (30-50°C) and normal pressure, making it safe and cost-effective for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Drospirenone Supplier
The technological advancements detailed in patent CN110407905B underscore the evolving landscape of steroid synthesis, where efficiency and sustainability are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this green oxidation process, ensuring that we can deliver Drospirenone and its intermediates with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the absence of heavy metals and ensure that every batch meets the highest international pharmacopoeia standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this heavy-metal-free process. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials against your internal benchmarks. Together, we can drive down costs and enhance the sustainability of your hormonal contraceptive portfolio.
